Authors: Nikita Desai, MD (EM Resident Physician, Hospital of the University of Pennsylvania) and David Jang, MD (EM Attending Physician, Hospital of the University of Pennsylvania) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Tim Montrief, MD (@EMinMiami); Brit Long, MD (@long_brit)
Case
An otherwise healthy 24-year-old woman is brought into the emergency department by her roommate with a chief complaint of altered mental status. Her roommate confirms her last known normal was at a party they both attended last night; however, they left separately. By the time the patient returned home in the early morning, she was acting strange and had become progressively more somnolent. Vital signs include HR 105 bpm, RR 14, BP 140/90 mmHg, T 99º F. She is arousable to sternal rub, but she does not follow any commands. Her physical exam is otherwise unremarkable. Initial fingerstick is 100 mg/dL. You order a non-contrast Head CT, while basic labs are sent along with a urine drug screen.
Background
Hyponatremia is defined as a serum sodium concentration < 135 mEq/L.1-3 Although the value in and of itself is not pathogenic, serum sodium concentration remains a measurable surrogate for the distribution of water across cell membranes. A breach in this delicate balance can lead to catastrophic effects including cerebral edema, tentorial herniation and ultimately respiratory arrest. 4 One epidemiologic study found hyponatremia (< 135 mEq/L) to be an independent predictor of mortality with a hazard ratio of 2.10 (95% CI: 1.41–3.12) even in an ambulatory patient population.5 Prompt treatment, however, must be balanced by the risk of osmotic demyelination syndrome (ODS) secondary to overcorrection.4 While the treatment of hyponatremia can be prolonged, outlined below is a practical guide for the acute management in the emergency department .4
Classifications: Concentration and Tonicity
Although the literature varies on the exact numerical cut-offs, the following parameters are good starting points when evaluating the hyponatremic patient.1-3

As a general principle, a sodium concentration < 130 mEq/L is to be considered pathologic until proven otherwise.6 Interestingly, the relationship between serum sodium concentration and mortality may not be as conversely linear as previously thought. One study found an increasing mortality rate with decreasing serum sodium concentrations until 120 mEq/L, at which point paradoxically mortality decreased (Figure 1) in the hospitalized patient population.
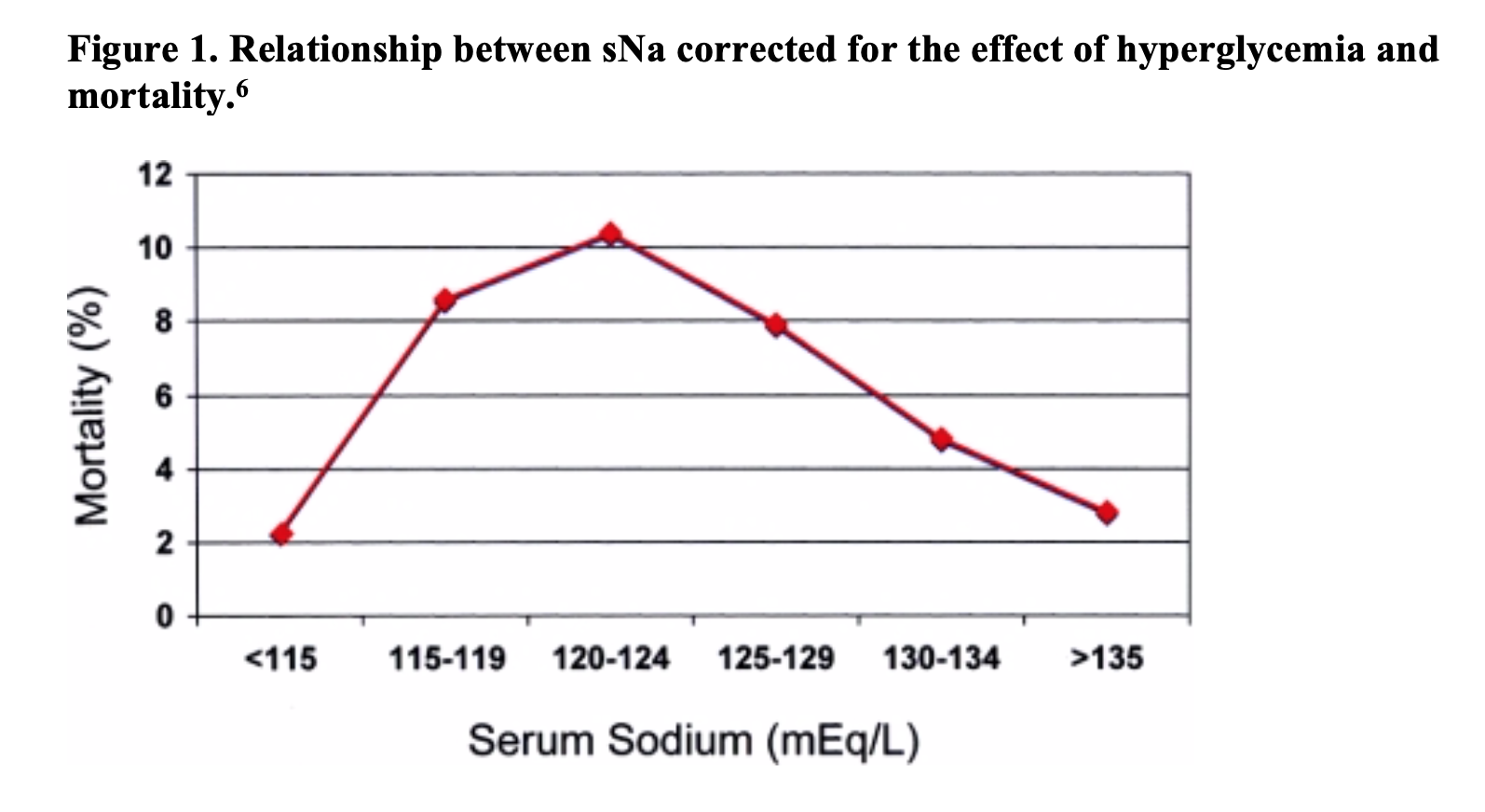
They propose that patients with extreme derangements may have more easily reversible underlying pathology such as medication side effect. In brief, treatment based on numerical value alone may lead to iatrogenic harm. Rather, emergent treatment of dysnatremia should be dependent upon the acuity of the hyponatremic state guided by the clinical evaluation of the patient. 4-7
In addition to serum concentration, hyponatremia is further characterized by the tonicity of the fluid. Tonicity refers only to the solutes that cannot pass through a semipermeable membrane and therefore influence the osmotic gradient. These effective solutes include sodium and glucose in the extracellular fluid or exogenous sugars. The large fluid fluctuations which can lead to cerebral edema are limited to hypotonic hyponatremia measured as a serum osmolality (sOsm) < 280 mosm/kg. Prior to engaging in treatment, identification of the tonicity of fluid is crucial as this aids in rapid diagnosis and treatment of hyponatremia.4
sOsm <280 mosm/kg
Hypotonic hyponatremia will be discussed below. Traditionally categorized by volume status, we will discuss an alternative algorithm that aids treatment management.
sOsm 280 – 295 mosm/kg
Definition: This range is referred to as isotonic hyponatremia and can be secondary to lab artifact (pseudohyponatremia) or isotonic infusions of osmotically active solutes.1
Pseudohyponatremia: hypertriglyceridemia, paraproteinemia (multiple myeloma)1
Isotonic infusions: recent IVIG infusion, IV glucose/mannitol, irrigants for urethral procedures4
Management: No emergent treatment required, direct ion detection for lab artifact (if available at your lab)
sOsm >295 mosm/kg
Definition: This indicates there are osmotically active solutes in the serum and is also referred to as hypertonic hyponatremia.
Examples: glucose, mannitol 1-2,8
Management: For hyperglycemia, calculate corrected sodium concentration by adding 2 mEq/L to the serum sodium for each 100 mg/100 mL increase in glucose concentration. In addition, patients will require IV or subcutaneous insulin along with judicious intravenous fluids.
Clinical Presentation
The central nervous system is the primary site of organ dysfunction in the hyponatremic state. Unfortunately hyponatremia, similar to other metabolic derangements, can mimic a wide array of disease states ranging from a gastrointestinal virus to status epilepticus.9 Therefore, a high clinical suspicion and a low threshold for obtaining labs is essential for diagnosis in the setting of deceivingly benign presentations. The severe features listed below clinically correspond to cerebral edema and increased intracranial pressure that require emergent treatment. Risk stratification of the subtler presentations such as nausea and dizziness mount a more difficult challenge for the emergency physician highlighting the importance of obtaining a good history.1,9

Time of onset
The brain has developed many adaptive mechanisms to counteract stressed conditions; as is the case in hypotonic hyponatremia. In short, astrocytes will transport osmotically active cytoplasmic solutes into the extracellular space in an attempt to counteract a hypotonic environment. This mechanism decreases the osmotic gradient, preventing excess water from entering the intracellular space which would cause cerebral edema. This re-establishment of the equilibrium is estimated to occur over 24-48 hours and is the basis for why acute hyponatremia is managed differently than chronic hyponatremia.10 Conversely, rapid correction of chronic hyponatremia, where osmotic equilibrium has already been established, may lead to unwanted fluid shifts into the extracellular space; effectively causing cellular apoptosis and demyelination of the central nervous system. Understanding the pathophysiology behind hyponatremia is crucial in risk stratification of the hyponatremic patient.1,10 Inappropriate treatment of hyponatremia can lead to severe morbidity and mortality.
Acute Hyponatremia
- Symptom onset < 48 hours
- Higher risk of cerebral edema
- Typically present with a moderate to severe hyponatremia
- Patients are more symptomatic with neurologic manifestations
- Lower chance of rapid overcorrection and ODS
Chronic Hyponatremia
- Symptom onset > 48 hours or unknown time of onset
- Typically have multiple comorbidities such as Congestive heart failure, Cirrhosis, Nephrotic syndrome, Chronic kidney disease
- Lower risk for cerebral edema as the brain has had time to adapt1
- Higher risk for rapid overcorrection and subsequent ODS
An unknown time of onset may in fact be secondary to an acute precipitant. However, without definitive data such as a reliable patient history or collateral information, an unknown time of onset is treated in a similar fashion to chronic hyponatremia requiring a slower correctional rate.
You are notified that your patient has a sodium level of 120 mEq/L. The head CT was unrevealing. The patient remains lethargic. The nurse is unable to collect a urine sample.
History
The bedside examination of the patient provides an abundance of information in regards to treatment initiation in the emergency department. Listed below are pertinent questions that need to be addressed if mental status permits. Obtaining collateral from family and friends is equally important.
Onset and classification of symptoms:
- Hyponatremia with severe symptoms requires emergent treatment regardless of chronicity
- Symptom onset: acute vs chronic
Free water intake/output:
- Increased free water intake: athlete with increased free water intake, psychogenic causes
- Decreased free water output: renal failure, medication side effects: desmopressin
Solute intake/secretion:
- Exercise induced (increased sweating)
- Ask about diet: low solute diets such as tea/toast, beer potomania
Past medical history:
- Congestive heart failure, cirrhosis, malignancy, thyroid disorder, chronic steroids
- Review psychiatric history (psychogenic polydipsia, SSRI initiation, etc)1
- Recent surgical history (post-operative fluid administration, intracranial surgery)
Medication review:
- Refer to Table 3 for a brief list of medication induced SIADH11
- Ask about recreational drug use (MDMA)11

ADH dependence/ Volume exam
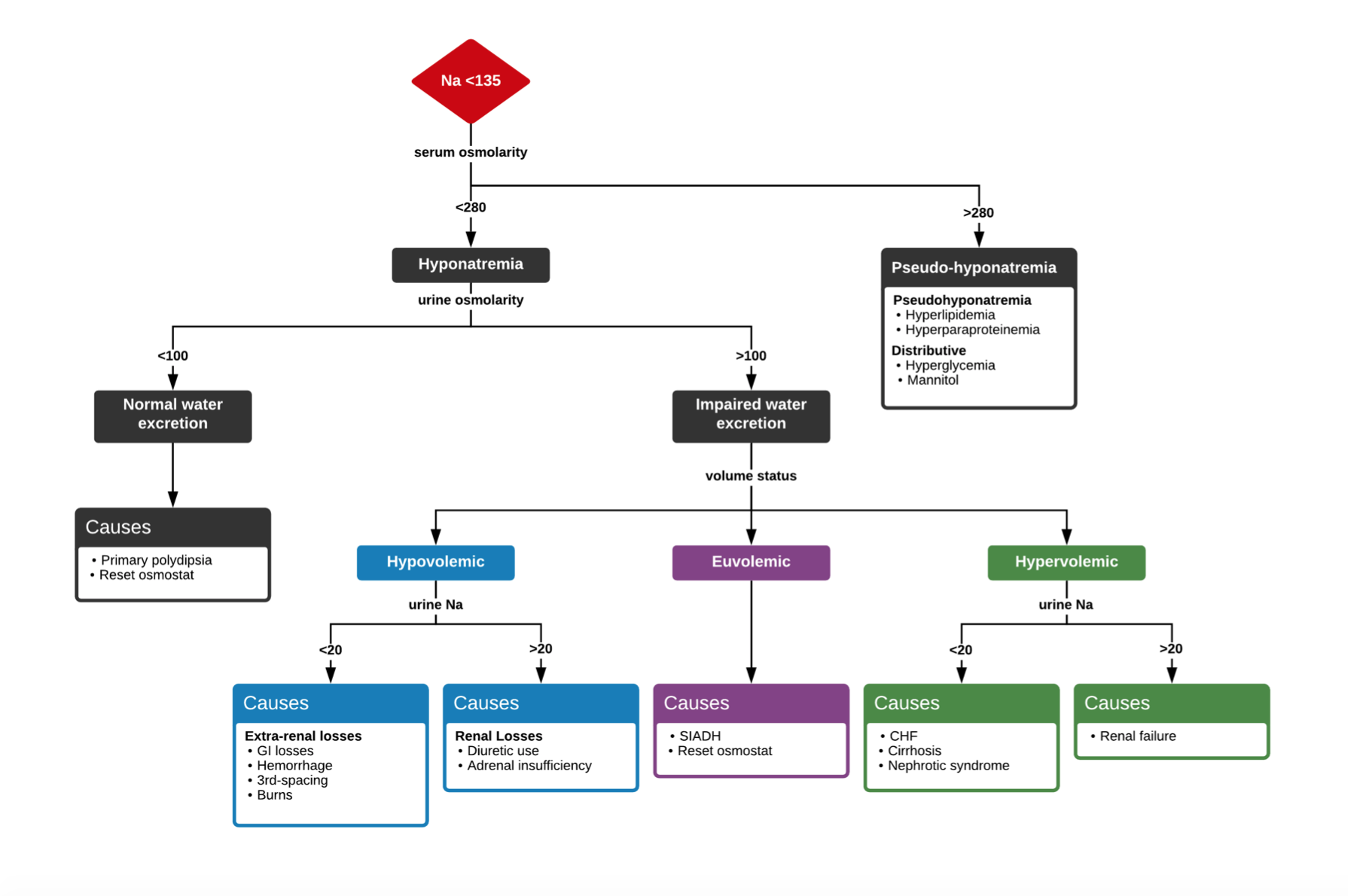
Although volume exam has been historically used to differentiate between the causes of hyponatremia, an alternative and more physiologically driven pathway to consider is antidiuretic hormone dependence (ADH). ADH otherwise known as vasopressin acts on the V2 receptors in the kidney increasing free water absorption in the collecting tubules. It is a hormone produced in the hypothalamus responsible for maintaining intracellular and extracellular tonicity. ADH secretion is triggered by an increase in serum osmolality (detected by osmoreceptors in the hypothalamus) and/or a decrease in effective arterial blood volume with the latter having a much larger influence.1-2,12 If there is low effective arterial blood volume (meaning less renal perfusion), there will be an increase in ADH secretion even in the presence of decreased plasma osmolality.1-2,12 A urine osmolality greater than plasma osmolality (typically > 100 mosm/kg) indicates ADH dependent hyponatremia and can not only help categorize patients but can also aid in treatment management. Once ADH dependence is identified by the urine studies, action can be taken to reverse the trigger- physiologic or pathologic in nature.1-2
What causes low effective arterial blood volume?
- Hypervolemic states such as CHF, Cirrhosis, Nephrotic syndrome as volume is redistributed to the venous system or the extravascular space.
- Hypovolemic states such as increased NG tube output, emesis/diarrhea, diuretic use, burns
Pathologic ADH secretion
- Hypothyroidism
- Primary or secondary adrenal insufficiency
- SIADH
Figure 2. Hyponatremia Flowchart1,13
Laboratory Evaluation
Patients may present to the emergency department with minimal information in regard to pertinent medical conditions or recent precipitating events and therefore it is important to cast a wide net in the initial work up of hyponatremia. A fingerstick blood glucose measurement, basic metabolic panel, and serum osmolality should be obtained for patients with possible hyponatremia. These lab results can rapidly categorize and aid in diagnosis of the hyponatremic patient based on tonicity alone. The remaining lab work presented below is tailored towards individual disease states associated with hyponatremia which can be ordered on the basis of clinical suspicion. We strongly advocate for sending urine studies (urine osmolality, urine electrolytes) on patients without a clear-cut etiology as this can help differentiate ADH dependence as illustrated above.

Serum uric acid and urine uric acid may assist if the patient is on recent diuretics to help differentiate between diuretic induced hypovolemia vs SIADH when volume assessment is equivocal. In both cases urine sodium will be >20 mEq/L. 14
Adrenal insufficiency typically presents with specific electrolyte abnormalities: hyponatremia, hyperkalemia and low/normal glucose.
Treatment
Symptomatic Hyponatremia without CNS dysfunction
- Send off aforementioned labs if clinically indicated
- Review medications: common culprits of mediation induced SIADH include analgesics (ibuprofen, opioids), thiazide diuretics, antipsychotics (SSRIs, haldol) and temporarily stop offending agents1-2
- Fluid restriction and NPO (nothing by mouth) status with two caveats
- Trial of loop diuretics in the grossly overloaded patient with a history of CHF or Cirrhosis
- Trial of small fluid bolus (250-500 cc Lactate Ringers for the hypovolemic patient (emesis, nasogastric tube output, diarrhea)
- Trend sodium concentration q1-2 hours
- Follow up on urine studies to tailor treatment options

Tolvaptan (oral V2 inhibitor) and Conivaptan a V1a/V2 inhibitor remains an option for ADH dependent, hypervolemic patients with congestive heart failure or cirrhosis however they are expensive and hepatotoxic. Administration should rely on a multi-disciplinary discussion between the emergency medicine physician, nephrology and pharmacy.2,3,15,16
Hyponatremia with CNS dysfunction
- If the patient is significantly altered or seizing with serum sodium concentration < 130 mEq/L, administer 3% hypertonic saline 100 cc IV over 10 min peripherally or centrally (can substitute for an ampule of bicarbonate)8
- Up to three boluses of hypertonic saline may be administered over a span of 30 minutes, symptom resolution is the target8
- If symptom resolution is not achieved after bolus administration, patient may be placed on a hypertonic infusion with a target of 4-6 mEq/L increase in the first six hours (use a weight-based formula)
- Check serum sodium concentrations hourly, expect sodium concentrations to rise 1-2 mEq per bolus
- Once symptom resolution is achieved, transition to continuous isotonic intravenous infusion with a target of 4- 6 mEq/L increase in the first six hours (use a weight-based formula)
- Consider concurrent administration of desmopressin (1-2mcg IV q8 hours) if the patient examines as hypovolemic (discussed below)3,16
Current recommendations have defined correction parameters in both acute and chronic settings as follows: The rate of correction should be no greater than 8 mEq/L in a 24-hour period and a total of 16 mEq/L in a 48-hour period.2Unfortunately, there have been reported cases of ODS with a stated correction rate of 9 mEq/L in a 24 hour period which highlights the weakness of such an approach.17 Alternatively, the rule of 6 (defined below) can help safely navigate the balance between treating CNS dysfunction and overcorrection.1,3,14
“Six [mEq/L] in six hours for severe symptoms, then stop. Six a day makes sense for safety.”
Restated, aim for a 4-6 mEq/L increase in the first six hours of treatment if the patient has signs of CNS dysfunction listed above. Do not correct greater than 6 mEq/L in the first 24 hours to avoid the risk of ODS.1,14,18
Disposition
Intensive care unit admission
- Patient remains symptomatic (altered, lethargic)
- Current hypertonic infusion 8
- Administration of hypertonic saline bolus 8
- Rapid rise in serum sodium concentration greater than 1 mEq/L/hr
- Follow local hospital protocols; serum sodium concentration parameters
Medical floor admission
- Patient is back to neurologic baseline
- Stable serum sodium trend over the course of 4-6 hours (may differ based on local protocols)
- Q 4-6 hour labs
- Nephrology should be consulted in the emergency department for symptomatic hyponatremia or rapidly rising serum sodium concentrations to aide in treatment adjuncts such as addition of desmopressin or vaptans
- Ensure accurate monitoring of urine output with placement of a Foley catheter or condom catheter as early as possible
The Contingency Plan: You overcorrected…now what?
As discussed above, if you correct the trigger for ADH secretion whether it be physiologic or pathologic in etiology, the serum sodium concentration will rise clinically correlated to aquaresis (electrolyte free urine output). If this level rises too quickly, the patient is at risk for ODS which is a true emergency. Treatment remains to administer or limit excretion of free water which can be accomplished orally, via intravenous infusions (D5W) or by medications. Desmopressin is a man-made form of vasopressin and can be a good prophylactic as well as reactive rescue medication as it essentially stops free water excretion from the kidneys.16 Desmopressin should only be considered as an adjunct to hyponatremia management in the patients that are high risk for reversing their ADH trigger too quickly causing overcorrection. This special cohort of patients is listed below. Desmopressin can be administered concurrently with initial fluid administration (including hypertonic bolus) or as a reactive measure to rapidly rising sodium concentration.20-22
Prophylactic Desmopressin
- Consider in the severely hypovolemic patient and the undifferentiated patient with multiple comorbidities (liver disease, alcoholism, hypokalemic)19
- These are the patients that are at highest risk for overcorrection19
- Desmopressin 1-2 mcg every 8 hours until sodium level normalizes20,21
- No additional oral intake
- Do not administer desmopressin to the grossly hypervolemic patient
- Desmopressin will not affect the blood pressure like vasopressin20-22
Reactive Desmopressin
- Red flags: Urine output > 100 cc/hr, sodium correction > 1 mEq/L/hr or breach in the rule of six
- Desmopressin 1-2 mcg q8 hrs, this will be continued until sodium concentration normalizes20,21
- Consider concurrent D5W infusion 3-6 cc/kg over 1-2 hours14,21
- Nephrology should be consulted immediately
After obtaining the results of the metabolic panel you decide to administer 100 mL 3% hypertonic saline. A foley catheter is placed to help monitor urine output. The urine drug screen later results positive for MDMA (ecstasy), a trigger for SIADH. The patient’s mental status improves after the first bolus and she is admitted to the medical floor for further stabilization.
Pearls:
- Rate of serum sodium concentration change is far more important to quantify than the numerical number itself. Rely on symptoms and good history taking skills.
- Just because you can correct acute hyponatremia quickly does not mean you should. Aim for a 4-6 mEq/L increase in the first six hours of treatment if the patient has signs of CNS dysfunction listed above. Do not correct greater than 6 mEq/L in the first 24 hours to avoid the risk of osmotic demyelination syndrome.
- If you do not have ready access to hypertonic saline (100 cc of 3% hypertonic saline IV over 10 min), you can substitute with an ampule (50 ml) of bicarbonate (8.4% sodium bicarbonate).
- Confirm tonicity of fluid (hypo-, iso-, hyper-) prior to initiating treatment in the emergency department.
- Be comfortable with the use of desmopressin in the case of undifferentiated hyponatremia, severely hypovolemic hyponatremia and rapid overcorrection of hyponatremia. You can administer this concurrently during hypertonic or intravenous fluid administration for prophylactic measures.20-22
Pitfalls:
- Be very cautious in administration of intravenous fluids (Lactate preferably over NaCl or 3% hypertonic) in patients with hypovolemic history (poor oral intake, diarrhea, increased NG tube output, vomiting) as they tend to overcorrect rapidly leading to an increased risk of osmotic demyelination syndrome. Be judicious with fluid challenges (250cc – 500cc per bolus).
- If you administer 3% hypertonic saline, pay close attention to the urine output and consider placing a foley early, urine output >100 cc/hr is an early red flag for overcorrection.21
Further FOAM Resources:
- CORE EM: Severe Hyponatremia
- Life in the Fastlane: Hypertonic Saline
- EM Cases: Emergency Management of Hyponatremia
- EM Basic: Hyponatremia
- Life in the Fastlane: Hyponatremia
- DDxOf: Hyponatremia
References
[1] Verbalis JG, Goldsmith SR, Greenberg A, et al. Diagnosis, evaluation, and treatment of hyponatremia: expert panel recommendations. The American journal of medicine. https://www.ncbi.nlm.nih.gov/pubmed/24074529. Published October 2013.
[2] Hoorn EJ, Zietse R. Diagnosis and Treatment of Hyponatremia: Compilation of the Guidelines. Journal of the American Society of Nephrology. 2017;28(5):1340-1349. doi:10.1681/asn.2016101139.
[3] Braun MM, Barstow CH, Pyzocha NJ. Diagnosis and Management of Sodium Disorders: Hyponatremia and Hypernatremia. American Family Physician. March 2015:299-307. https://www.aafp.org/afp/2015/0301/p299.html.
[4] Filippatos, T.D., et al. “Ten Common Pitfalls in the Evaluation of Patients with Hyponatremia.” European Journal of Internal Medicine, vol. 29, 2016, pp. 22–25., doi:10.1016/j.ejim.2015.11.022.
[5] Gankam-Kengne, Fabrice, et al. “Mild Hyponatremia Is Associated with an Increased Risk of Death in an Ambulatory Setting.” Kidney International, vol. 83, no. 4, 2013, pp. 700–706., doi:10.1038/ki.2012.459.
[6] Chawla A, Sterns RH, Nigwekar SU, Cappuccio JD. Mortality and serum sodium: do patients die from or with hyponatremia? Clin J Am Soc Nephrol. 2011;6(5):960–965. doi:10.2215/CJN.10101110
[7] Mohan S, Gu S, Parikh A, Radhakrishnan J. Prevalence of hyponatremia and association with mortality: results from NHANES. Am J Med. 2013;126(12):1127–37.e1. doi:10.1016/j.amjmed.2013.07.021
[8] Dineen, Rosemary et al. “Hyponatraemia – presentations and management.” Clinical medicine (London, England) vol. 17,3 (2017): 263-269. doi:10.7861/clinmedicine.17-3-263
[9] Sterns, Richard H., et al. “Treatment of Hyponatremia.” Current Opinion in Nephrology and Hypertension, vol. 19, no. 5, Sept. 2010, pp. 493–498., doi:10.1097/MNH.0b013e32833bfa64.
[10] Gankam Kengne, Fabrice, and Guy Decaux. “Hyponatremia and the Brain.” Kidney international reports vol. 3,1 24-35. 1 Sep. 2017, doi:10.1016/j.ekir.2017.08.015
[11] Shepshelovich, Daniel et al. “Medication-induced SIADH: distribution and characterization according to medication class.” British journal of clinical pharmacology vol. 83,8 (2017): 1801-1807. doi:10.1111/bcp.13256
[12] John, Savio, and Paul J Thuluvath. “Hyponatremia in cirrhosis: pathophysiology and management.” World journal of gastroenterology vol. 21,11 (2015): 3197-205. doi:10.3748/wjg.v21.i11.3197
[13] Fadial, Tom. “Differential Diagnosis of Hyponatremia.” Differential Diagnosis Of, 10 Oct. 2019, https://ddxof.com/hyponatremia/.
[14] Weingart S. Severe Hyponatremia in the Emergency Department. EMCrit Project. https://emcrit.org/emcrit/hyponatremia/. Published July 30, 2017.
[15] Bulloch MN. The Treatment of Acute Hyponatremia with Tolvaptan. American Research Journal of Pharmacy. 2018;3(1). doi:10.21694/2380-5706.18001.
[16] Tzamaloukas AH, Shapiro JI, Raj DS, Murata GH, Glew RH, Malhotra D. Management of severe hyponatremia: infusion of hypertonic saline and desmopressin or infusion of vasopressin inhibitors? Am J Med Sci. 2014;348(5):432–439. doi:10.1097/MAJ.0000000000000331
[17] Adrogué HJ, Madias NE. The challenge of hyponatremia. J Am Soc Nephrol 2012; 23:1140.
[18] Kilian M. Emergency Management of Hyponatremia: EM Cases. Emergency Medicine Cases.https://emergencymedicinecases.com/episode-60-emergency-management-hyponatremia/. Published June 16, 2019.
[19] George JC, Zafar W, Bucaloiu I, Chang AR. Risk Factors and Outcomes of Rapid Correction of Severe Hyponatremia. Clinical Journal of the American Society of Nephrology. July 2018:984-992. doi:https://doi.org/10.2215/CJN.13061117 .
[20] Ward FL, Tobe SW, Naimark DMJ. The Role of Desmopressin in the Management of Severe, Hypovolemic Hyponatremia: A Single-Center, Comparative Analysis. Can J Kidney Health Dis. 2018;5:2054358118761051. Published 2018 Mar 21. doi:10.1177/2054358118761051
[21] Farkas J. Taking control of severe hyponatremia with DDAVP. EMCrit Project. https://emcrit.org/pulmcrit/taking-control-of-severe-hyponatremia-with-ddavp/. Published December 13, 2016.
[22] Hypertonic saline and desmopressin: A simple strategy for safe correction of severe hyponatremia. Sood L., Sterns R.H., Hix J.K., Silver S.M., Chen L. (2013) American Journal of Kidney Diseases, 61 (4) , pp. 571-578.






1 thought on “Critical Hyponatremia: Pearls and Pitfalls”
Pingback: Quiz 58, January 17th, 2020