Author: David Cisewski, MD (@dhcisewski, EM Resident Physician, Icahn School of Medicine at Mount Sinai) // Reviewed by: Brit Long, MD (@long_brit) and Alex Koyfman, MD (@EMHighAK)

Background
Sore throat (aka, pharyngitis) is the 3rd most common complaint for physician visits with over 30 million cases diagnosed annually. Although the overwhelming majority of these cases are viral in origin (rhinovirus ~20%), the major focus of our attention in the ED stems around the question of “strep (GAS) vs not strep” and who gets antibiotics…. does anyone need antibiotics? Read on.
Why do we care if it’s strep (GAS) pharyngitis?
GAS is the most common bacterial cause of acute pharyngitis, resulting in 5%–15% of sore throat visits in adults and 20%–30% in children (Ebell, 2000). As opposed to a self-limiting viral pharyngitis requiring only symptomatic relief and a period of recovery, the popular dogma is that treating strep pharyngitis with a course of antibiotics is will hasten recovery and prevent the incidence of acute rheumatic fever (ARF)- and particularly, rheumatic heart disease (RHD) (Gerber, 2009). More on that to come.
Is everyone at risk of strep pharyngitis-induced ARF?
Not kids less than 3 years old. It is presumed that repeat GAS exposures are required to prime the immune system before an autoimmune response could result in anti-streptolysin O (ASO) antibodies and ARF. The immature immune systems of children <3 years of age are not primed for this capability, and thus the recommendations are to avoid treatment and workup for strep pharyngitis in this population.
If acute rheumatic fever is a concern, why not just treat everyone?
Beyond just antibiotic stewardship many fail to consider the adverse effect profile that comes with antibiotic use. In addition to nausea, vomiting, and diarrhea, unnecessary antibiotic use may lead to hypersensitivity reactions (SJS, TENS), anaphylaxis, imbalance of gut flora, and an increased rate of clostridium difficile infection. Consider for a moment that penicillin administration has an anaphylaxis incidence of 0.2% (1.2/10,000 injections), or a number needed to harm (NNH) of 500. Keep that number in the back of your mind.
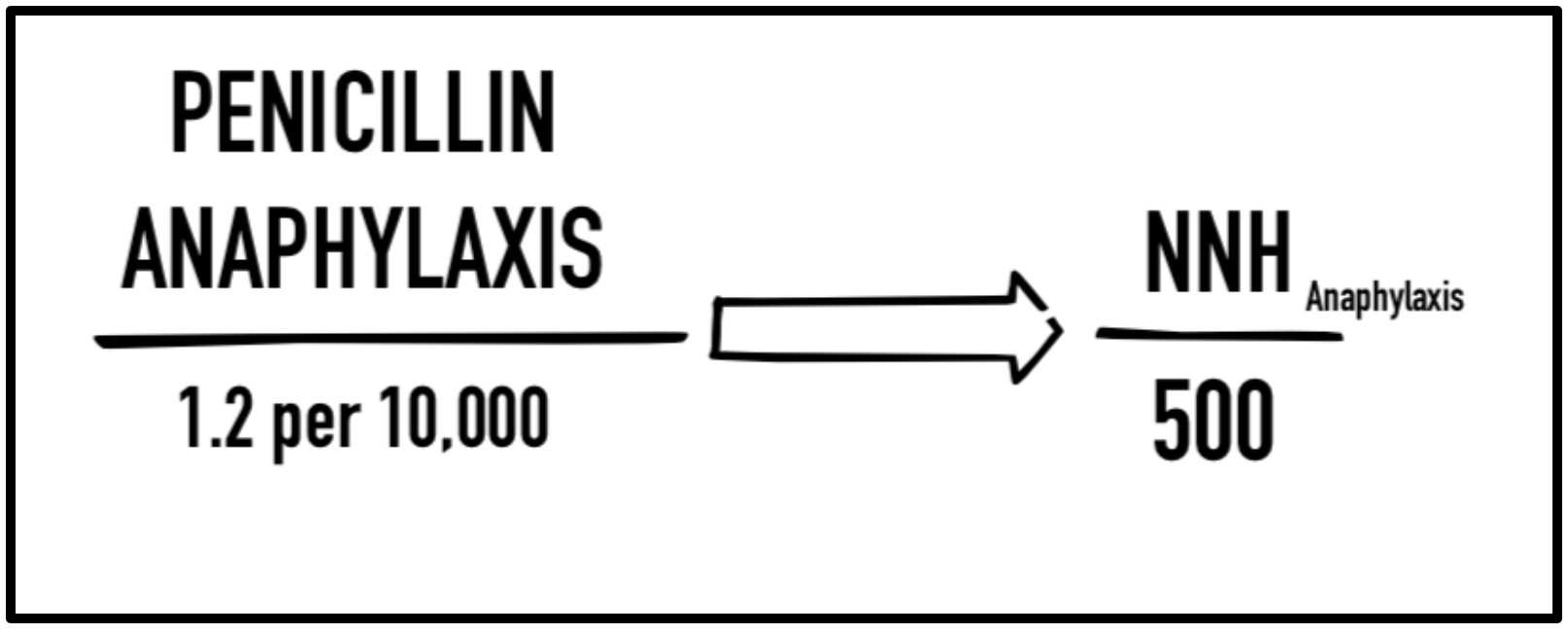
Risk of anaphylaxis from penicillin administration: penicillin administration has an anaphylaxis incidence of 0.2% (1.2/10,000 injections), a number needed to harm (NNH) of 500.
How can we know if its viral vs bacterial pharyngitis?
Patients with viral pharyngitis will present with symptoms such as cough, coryza, conjunctivitis, sneezing, voice hoarseness, sinus discomfort, oral ulcers, and occasionally a recognizable viral exanthem. Patients with clear-cut viral symptoms should not be pursued further for bacterial etiology due to the risk of false negatives and unnecessary workups.
In patients with indeterminate symptoms, consider the following diagnostic modalities:
CENTOR Criteria: Many may have heard of the CENTOR Criteria, a listing of 4 criteria that can be used to estimate the probability that pharyngitis is streptococcal, and to suggest a potential management course. The 4 criteria can be remembered using the mnemonic “CAFE”: Cough (absent), Adenopathy, Fever, Exudate (tonsillar). The increase number of CENTOR criteria present, the higher the probability of strep pharyngitis. But how accurate is this? Estimates suggest that a score of 1 out of 4 has a 10% probability of strep pharyngitis, 2 out of 4 has a 20% probability, 3 out of 4 a 35% probability, and 4 out of 4 a 50% probability. So even a slam dunk 4 out of 4 CENTOR still means you’re basically a coin flip away from knowing whether the patient does or doesn’t have strep. It is suggested that for a score of 2 or less, no further treatment is recommended and that for a score of 3 or 4 you can pursue a rapid strep antigen detection test (RADT) to determine treatment based on your ‘clinical gestalt’.

Rapid strep antigen detection test (RADT): The rapid strep can also be used to assess for bacterial colonization. Although this is faster than the throat culture you’re still only assessing for colonization, not infection. The rapid strep has a sensitivity of 70-90% and a specificity of 95%. Not the perfect test – you’re still susceptible to numerous false negatives and false positives.
Throat culture: Throat culture remains the gold standard. However, results can take up to 48 hours to return, delaying the diagnosis and potential initiation of antibiotics. Additionally, a positive throat culture implies colonization but not necessarily infection (ie, carrier state) – potential false positive.
ASO Antibodies: ASO antibody titers are not recommended in the routine diagnosis of acute pharyngitis as they reflect past but not current events (IDSA, strong rec).
If the patient is rapid strep negative, should we follow up with a throat culture?
It depends – yes for kids, no for adults. According to the IDSA recommendations, “In children and adolescents, a negative rapid strep test should be backed up by a throat culture [] however, routine use of back-up throat cultures for adults with a negative RADT is not necessary in usual circumstances, because of the low incidence of GAS pharyngitis in adults and because the risk of subsequent acute rheumatic fever is generally exceptionally low in adults with acute pharyngitis”.
If they’re rapid strep positive, should we follow up with a culture?
Yes, if you’re planning to initiate antibiotics (which you most likely are if you pursued the RADT in the first place) – for patients in which non-GAS is confirmed by throat cultures, antibiotics should be promptly discontinued.
“Treat it and you’ll be better in 7 days; don’t treat it and you’ll be sick for a week”
Untreated, the symptoms of strep pharyngitis are believed to last approximately 7 days. Although the research is limited, bacterial treatment has been shown to reduce these symptoms on average 14 hours (Spinks, 2013). Of note, all the studies in this Cochrane review compared antibiotics to placebo. They did not compare symptom relief with antibiotics to analgesics such as a combination of acetaminophen/ibuprofen, which may have been equally as efficacious at reducing the duration of symptoms. Bottom line – this short decrease in duration may not justify the added risks associated with antibiotic use.
What is the data to support a reduction in rheumatic fever?

If you’ve ever combed the literature on strep pharyngitis, you would know that a recurring statistic pops up when promoting antibiotic treatment of strep pharyngitis: specifically, antibiotics reduce the risk of acute rheumatic fever from 2.8% to 0.2%. This is the only figure I’ve seen, and it’s used in all the papers published for over half a century. Interestingly, this statistic came from a single military study conducted in the 1940’s (Denny, 1950). In the study, 1,600 young males enrolled in the US Air Force at Fort Warren, Wyoming. Each of these military recruits who presented with tonsillar exudate were randomized to receive either penicillin G (300,000U IM) and repeated in 72h (or again at 48h and 600kU at 96h) or placebo. The patients then returned to their military barracks for a 4-week follow-up to assess for signs of ARF (JONES Criteria – 2 major or 1 major + 2 minor). Of the patients taking antibiotics, 2 out of 800 (0.2%) developed rheumatic fever by follow up compared to 17 out of 800 patients (2.8%) in the control group; this was a number needed to treat (NNT) of 53. Given that approximately 50% of patients with ARF develop RHD (McMurray & Garber, 2015), there was an extrapolated NNT of 106 to prevent one case of RHD (see calculation below). The authors concluded that treatment of strep reduced development of rheumatic fever and suppressed the ASO response compared to placebo.
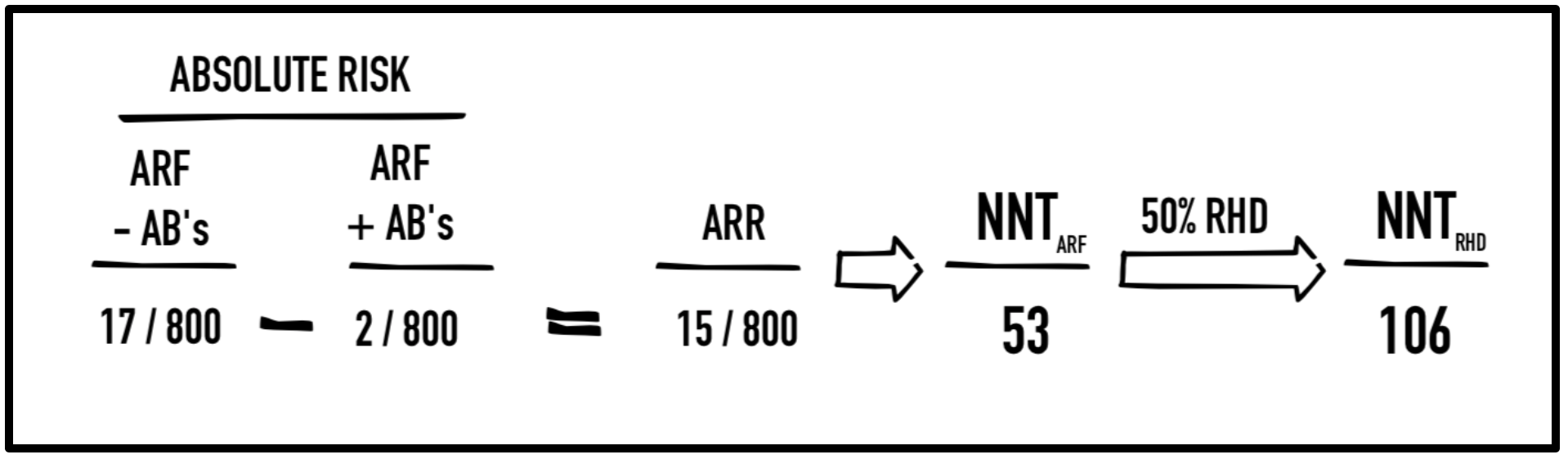
Benefit of antibiotics in treating strep pharyngitis (Denny, 1950): Acute rheumatic fever (ARF) treated with antibiotics (AB’s) resulted in an absolute risk reduction (ARR) of 15/800 equating to a number needed to treat (NNT) of 53 to prevent 1 case of ARF and 106 to prevent one case of rheumatic heart disease (RHD).
The major question, are these results generalizable? This was a very specific young, all-male population living in close-quarters under daily environmental stressors (military training). It is important to note that these results have never been replicated in a community setting. The NNT of 53 to prevent one case of ARF is based on a relatively high incidence of ARF seen only in military barrack epidemics and third world countries. More recent estimates suggest a nationwide community incidence of 300 per 2.2M without antibiotics compared to 75 per 2.2M with antibiotics. This is an absolute risk reduction (ARR) of approximately 1 per 100,000, or an NNT of 10,000 to prevent one case of ARF (McMurray & Garber, 2015) and an extrapolated NNT of 20,000 to prevent 1 case of RHD.

Benefit of antibiotics in treating strep pharyngitis (current US estimates): Acute rheumatic fever (ARF) treated with antibiotics (AB’s) resulted in an absolute risk reduction (ARR) of 15/800 equating to a number needed to treat (NNT) of 53 to prevent 1 case of ARF and 106 to prevent one case of rheumatic heart disease (RHD).
Now do you remember that NNH of 500 for anaphylaxis following penicillin administration and all the other adverse effects associated with antibiotics? This is the information you must use to decide whether antibiotics are doing more harm than good in treating strep pharyngitis.
But if antibiotics don’t prevent ARF, why the large decrease in incidence of ARF in the US?
The most consistent argument for reduced incidence of ARF is that changes in streptococcal M-protein types have led to reduced rheumatogenicity and reduced ability to cause ARF, not an increase in antibiotic use (Berkley, 2018). This is supported by data showing a reduced incidence in streptococcal infection occurring long before the advent of antibiotics (see image below). Additionally, an analysis of the 1987 Utah ARF outbreak, which showed no increased incidence of strep pharyngitis despite an increased incidence of AR, suggests that it is a specific rheumatogenic strain of GAS – rather than the GAS species alone – that is causing the ARF outbreaks (CDC, 1987). Worth noting, 50% of patients with ARF in this population study did not have a sore throat during the 2-month period before onset.

Bottom line: Outbreaks of acute rheumatic fever are now believed to occur because of sporadic changes in GAS rheumatogenicity, not decreased diagnosis or reduced antibiotic administration.
If we’re not giving antibiotics, what else can we do for patients with pharyngitis?
Steroids help reduce symptoms. In a large systematic review and meta-analysis, patients receiving low-dose corticosteroids were twice as likely to have complete pain relief at 24 hours (NNT=3) with complete pain resolution 11 hours sooner than placebo (BMJ, 2017). Adverse events were not appreciably increased in patients who received steroids. Consider a one-time dose of 10mg PO or IM dexamethasone (0.6mg/kg for peds, max 10mg) prior to discharge. Additionally, a 3-day course of acetaminophen/ibuprofen combination (650-975mg/ 400mg, q 6-8 hours) for symptomatic relief. Got a cough with that sore throat? Anyone who has worked with Jim Tsung know buckwheat honey has been shown to be efficacious in reducing cough compared to dextromethorphan or placebo (Paul, 2007).
The Upshot
The majority of sore throat is viral. Although the historical incidence of ARF and its correlation to strep pharyngitis has led many to believe an antibiotic regimen is necessary to prevent consequential sequelae, no definitive community-based data has ever supported this claim. Unfortunately due to the rarity of ARF in the United States it seems unlikely that such a study will ever be formally conducted. However, correlative data leaves us with an NNT that many would argue does not justify the added risks associated with an antibiotic regimen. For patients in whom symptoms remain the major concern, consider a cocktail of dexamethasone, acetaminophen/ibuprofen, and buckwheat honey instead.





