Originally published on Ultrasound G.E.L. on 8/31/20 – Visit HERE to listen to accompanying PODCAST! Reposted with permission.
Follow Dr. Michael Prats, MD (@PratsEM) from Ultrasound G.E.L. team!
Early Screening for Aortic Dissection With Point-of-Care Ultrasound by Emergency Physicians
J Ultrasound Med July 2020 – Pubmed Link
Take Home Points
1. In a high risk population, POCUS findings of aortic dissection can be very specific.
2. POCUS can decrease time to diagnosis, but this may not translate into downstream patient-centered benefits in this population.
Background
We have talked about aortic dissection before. It is often considered, often missed, often deadly. Most of the time you need to get a CT to make this diagnosis, but it can show up on POCUS. Here’s what we know: we know that it is feasible to get these views (often including the aortic arch) in the emergency department. We also suspect that incorporating POCUS might decrease time to diagnosis, but much of the existing literature is of moderate quality suffering from retrospective collection and issues of patient selection. The last study we discussed showed a tremendous sensitivity in low-risk patients if you combine POCUS with a d-dimer (100%) . If you find direct signs of a dissection (like a flap), POCUS is also very specific. In the paper at hand, the authors want to look at a high-risk population, scrap the d-dimer and see how well POCUS alone performs compared to CT in patients suspected of having this disease process. They also look at a number of interesting patient-centered outcomes including time to diagnosis, time to CT, time to treatment, and mortality.
Check out more on the prior evidence of POCUS for Aortic Emergencies in the Evidence Atlas!
Questions
What is the accuracy of point-of-care ultrasound evaluation for acute aortic dissection?
What is the time delay caused by point-of-care ultrasound evaluation for acute aortic dissection?
Population
Single center in China, data collected in 2016
Inclusion:
- Adults ≥18
- Presented to the emergency department with suspicion for aortic dissection (high risk based on concerning historical or physical exam findings)
Exclusion:
- Refused the ultrasound
- Trauma with chest pain
- Cardiac arrest
Design
Prospective nonrandomized convenience sample
Patients who presented meeting inclusion criteria were placed into the POCUS group or control group based on the presence or absence of an ultrasound-trained physician.
After the initial medical contact, if the POCUS providers were present, they were called to do the exam. They were blinded to history or exam, but knew that the patient was suspected of having an aortic dissection. They were not the treating provider. The treating provider was blinded to the POCUS. POCUS was done prior to CTA.
Only patients in the POCUS group received an ultrasound. All patients received ecg, labs, and CT angiography (CTA) of the aorta.
Primary outcomes was the sensitivity and specificity of POCUS compared to CTA.
They also recorded time to diagnosis (ended when “definitive diagnosis” was achieved), CT completion (when CT exam ended), initiation of treatment (when medication for blood pressure or heart rate was started) as well as in-hospital and 3 month mortality to compare between groups.
Divided into subgroups of Stanford A or B aortic dissections.
Who did the ultrasounds?
2 senior emergency medicine residents
They received a 1 week didactic and hands on training in ultrasound (not just aorta but cardiac, abdominal, and vascular). Did a minimum of 20 scans of each type.
The Scan
Phased array transducer

They stated that the bedside scan was performed according to the “A-F” mnemonic described in this letter to the editor from 2014. Aorta, Both Ventricles, Contractility, Dimensions (of aorta and chambers), Effusion, Further abnormalities
This current article does not reference what cardiac views were done, but it seems to follow the FATE (Focused Assessment Transthoracic Echocardiography) protocol with includes the four standard focused echo views.
Thoracic
- 4 views of heart (parasternal, subcostal, apical)
- Suprasternal notch view
- Abdominal Aorta (not further specified)
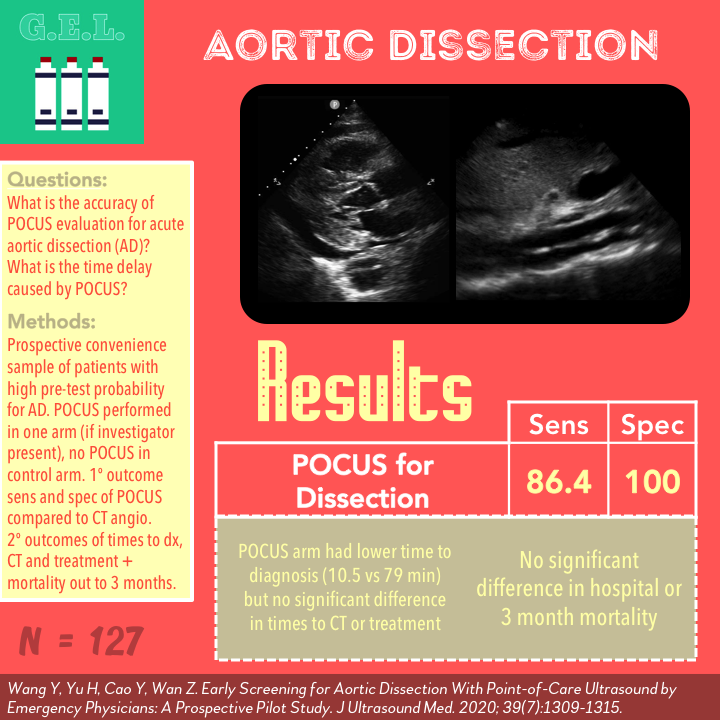
Diagnosis of Aortic Dissection = seeing a “flap”
- Undulating motion concordant with pulsatile blood flow (independent of excursion of the aortic wall)
- Seen in at least 2 planes
- “Clear distinction” from a reverberation of surrounding tissue
Learn how to do Ultrasound for Aortic Dissection from 5 Minute Sono!
Check out Aortic Dissection Pathology on the POCUS Atlas!
Results
N = 127
- 72 POCUS
- 55 control
- No significant differences between groups (both were ~70% male)
- Aortic dissection was in 61.1% of POCUS group (n=44). Appears not to report incidence in the entire population.
Now realize that for the accuracy outcomes, they only looked at the POCUS arm (n = 72). For the patient centered outcome time interval, they looked at all patients (n = 127) to compare the POCUS and control arms.
Primary Outcome – Diagnostic Accuracy for Aortic Dissection
All patients
Sensitivity 86.4% (CI 72.0-94.3)
Specificity 100% (CI 85.0-100%)
Secondary Outcomes Diagnostic Accuracy by Type
Type A (ascending)
Sensitivity 90.9% (CI 69.4-98.4)
Specificity 100% (CI 91.1-100)
Type B (distal to left subclavian)
Sensitivity 81.9% (CI 59.0-94.0)
Specificity 100% (CI 91.1-100)
Summary: overall 100% specific, less sensitive seemingly because it missed more Type B dissections.
Time Intervals
Door to Diagnosis (p <0.01)
POCUS 10.5 min (IQR 5.0-34.0)
Control 79.0 min (IQR 40-115)
Door to CT (p 0.84)
POCUS 63.0 (IQR 32.0-116.5)
Control 66.0 (IQR 38.0-93.0)
Door to Treatment (p 0.55)
POCUS 49.0 (IQR 7-102.0)
Control 53.0 (IQR 24.0-140.8)
Mortality
In hospital (p 0.25)
- POCUS 4.2%
- Control 9.1%
Within 3 Months (p <0.99)
- POCUS 25.0%
- Control 20.8%
Summary: Only significant difference between arms was a shorter time to diagnosis in POCUS arm by 70 minutes. This did not lead to faster CT, treatment, or improved mortality. Note that door to treatment times are faster than door to CT reflecting that treatment was initiated prior to CT in many cases in both arms.
Other findings
Door to US Time was 9 minutes (IQR 4.0-15.0)
Time for US exam was 5 minutes (IQR 4.0-6.8)
0 false positives
6 false negatives (not discussed)
Strengths
Prospective
Control arm
Reasonable # of patients
Risk stratified
Patient centered outcomes
Limitations
Convenience sampling. Single center with seemingly high prevalence of aortic dissections. They also don’t report BMI which limits some of the generalizability. Part of the problem is that the “high risk” criteria for inclusion are not better defined.
The study was likely underpowered to detect differences in a lot of the main outcomes (mortality for example).
Time to treatment was not broken down between type A and type B. Would type A dissections go to surgical repair faster if the diagnosis was known sooner? I guess it is possible, but was not reported on here.
CTA gives more information, not just accuracy of diagnosis. This means that even if POCUS gets you the information more quickly. CT might still be needed and would could negate any time savings.
Discussion
So why did a time savings in making the diagnosis not translate to any other improvements? It could be that the POCUS diagnosis was not trusted, but I think that there is a more likely explanation. If you look at the times it took to get a CT (~1 hour) and start meds (~50 minutes), this might be the fastest these actions can occur in most centers. Remember this “door” time starts at time of emergency department arrival. Even if you knew the diagnosis immediately and there was no delay in placing the orders, it still takes time to get the patient triaged, get vitals, move to a room, history, physical, order tests, get to CT, get medications drawn up and administered. So perhaps what this study tells us is that the time to diagnosis does not lead to a significant delay, because people are going to get things moving either way in these high risk cases. Perhaps POCUS, therefore, would have a great impact in patients in whom you have a low suspicion for aortic dissection. In these patients, we are probably less likely to begin diagnostics/treatment empirically and therefore, POCUS might expedite that process by allowing for an earlier diagnosis. A similar finding was seen in an article on evaluating hydronephrosis in nephrolithiasis. POCUS had the largest impact on those who were low risk, then found to have severe hydronephrosis.
It is interesting to compare the results here to the aforementioned 2019 European Heart Journal study. The 2019 study was way larger (n = 839), but they had a different research question and population. They were attempting to see if POCUS, in combination with a risk-statification tool and a d-dimer, was sensitive enough to rule out acute aortic emergencies. The short answer is that yes – it was perfectly sensitive in their study. In the 2019 study, there was a 17.4% prevalence of acute aortic emergencies (which included dissections, ulcers, hematomas, ruptures). The current 2020 study had dissections in 61.1% of their POCUS arm – a huge portion, supporting that this is a higher risk population. The 2019 study noted that only 10% of the population with aortic emergency had direct signs (meaning seeing a flap). The sensitivity was kept high by including “indirect signs” such as pericardial effusion or increased aortic diameter. For direct signs only, the studies were in agreement – very specific (2019: 97.4%, 2020: 100%). For sensitivity of direct signs only, the 2020 study was actually surprisingly more sensitive (2019: 45.2%, 2020: 86.4%). This could reflect the differences in the definitions for positive findings or perhaps just the different populations. If the current study had included indirect signs it might have been more sensitive. Both studies showed that POCUS was more sensitive for type A dissections (perhaps because the parasternal and suprasternal views can visualize aorta better than transabdominal?). The 2019 study showed that in the higher-risk subpopulation, POCUS was actually less specific. This study somewhat refutes that finding. Regardless, the data from both of these studies is overall consistent. POCUS findings of a dissection flap are specific for the disease.
It is unfortunate that this study did not show that this actually changes any of the important patient centered outcomes measured. The next studies will have to carefully consider how this diagnostic test could best be used to benefit patients. Is this another application that could help pre-hospital or no-CT environments?
Summary
This prospective nonrandomized observational study from a Chinese emergency department evaluated 127 patients who were at high risk for aortic dissection. They found a sensitivity of 86.4% and a specificity of 100% compared to CT angiography. Although there was a 70 minute faster time to diagnosis in the POCUS group, this did not translate into any other measured benefits.
Take Home Points
1. In a high risk population, POCUS findings of aortic dissection can be very specific.
2. POCUS can decrease time to diagnosis, but this may not translate into downstream patient-centered benefits in this population.
More Great FOAMed on this Topic
Retroperitoneal Ultrasound (Aorta/Kidneys) with Claire Heslop on The Ultrasound Podcast
Our score

Expert Reviewer for this Post

Joseph Minardi, MD @jminardi21
Chief, Division of Emergency and Clinical Ultrasound, West Virginia University Departments of Emergency Medicine and Medical Education
Reviewer’s Comments
Well done review of this article. I would add that it’s possible, maybe even likely, that no reductions in times were seen due to the patient selection in these cases. These cases were already identified as high risk prior to POCUS and on the dissection CTA train. All of these patients were also hemodynamically stable. POCUS is unlikely to decrease times or impact these patients. POCUS may have a more important role in the lower or moderate risk patients, where it could prompt further imaging that may not have necessarily been planned. Also, in some cases, a POCUS, then direct to treatment strategy may be more effective in reducing time to treatment, and possibly patient centered outcomes. There may be a greater impact of POCUS in less stable patients where CTA is not feasible.
I’m obviously a proponent of liberal echo in patients with chest pain to aid in risk stratification and identification of unexpected findings. I think the literature supports this approach (although, admittedly, not all high quality evidence), and I have seen the impact for patients in my care and in my group’s practice.
Cite this post as
Michael Prats. Return of the Aortic Dissection – POCUS Accuracy and Time. Ultrasound G.E.L. Podcast Blog. Published on August 31, 2020. Accessed on June 24, 2022. Available at https://www.ultrasoundgel.org/97.