Author: Juliette Conte, MD (EM Resident Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case
A 73-year-old male with a history of hypertension, coronary artery disease, and peripheral vascular disease presents with fever and right-upper quadrant pain over the past 6 hours. He denies nausea/vomiting, diarrhea, recent travel, and history of abdominal surgeries. He has never experienced pain like this before.
Triage VS: T 101.3F, HR 112, BP 143/87, RR 18, SpO2 100% on room air. Exam reveals an ill-appearing gentleman in obvious distress. His exam is otherwise remarkable for right upper quadrant tenderness with a positive Murphy’s sign, normal bowel sounds, and no rebound tenderness or guarding.
You walk out of the room feeling pretty confident that you’ve nailed the diagnosis. You order a right upper quadrant ultrasound (RUQUS).
His labs are remarkable for a white blood cell count of 14, with normal liver aminotransferases and a normal total bilirubin. His labs are otherwise unremarkable. The RUQUS shows a thickened anterior gallbladder wall (4.5mm) with pericholecystic fluid, but no gallstones or common bile duct dilation. You have administered fluids and analgesics, but the patient still appears toxic. With how sick the patient is, you order a CT Abdomen/Pelvis, which reveals pericholecystic fluid and gallbladder wall subserosal edema, but no evidence of abscess or perforation.
Background
Acute acalculous cholecystitis (AAC) is defined as inflammation of the gallbladder without evidence of gallbladder calculi1,2. Though relatively uncommon amongst the spectrum of biliary disease, AAC accounts for approximately 5-10% of cases of acute cholecystitis and has an incidence of 0.2-0.4% in critically ill patients1,3,4. The annual incidence of AAC has been reported ranging from 7-22% in adults5.
AAC is commonly associated with critically ill patients in the ICU, i.e. polytrauma patients, massive burns, cardiac surgery, abdominal vascular surgery, immunosuppression, sepsis, prolonged TPN administration6. The proposed common pathophysiology among many of these conditions is gallbladder ischemia from hypoperfusion. Hypoperfusion is often multifactorial, secondary to splanchnic vasoconstriction from hypotension or vasopressors, atherosclerotic vascular disease, and increased gallbladder pressure from biliary stasis6–8. Biliary stasis is also thought to render the gallbladder mucosa more susceptible to injury6. Factors that contribute to biliary stasis in critically ill patients include sepsis, TPN, fasting, mechanical ventilation with positive end-expiratory pressure, and opioid analgesia causing ileus6.
Despite this longstanding association, AAC may have a higher prevalence in the outpatient setting. Literature evaluating AAC in this setting is extremely limited. A single retrospective review study at Yale demonstrated that of 2346 patients diagnosed with AAC over a 7-year period, 77% had developed AAC as outpatients without acute inciting illness or trauma4, suggesting that perhaps the pre-existing association with ICU patients represents only a minority of patients. Another study evaluating all cases of acute cholecystitis over a 4-year period in South Korea found that 90% of cases of AAC developed in outpatients with no acute illness7. Further studies are necessary to further investigate whether the incidence of AAC is truly higher in outpatients than previously estimated.
Risk Factors
Risk factors for AAC in the outpatient setting include atherosclerotic cardiovascular disease4,7 and age greater than 60 (average age 64.5 years)7,9. In contrast to calculous cholecystitis which occurs more commonly in women, male sex was shown to have a stronger association with acalculous cholecystitis in multiple studies7,9–12. Another study evaluating AAC in postoperative patients found 80% of patients with post-operative of AAC were male 6. In the Savoca study of outpatients with AAC, 83% had significant comorbidities, most commonly cardiovascular disease (72%) , alcoholic liver disease (15%), COPD, (15%) and peptic ulcer disease (4%)4. The Ryu et al. study conducted in South Korea had a lower rate of comorbidities, with patients having risk factors of hypertension (25%), diabetes (20%), and chronic renal failure (10%)7. There is limited data evaluating the prevalence of these risk factors in other populations.
Risk factors for AAC in the inpatient, critically ill adult population are numerous and are listed in Table 1.

Clinical Presentation
AAC is nearly clinically indistinguishable from calculous cholecystitis. The most common findings include right upper quadrant pain, fever, and leukocytosis. Less common findings in AAC include nausea, vomiting, biliary colic, elevated LFTs, alkaline phosphatase, total bilirubin. In the Savoca study of 47 outpatients who presented with AAC, the most common presenting signs and symptoms were leukocytosis (93%), right upper quadrant pain (83%), and fever (55%). In this patient population, other symptoms were less common, i.e. jaundice (4%) and nausea and vomiting (6%)4.
In critically ill patients and sedated patients, it can be difficult to elicit any specific symptomatology, and a common presentation is sepsis of unknown origin.
Evaluation
As with calculous cholecystitis, ultrasound is the primary imaging modality of choice in the evaluation of AAC. However, the sensitivity and specificity of ultrasound for AAC are not well established, and its primary role in the diagnostic algorithm of AAC remains debated.
Studies have reported sensitivities ranging from 30-92% and specificities from 89-100%1 in the ultrasonographic diagnosis of AAC. Of note, the study reporting a sensitivity of 92% incorporated both calculous and acalculous etiologies of acute cholecystitis13. This wide range in sensitivities is reflective of the relatively limited data on acalculous cholecystitis. The primary role of US in the evaluation of AAC may be largely due to its ease of performance at the bedside, its rapidity of performance, and its ability to be repeated without harm.
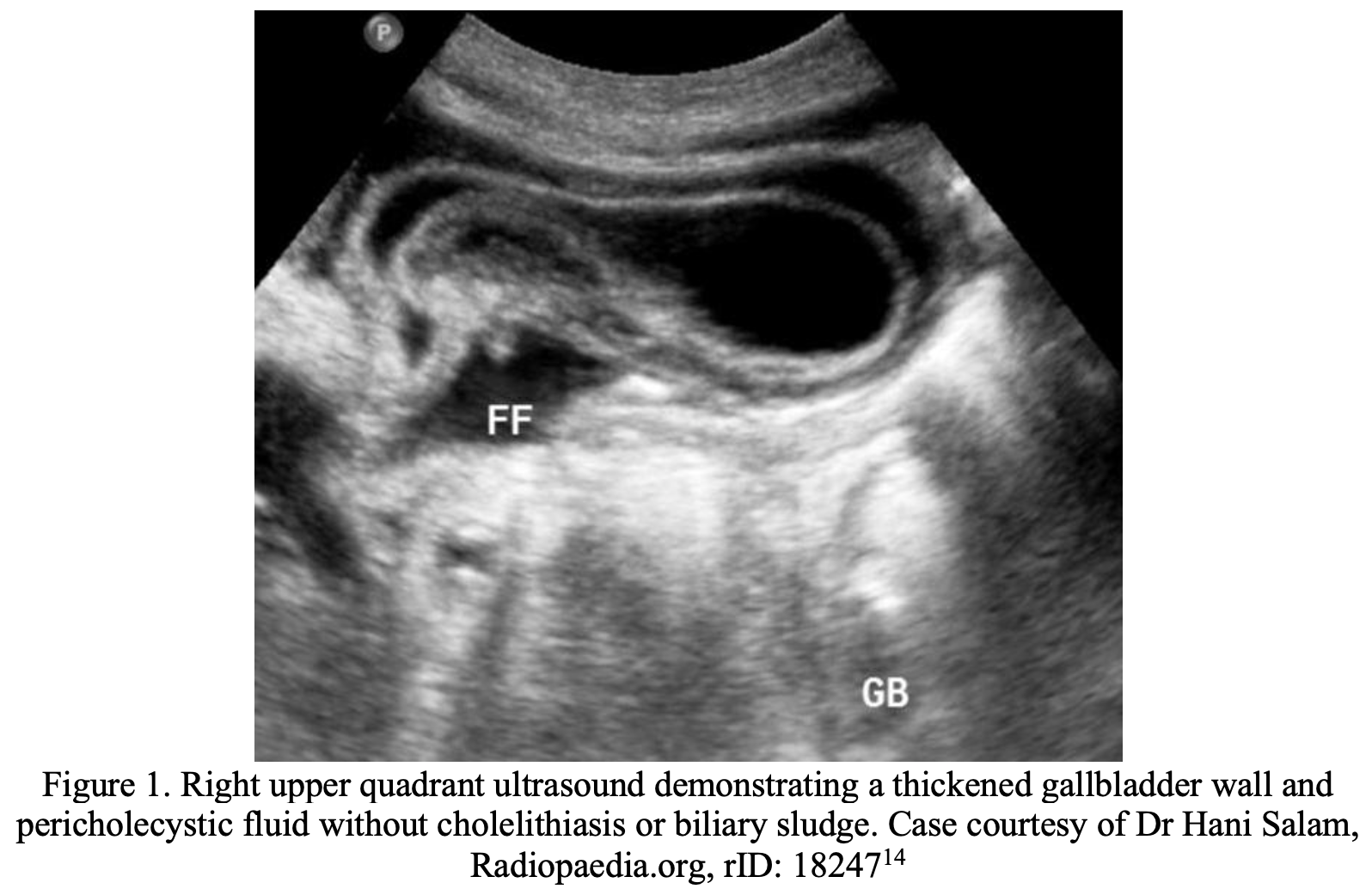
The proposed “diagnostic triad” of AAC is gallbladder wall thickness >3.5mm, biliary sludge, and gallbladder hydrops, defined as gallbladder distention >5cm in the transverse plane or >8cm in the longitudinal plane1. In the setting of ICU patients, these criteria have proven to be sensitive, but not specific, for the diagnosis of AAC, and ICU patients without AAC may manifest all three diagnostic criteria1. The sensitivity of this triad in the outpatient setting has yet to be reported. Other findings include pericholecystic fluid and a gallbladder halo sign, indicated by irregular, discontinuous, alternating echogenic bands, which are indicative of subserosal edema and necrosis2,13.


In the setting of a high clinical suspicion for AAC, the diagnostic algorithm for evaluation of AAC if ultrasound is equivocal/negative is not well established. Further imaging with CT scan or nuclear imaging should be pursued.
CT Scan
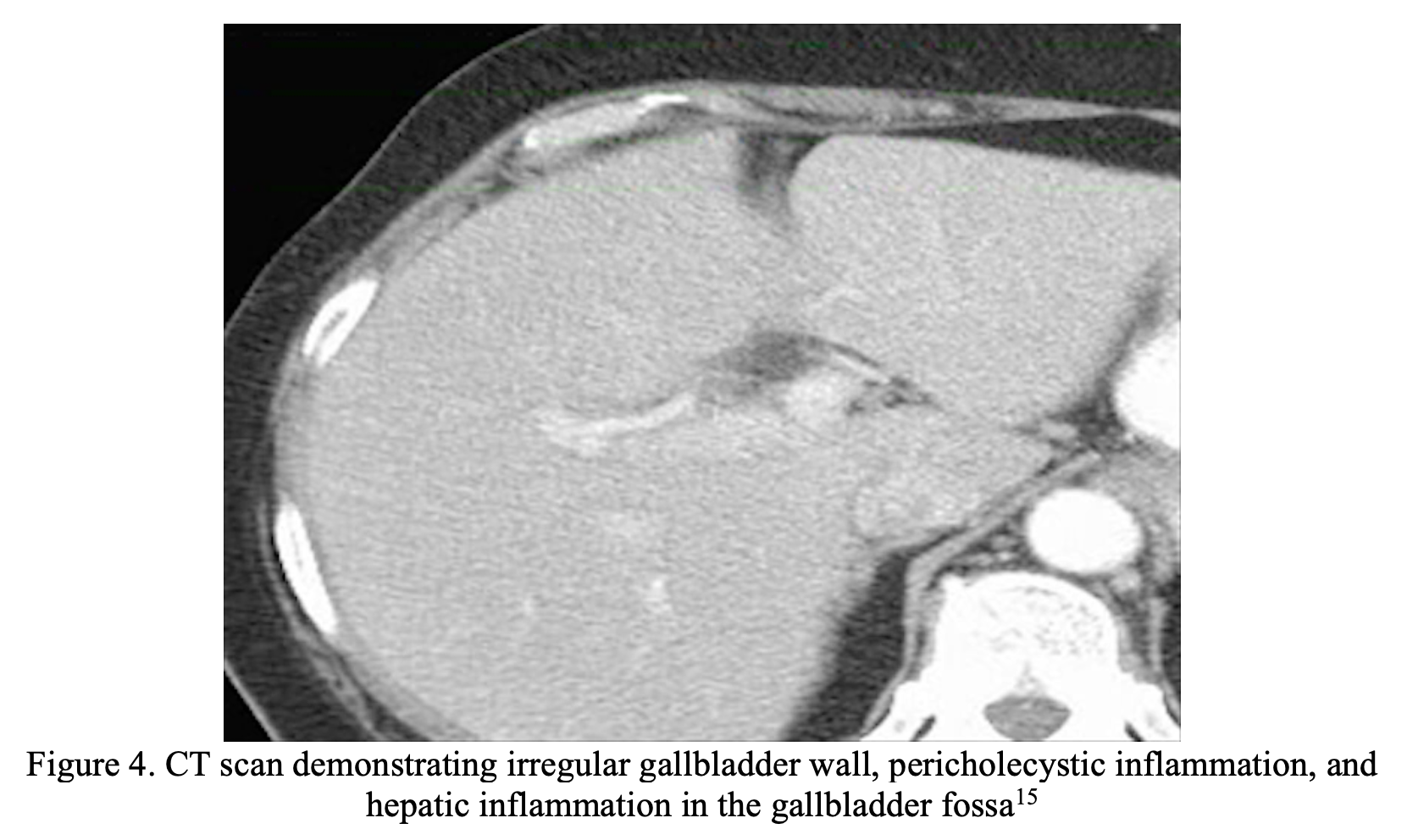
CT scan has little benefit over US in the evaluation for AAC, however is often employed to investigate alternative intra-abdominal processes and can demonstrate findings suggestive of AAC. CT scan may show findings similar to those in acute calculous cholecystitis, including gallbladder wall thickening (>3mm) or nodularity, poor definition of gallbladder/liver interface, and pericholecystic fluid16. However, findings are often nonspecific. In one retrospective study of trauma patients with multisystem injury with clinical suspicion for AAC, 15 patients were found to have CT scans performed in the diagnostic evaluation of AAC, and CT scan was 100% sensitive and specific in this small sample size16. In a retrospective analysis of 127 intensive care unit patients diagnosed with AAC by CT scan (+/- US) found that higher bile density within the gallbladder and subserosal edema was had a sensitivity of 23.1% and specificity of 93.6%for an edematous gallbladder17.

Hepatobiliary iminodiacetic acid (HIDA) Scan
HIDA is a class of nuclear imaging studies that includes Radionuclide Cholescintigraphy (RC), Morphine Cholescintigraphy (MC) and cholecystokinin (CCK)-augmented HIDA. Though logistically more involved and more time-intensive, multiple studies recommend HIDA scan as the primary imaging modality for acalculous cholecystitis, as it evaluates gallbladder function rather than anatomy5,18,19. It is performed in a nuclear medicine suite by injecting a 99-Technectium labeled analogue of iminodiacetic acid into the bloodstream, which is taken up by the liver and concentrated in bile. A HIDA is considered positive for AAC if there is nonvisualization of the gallbladder 1 hour after injection of radiolabeled technetium (RC), or nonvisualization of the gallbladder 30 minutes after injection of morphine (after initial radiolabeled technetium) (MC)1. The sensitivity has been reported as 67-100% with a specificity of 38-100%1. Additionally, AAC diagnosed by HIDA has been reported after negative US and CT imaging20.
Diagnostic Laparoscopy
In the setting of negative ultrasound, CT scan and HIDA scan, a laparoscopy is recommended by the Society of American Gastroenterological and Endoscopic Surgeons21. Aspiration and culture of gallbladder contents are performed during diagnostic laparoscopy. Limited studies on diagnostic laparoscopy have reported a diagnostic accuracy of 90-100%1.
Initial Management
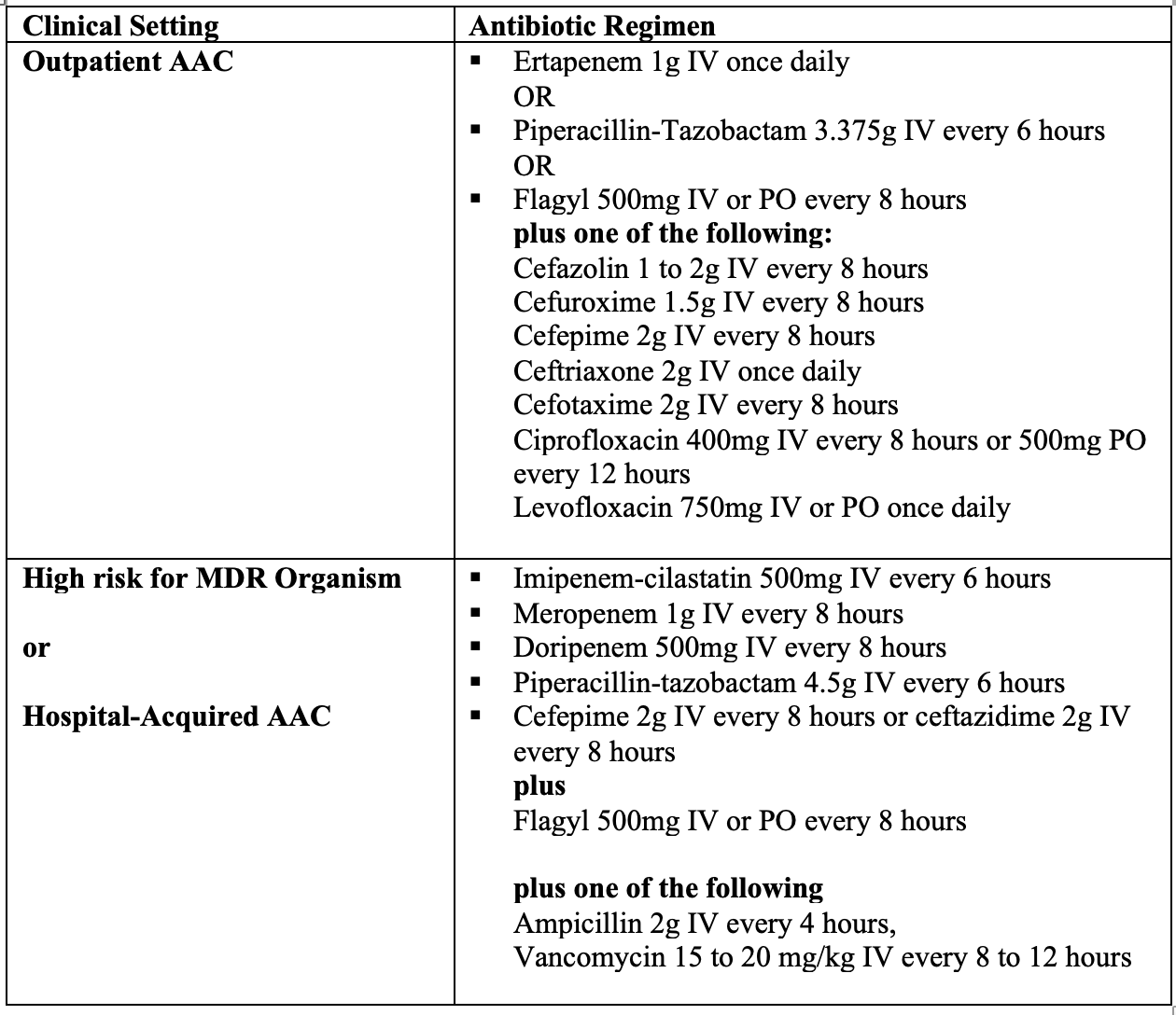
Initial management should include fluid resuscitation and systemic IV antibiotics. Antibiotic regimens should provide gram-negative coverage. Of note, while many recommend using vancomycin for hospital-acquired infections, in biliary disease, MRSA is a rare cause of infection. Rather, in the toxic-appearing patient who has previously received antibiotics, vancomycin is utilized to treat enterococcal infection22.
Treatment
The two mainstays of treatment for AAC are cholecystostomy and cholecystectomy2,8. For uncomplicated cases of AAC with no evidence of gangrene, perforation, or empyema, percutaneous cholecystostomy is preferred for decompression and drainage8,9. If the patient experiences improvement in symptoms, a cholecystectomy may not be necessary. For more critically ill patients, if there is evidence of significant gallbladder ischemia or necrosis, or persistent symptoms beyond 24-72hrs, cholecystectomy (laparoscopic vs open) is performed. Endoscopic retrograde cholangiopancreatographic GB drainage with stenting has been attempted without much success1,8.
Prognosis
ACC follows a more fulminant course than calculous cholecystitis and is associated with gangrene, perforation, empyema, and abdominal sepsis in approximately 40% of cases1, leading to significantly higher morbidity and mortality rates9. Mortality rates are reported as high as 67%, whereas that of acute calculous cholecystitis is approximately 3%4. Whether this higher complication rate is due to a more severe course of the disease versus delays in diagnosis remains debated.
Disposition
Patients are often critically ill, and with poor prognosis, will require admission for resuscitation and further therapy. IV antibiotics, cholescintigraphy, possible percutaneous cholecystostomy, and/or cholecystectomy are needed.
Key Points
- AAC may have a higher incidence in outpatients than hospitalized, critically ill patients. Maintain a high index of suspicion in patients who present to the ED with right upper quadrant pain even in the absence of a history of cholelithiasis.
- Men > 60 years old with atherosclerotic cardiovascular disease are the most common outpatient population to develop AAC.
- The sensitivity of ultrasound for acute acalculous cholecystitis is not well established. If you have a high clinical suspicion and a negative ultrasound, pursue further diagnostic imaging with HIDA scan +/- CT imaging, followed by admission for diagnostic laparoscopy if all noninvasive testing is negative.
- Acute acalculous cholecystitis follows a more fulminant course than calculous cholecystitis. Broad spectrum antibiotics with gram-negative coverage and fluid resuscitation should be started immediately if the diagnosis is suspected, with an emergent consultation to general surgery.
Case Resolution
The patient was admitted to the General Surgery service. His initial ultrasound was consistent with acute acalculous cholecystitis, given that he met 2 major diagnostic ultrasonographic criteria. His CT scan showed no findings concerning for empyema, gangrene or perforation, and his symptoms were present <24hrs, therefore he underwent placement of a percutaneous a percutaneous cholecystostomy tube by interventional radiology. His symptoms resolved within two weeks of cholecystostomy tube placement, and the tube was removed without complication and without subsequent cholecystectomy.
References/Further Reading
- Huffman JL, Schenker S. Acute Acalculous Cholecystitis: A Review. Clinical Gastroenterology and Hepatology. 2010:8(1):15-22.
- Babb RR. Acute Acalculous Cholecystitis. A Review. Journal of Clinical Gastroenterology. 1992:15(3):238-241.
- Jones MW, Ferguson T. Gallbladder, Cholecystitis, Acalculous. StatPearls Publishing: 2019.
- Savoca PE, Longo WE, Zucker KA, McMillen MM, Modlin IM. The increasing prevalence of acalculous cholecystitis in outpatients: Results of a 7-year study. Annals of Surgery. 1990:211(4):433-437.
- Kalliafas S, Ziegler DW, Flancbaum L, Choban PS. Acute acalculous cholecystitis: Incidence, risk factors, diagnosis, and outcome. American Surgeon. 1998:64(5):471-475.
- Barie PS, Fischer E. Acute acalculous cholecystitis. Journal of the American College of Surgeons. 1995:180(2):232-244.
- Ryu JK, Ryu KH, Kim KH. Clinical features of acute acalculous cholecystitis. Journal of clinical gastroenterology. 2003:36(2):166-169.
- Owen CC, Jain R. Acute Acalculous Cholecystitis. Current Treatment Options in Gastroenterology. 2005:8:99-104.
- Gu MG, Kim TN, Song J, Nam YJ, Lee JY, Park JS. Risk factors and therapeutic outcomes of acute acalculous cholecystitis. Digestion. 2014:90(2):75-80.
- Savoca PE, Longo WE, Zucker KA, McMillen MM, Modlin IM. The increasing prevalence of acalculous cholecystitis in outpatients: Results of a 7-year study. Annals of Surgery. 1990:211(4):433-437.
- Glenn F, Becker CG. Acute acalculous cholecystitis. An increasing entity. Annals of surgery. 1982:195(2):131-136.
- Johnson LB. The importance of early diagnosis of acute acalculus cholecystitis. Surgery Gynecology and Obstetrics. 1987:164(3):197-203.
- Marchal GJF, Casaer M, Baert AL, Goddeeris PG, Kerremans R, Fevery J. Gallbladder Wall Sonolucency in Acute Cholecystitis. Radiology. 1979:133(2):429-433.
- Di Muzio B, Weerakkody Y. Acute acalculous cholecystitis | Radiology Reference Article | Radiopaedia.org. Radiopaedia. https://radiopaedia.org/articles/acute-acalculous-cholecystitis?lang=us.
- Lane J. Acalculous Cholecystitis Imaging: Practice Essentials, Radiography, Computed Tomography. Medscape. https://emedicine.medscape.com/article/365553-overview. Published 2017.
- Mirvis SE, Vainright JR, Nelson AW, et al. The diagnosis of acute acalculous cholecystitis: A comparison of sonography, scintigraphy, and CT. American Journal of Roentgenology. 1986:147(6):1171-1175.
- Ahvenjarvi L, Koivukangas V, Jartti A, et al. Diagnostic accuracy of computed tomography imaging of surgically treated acute acalculous cholecystitis in critically ill patients. Journal of Trauma – Injury, Infection and Critical Care. 2011:70(1):183-188.
- Weissmann HS, Berkowitz D, Fox MS, et al. The role of technetium-99m iminodiacetic acid (IDA) cholescintigraphy in acute acalculous cholecystitis. Radiology. 1983:146(1):177-180.
- Swayne LC. Acute acalculous cholecystitis: sensitivity in detection using technetium-99m iminodiacetic acid cholescintigraphy. Radiology. 1986:160(1):33-38.
- Shafiq M, Zafar Y. Acute Acalculous Cholecystitis in the Setting of Negative Ultrasound and Computed Tomography Scan of the Abdomen. Cureus. 2018:10(2):2-7.
- Guidelines for Diagnostic Laparoscopy. Society of American Gastrointestinal and Endoscopic Surgeons. https://www.sages.org/publications/guidelines/guidelines-for-diagnostic-laparoscopy/. Published 2019.
- Solomkin JS, Mazuski JE, Bradley JS, et al. Diagnosis and management of complicated intra-abdominal infection in adults and children (IDSA guidelines). Clinical Infectious Diseases. 2010:50:133-164.