Authors: Brit Long, MD (@long_brit, EM physician at SAUSHEC) and Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital) // Edited by: Jennifer Robertson, MD, MSEd
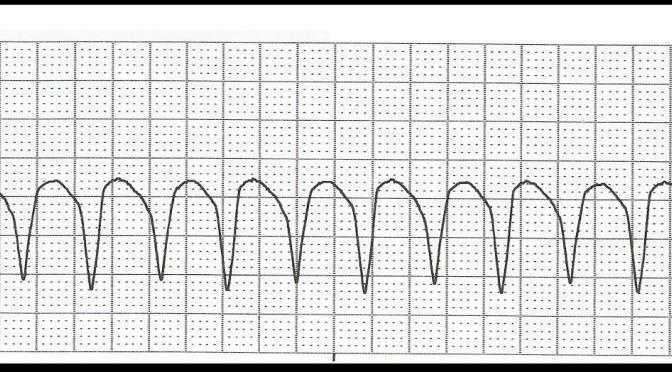
You are five minutes into your shift, and the charge nurse asks you to immediately see a patient who was just roomed. The patient is a 62-year-old male with a history of coronary artery disease (CAD), hypertension (HTN), hyperlipidemia (HLD) and two prior myocardial infarctions (MIs). He complains of lightheadedness, but otherwise feels well without chest pain or dyspnea. He is actually not sure why he was rushed back so quickly. The technician hands you an electrocardiogram (ECG) which displays a wide complex, monomorphic rhythm at 134 beats per minute (bpm). With the exception of a rapid heart rate, the patient’s vital signs are unremarkable, with a blood pressure (BP) of 144/70 and a respiratory rate (RR) of 17 breaths per minute.
He does not show any signs of hemodynamic instability, and his ECG is consistent with monomorphic ventricular tachycardia. What are the best treatment options for this patient?
Background
Ventricular tachycardia (VT) and ventricular fibrillation (VF) cause approximately 300,000 deaths per year in the United States (US) and account for 5.6% of all mortality.1 VT is considered the most common wide complex tachycardia.2-7 Risk factors for VT include known cardiac ischemia, male gender, and older age. Underlying causes of VT include ischemic heart disease, structural heart disease (cardiomyopathy), congenital heart disease, channelopathies, electrolyte derangements, sympathomimetic agents, digitalis toxicity, and infiltrative cardiomyopathy.2-6,8
Wide complex tachycardias are defined by a rate greater than 100 bpm and a QRS complex duration greater than 120 milliseconds (ms). The differential diagnosis of wide complex tachycardia includes supraventricular tachycardia with aberrant ventricular conduction, preexcited tachycardia in patients with Wolff-Parkinson-White syndrome, ventricular tachycardia, toxic/metabolic derangement, pacemaker-related WCT, and artifact.3-6 VT accounts for 80% of wide complex tachycardias, SVT with aberrant conduction accounts for the other, approximately 20% of cases.9,10
The rhythm of VT originates distal to the bundle of His in the ventricular myocardium. Actual sites of origin may include the ventricular myocardium, distal conduction system beyond the bundle of His, or both. VT is associated with ventricular rates of > 120 bpm and QRS complex intervals > 120 ms.3-6 The majority of patients with VT possess an area of fibrotic tissue in the myocardium. 3-6,10 This scar usually results from prior myocardial infarction or ischemic cardiomyopathy, while a small minority of patients may have normal myocardium with no structural disease.10,11
The classification of ventricular tachycardia is based on several factors: ECG appearance, duration of VT, and most importantly, hemodynamic status of the patient.
– Monomorphic VT is a wide complex rhythm with a QRS duration greater than 120 ms. It originates from a single focus and is identical from beat to beat.
– Polymorphic VT varies from beat to beat.
– Non-sustained VT occurs for less than 30 seconds.
– Sustained VT lasts longer than 30 seconds.3-5,12
– A patient with VT who is unstable demonstrates evidence of hemodynamic compromise including hypotension, altered mental status, chest pain, or heart failure but is awake with a pulse. If the patient is unresponsive or pulseless, then cardiac arrest is present.
– A stable patient with VT shows no signs of hemodynamic compromise.3-6,13
The medical management of ventricular tachycardia is controversial, specifically for stable, monomorphic VT. Multiple options exist, and expert recommendations have undergone multiple modifications.
Current VT Management Guidelines
The 2000 American Heart Association (AHA) guidelines for Advanced Cardiac Life Support (ACLS) recommended procainamide or sotalol (IIa) over the use of amiodarone or lidocaine (IIb) in the setting of stable, monomorphic VT with preserved ejection fraction (EF).13
In patients with decreased EF, amiodarone and lidocaine received IIb recommendations.
The American College of Cardiology (ACC)/AHA/European Society of Cardiology (ESC) 2006 modifications gave amiodarone a IIa recommendation for stable VT resistant to procainamide.14,15 Per the AHA Advanced Cardiovascular Support (ACLS) algorithm, amiodarone is the only anti-dysrhythmic mentioned for use in patients with VT (class IIb).15 However, the AHA still recommended procainamide in patients who have preserved EF and a normal QT interval (class IIa). Lidocaine was also recommended as a second line agent (class indeterminate). The AHA notes sotalol can be given but only with expert consultation.15 The European resuscitation guidelines also recommend amiodarone for treatment of stable, monomorphic VT.16
In 2010, further modifications were made by the AHA, where sotalol was given a class IIb recommendation.17 The 2015 update did not change the recommendations for procainamide and amiodarone.18
Despite these recommendations, direct-current cardioversion is the most effective therapy, supported by numerous studies.4,6,18-20 If possible, this should be the first-line treatment.
Management of Unstable VT
The management of patients with unstable monomorphic or polymorphic VT requires immediate synchronized direct-current cardioversion. If recurrent VT occurs, continue to electrically cardiovert. In addition, a patient with shock resistant unstable VT should receive amiodarone 300 mg IV with a second bolus of 150 mg IV.4,6,15,17,18
Management of Stable VT
Hemodynamically stable patients should be approached by first evaluating whether the VT is monomorphic or polymorphic. If the rhythm is polymorphic with normal QT interval, treatment is the same as monomorphic VT. Prolonged QT with polymorphic VT is concerning for Torsades de pointes. Magnesium sulfate, isoproterenol, pacing, or a combination of these treatments is required. Avoid procainamide and amiodarone in this setting, which can prolong the QT interval further.21
Several medications can be used for the management of stable, monomorphic VT. This post will evaluate several of these medications. Of note, we will not address suppression of stable VT, but rather termination of the dysrhythmia.
Lidocaine
Lidocaine is a class Ib antidysrhythmic. The medication antagonizes fast voltage-gated sodium channels, preventing depolarization and action potential transmission. Lidocaine is dosed at 1 to 1.5 mg/kg IV initially and repeated at 0.5 to 0.75 mg/kg every 5 to 10 minutes. Lidocaine can have numerous side effects that may affect the cardiovascular, gastrointestinal and central nervous systems. Hypotension, thrombophlebitis at the infusion site, tinnitus, dyspnea, bronchospasm, and allergic reactions can also occur from lidocaine therapy.4,6,14-120,22,23
Amiodarone
Amiodarone is a class III antidysrhythmic that prolongs phase 3 of the cardiac action potential. The medication has other antidysrhythmic effects, similar to classes Ia, II, and IV. The mechanism of this medication is complex: it increases the refractory period of sodium and potassium channels and slows intracardiac action potentials, similar to beta blockers and calcium channel blockers. Dosing is 150 mg IV over 10 minutes, followed by 1 mg/minute for 6 hours and 0.5mg/minute for at least 18 hours. Contraindications include prior hypersensitivity, severe sinus node dysfunction, second and third degree heart block, cardiogenic shock, and syncope from heart block. The medication affects multiple organs and organ systems: pulmonary (interstitial lung disease, pulmonary fibrosis), thyroid (hypothyroidism and hyperthyroidism), ophthalmologic (optic neuropathy, keratopathy, vision changes), liver (jaundice, hepatitis, hepatomegaly), skin (discoloration), neuropathy, gynecomastia, and epididymitis. 4,6,14-18,20,24
Procainamide
Procainamide is a class Ia antidysrhythmic that induces a rapid block of cardiac sodium channels. The blockage of sodium channels occurs intracellularly and extracellularly. Procainamide slows conduction velocity and increases the refractory period. Classically, the medication is dosed at 15 to 18 mg/kg IV as a slow infusion over 25 to 30 minutes, but the 2015 ACLS guidelines recommend a max dose of 17 mg/kg.18 Other studies have since modified this dosing regimen, which is discussed below. Several options are present for rate of infusion: either 20 to 50 mg/minute or 100 mg every 5 minutes may be given until resolution of VT, hypotension or QRS widening by 50% of its original length occurs. The infusion should also be stopped once a total dose of 17 mg/kg IV is reached. Several side effects can occur while using procainamide, including worsening dysrhythmia, bradycardia, hypotension, drug fever, allergy, blood dyscrasias, and systemic lupus erythematosus. 4,6,14-20,25
Sotalol
Sotalol is a class III antidysrhythmic that targets potassium channels and delays ventricular relaxation. This increases the amount of time before another signal can be generated in the myocytes. The medication is also a non-selective competitive beta-adrenergic receptor blocker, decreasing production of cyclic AMP and the activation of calcium channels.26,27 Dosing is 1.5 mg/kg IV over 5 minutes. Contraindications include sick sinus syndrome, long QT syndrome, shock, asthma, potassium less than 4 meq/L, and poorly controlled heart failure. Side effects include weakness, dizziness, headache, bradycardia, palpitations, nausea/vomiting, and headache.4,6,27-30
On the horizon? Nifekalant
A medication recently developed and only currently used in Japan is nifekalant, a class III antidysrhythmic that selectively blocks potassium channels, prolonging the refractory period. It has no intrinsic β-blocking capability.31-34 This medication is thought to have rapid onset of action, rapid clearance and rapid reduction of the defibrillation threshold, while having no negative inotropic effects. Nifekalant is dosed at 0.3-0.6 mg/kg IV, followed by an infusion rate of 0.15-0.50 mg/kg/hr IV. This medication has demonstrated efficacy in the termination and suppression of VT in multiple studies. Rates of successful VT termination with utilization of this medication range from 48.4% to 80%.31-39 This medication may also improve the defibrillation ability of direct current. Studies have also been conducted for ventricular fibrillation, shock-resistant ventricular fibrillation, and hemodynamically unstable ventricular tachycardia. It has not been evaluated against procainamide. Adverse effects include QT interval prolongation and Torsades de pointes.31-39
Some Literature…
The majority of literature evaluates VT suppression, rather than VT termination. The studies evaluating the use of antidysrhythmic termination of VT suffer from significant heterogeneity in exclusion criteria, medications studied, and outcomes.40-46
Ho et al. 1994: a randomized prospective trial including 33 patients with VT based on ECG criteria.41
– Investigators compared sotalol 100mg IV over 5 minutes and lignocaine (otherwise known as lidocaine) 100 mg IV over 5 minutes. 41
– Outcome was VT termination in 15 minutes or hemodynamic deterioration.
– Sotalol was more effective at VT termination, 69% versus 18% (95% confidence interval for absolute difference of 51%, 22-80%, p = 0.003).
– One patient in each group required electrical cardioversion. The authors concluded sotalol was more effective.40,41
Gorgels et al 1996: a randomized prospective study of 29 patients with VT based on ECG diagnosis.42 Investigators excluded patients with acute myocardial infarction and hemodynamic instability.
– Primary outcome consisted of VT termination in 15 minutes.
– The medications studied were procainamide 10mg/kg IV at 100 mg/min and lidocaine 1.5 mg/kg IV over 2 minutes.
– Procainamide resulted in VT termination in 12 of 15 patients, while lidocaine only terminated 3 of 14. Procainamide also stopped VT in 8 of 11 patients in which lidocaine was ineffective.40,42
Manz et al. 1988: a randomized, prospective trial. VT diagnosis was based on ECG or electrophysiological confirmation of VT.43
– Primary outcome was VT termination.
– Investigators compared ajmaline 50-75 mg IV over 3-5 minutes and lidocaine 100-200 mg IV over 3-5 minutes.43
– Ajmaline terminated VT in 10 of 15 patients, but lidocaine only terminated VT in 2 of 16 patients, bringing into question the efficacy of lidocaine.
– Both medications were well tolerated.40,43
Marill et al 2010: a retrospective, observational trial, unlike the prior studies. The investigators compared amiodarone and procainamide, with inclusion of 83 patients on ECG criteria.44
– Primary outcome was VT termination within 20 minutes.
– Amiodarone was dosed at 150 mg IV with a minimum rate of 10 mg/min, and procainamide was dosed at 500 mg IV at a minimum rate of 15 mg/min.
– Rates of VT termination were 25% and 30% for amiodarone and procainamide, respectively, with an adjusted odds of termination of 1.2 (95% confidence interval [CI] =0.4 to 3.6) when comparing procainamide to amiodarone.
– When comparing the two agents as initial agents alone, termination occurred in 8/34 patients receiving amiodarone (24%, 95% CI = 11% to 41%) and in 4/7 patients with procainamide (57%, 95% CI 18% to 90%).
–– Of the patients receiving infusion, 35 of 66 amiodarone patients (53%, 95% CI=40 to 65%) and 13 of 31 procainamide patients (42%, 95% CI=25 to 61%) required electrical therapy.40,44
Komura et al. 2010: included 90 patients based on ECG criteria, comparing procainamide and lidocaine.45 No patients with chest pain or acute myocardial infarction were included.45
– Outcome was termination of VT or hemodynamic deterioration.
– Procainamide was dosed at 100 mg IV every 1 to 2 minutes to a maximum of 800 mg IV versus lidocaine at 50 mg IV to a maximum of 150 mg IV.
– Medications were discontinued if VT was terminated, maximum dosage was reached, or if the patient experienced hemodynamic decompensation.
– VT was stopped in 53/70 patients receiving procainamide (75.7%) and in 7/20 patients receiving lidocaine (35.0%).
– Four of the patients who did not convert to normal sinus rhythm with lidocaine did convert with procainamide.
– QRS prolongation was seen with procainamide, but not with lidocaine.40,45
Ortiz et al. PROCAMIO study: just released this year, included 62 patients and compared procainamide with amiodarone.46
– Primary outcome was the presence of major cardiac adverse event (MACE) within 40 minutes after infusion initiation. MACE was defined by clinical signs of hypoperfusion, signs of heart failure, hypotension (systolic blood pressure ≤70 mmHg if the pre-treatment systolic pressure was ≤100 mmHg or systolic blood pressure ≤80 mmHg if the pre-treatment systolic pressure was > 100 mmHg), increase in HR of > 20 bpm with medication, and appearance of fast polymorphic VT.
– Fifteen patients (24%) experienced a MACE within 40 minutes of infusion discontinuation, though fewer events were found in patients receiving procainamide vs. amiodarone (9% vs. 41%, OR 0.1; 95% CI 0.03-0.6). Over a 24-hour period, adverse events occurred in 18% and 31% of the procainamide and amiodarone patients, respectively.
– VT termination occurred in 67% of procainamide patients and 38% of amiodarone patients within 20 minutes of infusion initiation.
– Among 49 patients with structural heart disease, MACE occurred in 11% procainamide and 43% amiodarone patients.46
These studies are summarized in the table below. If you’re interested in further evaluation of these studies, please see the “Deep Dive” section at the end of this post.
Table 1 – Comparison of Outcomes40-46
| Study | Sample Size | Medications | Outcome | Relative Risk (95% CI) | Number Needed to Treat (NNT) (95% CI) |
| Ho et al. | 33 | Lidocaine 100mg IV vs. Sotalol 100 mg IV | Lidocaine 18% vs. Sotalol 69% | 3.9 (1.3 to 11.5) | 2.0 (1.2 to 4.5) |
| Gorgels et al. | 29 | Lidocaine 1.5 mg/kg IV vs. Procainamide 10 mg/kg IV | Lidocaine 21% vs. Procainamide 80% | 3.7 (1.3 to 10.5) | 1.7 (1.1 to 3.4) |
| Manz et al. | 31 | Lidocaine 100 mg IV vs. Ajmaline 50 mg IV | Lidocaine 13% vs. Ajmaline 67% | 5.3 (1.4 to 20.5) | 1.9 (1.2 to 3.9) |
| Marill et al. | 41 | Procainamide 500 mg IV vs. Amiodarone 150 mg IV | Procainamide 57% vs. Amiodarone 24% | 4.3 (0.8 to 23.6) | 3.0 (-17.5 to 1.4) |
| Komura et al. | 90 | Lidocaine 50 mg IV up to 150 mg vs. Procainamide in 100 mg IV every 1-2 minutes (up to 800 mg) | Lidocaine 35% vs. Procainamide 76% | 2.2 (1.2 to 4.0) | 2.5 (1.6 to 5.7) |
| Ortiz et al. | 62 | Procainamide 10mg/kg IV over 20 minutes vs. Amiodarone 5mg/kg IV over 20 minutes | Procainamide 67% vs. Amiodarone 38% | 1.8 (1.04 to 3.0) | 3.5 (1.9 to 20.4) |
VT Management Pearls
If possible, cardioversion should be performed. This is the most effective treatment, supported by guidelines and the literature. If medications are utilized, physicians should carefully evaluate the patient’s hemodynamic status. If the patient becomes unstable at any point, direct current cardioversion is needed.4,6,18-20,24
In the U.S., if medical management is selected, procainamide provides the greatest efficacy and lowest risk of adverse outcomes. Guidelines for infusion recommend an infusion rate of 20-50 mg/min IV;13-18 however, infusion time at this rate can take 30 minutes to reach adequate dosing. The results from Gorgels et al. and Komura et al. suggest that rates of 50-100 mg/min IV are efficacious and safe, while decreasing the time for needed monitoring.40,42,45
If the patient is experiencing acute ischemia or myocardial infarction, direct current cardioversion is needed. If medical management is selected, amiodarone or lidocaine may be more efficacious, but the poor literature support prevents the authors of this manuscript to recommend one medication over another.
Figure 1 – Stable Monomorphic VT Algorithm
Procainamide Pearls
Procainamide IV infusion requires monitoring of blood pressure and ECG. The QRS complex duration should be carefully analyzed before and during the infusion. Stop points of infusion include dysrhythmia discontinuation, hypotension, QRS prolongation greater than 50% of the original duration, a total of 1 gram is infused, or acceleration of the tachycardia. The most feasible dosing of procainamide includes a maximum dose of 10 mg/kg (maximum 1 g) IV at 100 mg/min over 10 minutes. This approach allows rapid loading of procainamide, compared to ACLS infusion of 20 mg/min until a maximum dose of 17 mg/kg is provided.
Summary
– Stable, monomorphic ventricular tachycardia is defined by a rate faster than 120 beats/min with QRS greater than 120 ms.
– Hemodynamically unstable VT requires immediate synchronized direct current cardioversion.
– Medical management of hemodynamically stable monomorphic VT is controversial. Direct current cardioversion is most efficacious.
– Guidelines for the treatment of VT from the AHA provide a IIa recommendation for procainamide, compared to a IIb recommendation for both amiodarone and sotalol.
– Studies evaluating procainamide, lidocaine, amiodarone, and sotalol suffer from poor design, difference in inclusion and exclusion criteria, small sample size, and outcome determination.
– PROCAMIO demonstrates procainamide’s efficacy.
– Procainamide demonstrates the greatest efficacy. If procainamide is selected, a maximum dose of 10 mg/kg (maximum 1 g IV) at 100 mg/min over 10 minutes should be provided with monitoring of blood pressure and ECG.
Ready for a Deep Dive?
All of the mentioned studies above (Ho et al., Gorgels et al., Manz et al., Marrill et al., Komura et al., and Ortiz et al.) used ECG for initial diagnosis of VT. These studies differed in whether patients with acute myocardial infarction (MI) were included.40-46 Gorgels et al., Ortiz et al., and Komura et al. did not include patients with MI,42,45,46 whereas the others did.41,43,44 All of these studies except one (Manz et al.) limited evaluation to spontaneous VT.40-46
Lidocaine was the most commonly studied medication, with similar dosing. Two of the studies evaluated procainamide and lidocaine,42,45 with two studies comparing procainamide and amiodarone.44 Dosing and rate of infusion of procainamide differed between studies, with Marrill et al. providing procainamide at maximum 500mg infusion.44 Komura et al. and Gorgels et al. used higher dosing ranges with fast infusions.42,45 These two studies found a NNT of approximately two for terminating VT with procainamide compared to lidocaine.42,45 Ortiz et al. utilized 10 mg/kg IV over 20 minutes, with a NNT of 3.5 when procainamide was compared to amiodarone.46 Four studies used a specified time of termination (15 to 20 minutes), and if termination of VT did not occur in this time, the medication was deemed unsuccessful.41,42,44,46 In terms of adverse effects from these studies, death occurred in four patients, one from a large myocardial infarction after termination of VT with sotalol. Hypotension occurred in a wide range, as Ortiz et al. found hypotension occurred in 24% of patients receiving procainamide and 48% of patients receiving amiodarone.46 Neurologic symptoms such as dizziness, paresthesias, headache, visual problems, and hearing changes occurred in approximately 16% of patients receiving lidocaine infusion.40-46
The current literature has few prospective trials with small sample sizes, as well as retrospective observational studies with selection bias and multiple confounders. The PROCAMIO study conducted by Ortiz et al., which is prospective and randomized, provides some of the strongest evidence supporting procainamide.46 A greater amount of literature exists evaluating the use of medications for suppression of VT. This post did not cover these studies. Another important aspect is the inclusion of patients with MI. Studies evaluating lidocaine and procainamide did not evaluate patients with acute MI, where lidocaine may block sodium channels in ischemic myocardium more effectively. These studies also did not calculate sample sizes needed for outcomes, and it is difficult to determine whether patients were properly randomized.40-46
References/Further Reading:
- Chugh SS, Jui J, Gunson K, Stecker EC, John BT, Thompson B, et al. Current burden of sudden cardiac death: multiple source surveillance versus retrospective death certificate-based review in a large U.S. community. J Am Coll Cardiol 2004 Sep 15. 44(6):1268-75.
- Stewart RB, Bardy GH, Greene HL. Wide complex tachycardia: misdiagnosis and outcome after emergent therapy. Ann Intern Med 1986;104:766.
- Akhtar M, Shenasa M, Jazayeri M, et al. Wide QRS complex tachycardia. Reappraisal of a common clinical problem. Ann Intern Med 1988;109:905.
- Gupta AK, Thakur RK. Wide QRS complex tachycardias. Med Clin North Am 2001;85:245.
- Steinman RT, Herrera C, Schluger CD, et al. Wide QRS tachycardia in the conscious adult: Ventricular tachycardia is the most frequent cause. JAMA 1989;261:1013-1016.
- Delbridge TR, Yealy DM. Wide complex tachycardia. Emerg Med Clin North Am 1996;13:902-924.
- Herbert ME, Votey SR, Morgan MT, et al. Failure to agree on the electrocardiographic diagnosis of ventricular tachycardia. Ann Emerg Med 1996;271:35-38.
- Brugada J, Brugada R, Brugada P. Channelopathies: a new category of diseases causing sudden death. Herz 2007 May;32(3):185-91.
- Baerman JM, Morady F, DiCarlo LA, et al. Differentiation of ventricular tachycardia from supraventricular tachycardia with aberration: Value of the clinical history. Ann Emerg Med 1987;16:40-43.
- Josephson, ME. Electrophysiology of Ventricular Tachycardia: An Historical Perspective. J Cardiovasc Electrophysiol 2003;14:1134-1148.
- Stevenson WG. Catheter ablation of monomorphic ventricular tachycardia. Curr Opin Cardiol 2005;20(1):42-47.
- Morady F, Baerman JM, DiCarlo LA Jr, et al. A prevalent misconception regarding wide-complex tachycardias. JAMA 1985;254:2790.
- The American Heart Association in collaboration with the International Liaison Committee on Resuscitation. Guidelines 2000 for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Part 6: advanced cardiovascular life support: section 7: algorithm approach to ACLS emergencies: section 7A: principles and practice of ACLS. Circulation 2000;102(8 Suppl):I136–9.
- ACC/AHA/ESC. 2006 Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death: A Report of the American College of Cardiology/American Heart Association Task Force and the European Society of Cardiology Committee for Practice Guidelines (Writing Committee to Develop Guidelines for Management of Patients With Ventricular Arrhythmias and the Prevention of Sudden Cardiac Death). J Am Coll Card 2006;48:e247–346.
- The American Heart Association in collaboration with the International Liaison Committee on Resuscitation. American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Part 7.3: Management of Symptomatic Bradycardia and Tachycardia. Circulation 2005;112:IV-67–77.
- Nolan JP, Soar J, Zideman DA, et al. European Resuscitation Council Guidelines for Resuscitation 2010 Section 1. Executive summary. Resuscitation 2010;81:1219–76.
- Neumar RW, Otto CW, Link MS, et al. Part 8: adult advanced cardiovascular life support: 2010 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2010;122(Suppl 3): S729–67.
- Link MS, Berkow LC, Kudenchuk PJ, Halperin HR, Hess EP, Moitra VK, et al. Part 7: adult advanced cardiovascular life support: 2015 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care. Circulation 2015;132(Suppl 2): S444-S464.
- Lown B. Electrical reversion of cardiac arrhythmias. Br Heart J 1967;29:469–89.
- Atkins DL, Dorian P, Gonzalez ER, et al. Treatment of tachyarrhythmias. Ann Emerg Med 2001;37:S91–109.
- Passman R, Kadish A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med Clin North Am 2001;85(2):321-341.
- Carterall WA. Molecular mechanisms of gating and drug block of sodium channels” Sodium Channels and Neuronal Hyperexcitability. Novartis Foundation Symposia 2001;241:206–225.
- Sheu SS, Lederer WJ. Lidocaine’s negative inotropic and antiarrhythmic actions. Dependence on shortening of action potential duration and reduction of intracellular sodium activity. Circulation Research 1985 Oct;57(4):578–90.
- Tomlinson DR, Cherian P, Betts TR, et al. Intravenous amiodarone for the pharmacological termination of haemodynamically-tolerated sustained ventricular tachycardia: is bolus dose amiodarone an appropriate first-line treatment? Emerg Med J 2008;25:15–18.
- Zamponi GW, Sui X, Codding PW, French RJ. Dual actions of procainamide on batrachotoxin-activated sodium channels: open channel block and prevention of inactivation. Biophys J 1993;65(6):2324-2334
- Bertrix, Lucien et al. Protection against ventricular and atrial fibrillation by sotalol. Cardiovascular Research 1986;20:358-363.
- Edvardsson, N et al. Sotalol-induced delayed ventricular repolarization in man. European Heart Journal 1980;1:335-343.
- Antonaccio M, Gomoll A. Pharmacologic basis of the antiarrhythmic and hemodynamic effects of sotalol. Am J Cardiol 1993;72:27A-37A.
- Charnet P, et al. cAMP-dependent phosphorylation of the cardiac L-type Ca channel: A missing link? Biochimie 1995;77:957–962.
- Kassotis, J et al. Beta receptor blockade potentiates the antiarrhythmic actions of d-sotalol on re-entrant ventricular tachycardia in a canine model of myocardial infarction. Journal of Cardiovascular Electrophysiology 2003;14:1233-1244
- Naitoh N, Tagawa M, Yamaura M, Taneda K, Furushima H, Aizawa Y. Comparison of electrophysiologic effects of intravenous E-4031 and MS-551, novel class III antiarrhythmic agents, in patients with ventricular tachyarrhythmias. Jpn Heart J 1998; 39:457–467.
- Washizuka T, Chinushi M, Watanabe H, Hosaka Y, Komura S, Sugiura H, et al. Nifekalant hydrochloride suppresses severe electrical storm in patients with malignant ventricular tachyarrhythmias. Circ J 2005;69:1508–1513.
- Yusu S, Ikeda T, Mera H, Miyakoshi M, Miwa Y, Abe A, et al. Effects of intravenous nifekalant as a lifesaving drug for severe ventricular tachyarrhythmias complicating acute coronary syndrome. Circ J 2009;73:2021–2028.
- Pantazopoulos IN, Troupis GT, Pantazopoulos CN, Xanthos TT. Nifekalant in the treatment of life-threatening ventricular tachyarrhythmias. World Journal of Cardiology 2011;3(6):169-176.
- Sakurada H, Kobayashi Y, Sugi K, Katagiri T, Baba T, Enjoji Y, et al. Efficacy of intravenous doses of MS-551 for sustained ventricular tachycardia. J Clin Therap Med 1997;13:1773–1787.
- Aonuma K, Hiroe M, Nishimura S, Marumo F. Efficacy of intravenous doses of MS-551 for ventricular tachycardia after myocardial infarction. J Clin Therap Med 1997;13:1789–797.
- Katoh T, Tsunoo M, Mitsuhashi T, Atarashi H, Ino T, Kuroki S, et al. Phase I study of MS-551 (1)–A single intravenous injection study. J Clin Therap Med 1997;13:1659 – 1674.
- Katoh T, Mitamura H, Matsuda N, Takano T, Ogawa S, Kasanuki H. Emergency treatment with nifekalant, a novel class III anti-arrhythmic agent, for life-threatening refractory ventricular tachyarrhythmias: post-marketing special investigation. Circ J 2005 Oct;69(10):1237-43.
- Nakagawa K, Nakamura K, Kusano KF, et al. Use of Intravenous Amiodarone in the Treatment of Nifekalant-Resistant Arrhythmia: A Review of 11 Consecutive Cases with Severe Heart Failure. Pharmaceuticals 2011;4(6):794-803.
- deSouza IS, Martindale JL, Sinert R. Antidysrhythmic drug therapy for the termination of stable, monomorphic ventricular tachycardia: a systematic review. Emerg Med J 2015;32: 161–167.
- Ho DSW, Zecchin RP, Richards DAB, et al. Double-blind trial of lignocaine versus sotalol for acute termination of spontaneous sustained ventricular tachycardia. Lancet 1994;344:18–23.
- Gorgels AP, van den Dool A, Hofs A, et al. Comparison of procainamide and lidocaine in terminating sustained monomorphic ventricular tachycardia. Am J Cardiol Jul 1 1996;78:43–6.
- Manz M, Luderitz B. Emergency treatment of ventricular tachycardias: Ajmaline and lidocaine compared. Deutsche Medizinische Wochenschrift 1988;113:1317–21.
- Marill KA, deSouza IS, Nishijima DK, et al. Amiodarone or procainamide for the termination of sustained stable ventricular tachycardia: An historical multicenter comparison. Acad Emerg Med 2010;17:297–306.
- Komura S, Chinushi M, Furushima H, et al. Efficacy of procainamide and lidocaine in terminating sustained monomorphic ventricular tachycardia. Circ J May 2010;74:864–9.
- Ortiz M, Martin A, Arribas F, et a; PROCAMIO Study Investigators. Randomized comparison of intravenous procainamide vs. intravenous amiodarone for the acute treatment of tolerated wide QRS tachycardia: the PROCAMIO study. Eur Heart J 2016 Jun 28. pii: ehw230. [Epub ahead of print]







2 thoughts on “Stable Monomorphic Ventricular Tachycardia Management in the ED”
Pingback: #FOAMed Highlights
Pingback: emDOCs.net – Emergency Medicine EducationVentricular Tachycardia Mimics - emDOCs.net - Emergency Medicine Education