Cystic Fibrosis: ED Management, Pearls and Pitfalls
- Sep 24th, 2016
- Stephanie Tassin
- categories:
Authors: Stephanie Tassin, MD (EM Resident at SAUSHEC) and Brit Long, MD (@long_brit, EM Attending Physician at SAUSHEC) // Edited by: Jennifer Robertson, MD, MSEd and Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW Medical Center / Parkland Memorial Hospital)
Introduction
Cystic fibrosis (CF) is a life-shortening, autosomal recessive disease that affects multiple organ systems via mutations in the Cystic Fibrosis Transmembrane Conductance Regulator (CFTR) chloride channel. In short, a defective CFTR channel prevents mucous and exocrine glands from secreting chloride. Without chloride, water cannot follow, and the end result is thick, hyperviscous secretions in the lung, sinuses, pancreas, intestines, and biliary system [1]. In the sweat glands, chloride reabsorption is impaired, leading to excess sodium chloride loss. With excessive sweating, this can lead to hyponatremic hypochloremic dehydration. It also has detrimental effects on the reproductive, musculoskeletal, and urinary systems, but these are rarely relevant in the Emergency Department (ED) [2].
CF affects 1 in 3,200 Caucasian births and is primarily thought of as a Caucasian disease, though it is also seen in other ethnicities at lower frequencies. As of 2014, less than 2/3 of cases are detected by newborn screening, so providers must keep it on their differential, especially in young children with recurrent pulmonary infections, sinus infections, failure to thrive, and constipation or meconium ileus [2].
Case 1
A 22 year old female with cystic fibrosis presents to you complaining of increasing shortness of breath over the last week. Her chronic cough has gotten worse recently and it is productive of occasionally blood-tinged and green sputum. She reports low-grade fevers at home as well as fatigue, headache, and anorexia. She just moved to town and has no medical records in your system. Vital signs include blood pressure (BP) 110/64, heart rate (HR) 110, respiratory rate (RR) 34, oxygen saturation (SpO2) 92% on room air, temperature (T) 100.1°Farenheit (F) orally. Her examination is remarkable for coarse breath sounds with scattered expiratory wheezes, a slightly increased work of breathing, and clubbing of her fingernails. Her chest x-ray is shown below.
http://img.medscape.com/pi/emed/ckb/radiology/336139-354931-3315.jpg
You put her on oxygen by nasal cannula and give her a small intravenous (IV) fluid bolus, but you wonder what to do next. You do not see a lot of patients with CF, but you see a lot of chronic obstructive pulmonary disease (COPD), and she looks almost exactly like a patient with a COPD exacerbation. Do you just treat her like COPD with nebulizer treatments, steroids, antibiotics, and bi-level positive pressure ventilation (BiPAP)? What antibiotics do you use? Is there anything else you can offer?
CF Pulmonary Exacerbations
Progressive pulmonary disease accounts for 85% of the mortality in CF[3]. From a young age, the CF patient is colonized with a predictable spectrum of bacteria that persist and cause chronic infection and inflammation [1]. Typically, this causes a persistent, productive cough and an obstructive pattern on pulmonary function tests. This may progress to chronic bronchitis and bronchiectasis with occasional exacerbations. These pulmonary exacerbations are generally characterized by increasing dyspnea, tachypnea, changes in sputum production, increased adventitious lung sounds, malaise, anorexia, and a decrease in pulmonary function [3, 4, 5].
The majority of pulmonary exacerbations are caused by clonal expansion of existing strains of bacteria, rather than an acquisition of new bugs [6]. Still, other causes of pulmonary distress must be kept on the differential in cystic fibrosis patients. Allergic bronchopulmonary aspergillosis (ABPA) should be suspected in patients with significant wheezing. Though it will likely not be diagnosed in the emergency department [6], it is reasonable to start steroids in these patients as discussed below. Spontaneous pneumothorax can occur, especially in older patients with advanced disease. Recurrence is also common [7]. Minor hemoptysis is also common in CF, especially during a pulmonary exacerbation. Beyond checking the international normalized ratio (INR) to rule out any contributing vitamin K deficiency, this generally only requires reassurance. A complete blood count (CBC) is rarely needed unless in circumstances concerning for anemia.
In patients with suspected pulmonary exacerbations, plain chest radiographs should be ordered to exclude pneumothorax. Other findings such as mucous plugging, peribronchial thickening, and air space disease are common but not specific for an acute exacerbation [8].
Microbiology
Early in life, the most common bugs cultured from the lungs are Staphylococcus aureus and non-typeable Haemophilus influenzae [9]. Pseudomonas aeruginosa is also often isolated early and is the most significant pathogen in CF. Once established, P. aeruginosa is essentially impossible to eradicate due to a combination of genetic adaptations, biofilm formation, and an optimal environment in CF airways. Burkholderia cepacia complex is less common, but is still occasionally seen and can rapidly lead to necrotizing pneumonia, sepsis, and death. Other common organisms include S. maltophilia, and A. xylosoxidans, which tend to be less virulent. Aspergillus spp. is isolated from more than 25% of patients but it rarely causes invasive infection outside of the immunocompromised post-transplant patient. However, allergic bronchopulmonary aspergillosis (ABPA) can cause significant illness and it is a diagnosis to keep in mind if your patient presents with significant wheezing [9].
Treatment
Early recognition and treatment of pulmonary exacerbations has been associated with slower long-term decline in lung function [10]. Pulmonary exacerbations are treated with antibiotics and supportive measures aimed at airway clearance. In cases of ABPA, which is usually not diagnosed in the ED, patients may present with asthma-type symptoms and are usually treated with steroids [9].
Management is summarized in Table 1.
Table 1
| Management Considerations in CF Pulmonary Disease |
| 1. Antipseudomonal antibiotics (high doses): Aminoglycoside + β-lactam
2. Bronchodilator – either MDI or nebulized albuterol 3. Nebulized 7% saline, 4mL 4. BiPAP if needed 5. Only use steroids if there are significant asthma-type symptoms (i.e. wheezing) |
Antibiotics
In general, antibiotic choices should be tailored to the patient’s previous culture results if known. For mild exacerbations or presumed viral upper respiratory infections not requiring inpatient admission, it is reasonable to discharge patients on an oral antibiotic such as amoxicillin-clavulanate that will cover both H. influenza and S. aureus. If the patient has a known history of Pseudomonas infection, an anti-pseudomonal fluoroquinolone such as ciprofloxacin may be used (1 month – 5 years: 15mg/kg max 750mg/dose BID, 5-18 years: 20mg/kg BID). Recommended duration of treatment is 2 weeks [5]. Patients should continue to use their regular nebulized anti-pseudomonal antibiotic. In these cases, sputum should be sent for culture prior to starting therapy. If there is no improvement on oral antibiotics, there should be a low threshold to admit for intravenous treatment [6].
For more severe exacerbations or those failing outpatient therapy, treatment is usually aimed at Pseudomonas unless the previous cultures identify a different pathogen as the likely culprit. For presumed Pseudomonas infection in a patient with a severe exacerbation, double antibiotic therapy is preferred with two anti-pseudomonal drugs with different mechanisms of action. This is most often accomplished with an aminoglycoside and β-lactam [5]. While results of previous cultures are useful to determine which bug is most likely causing an exacerbation, antibiotic sensitivities for Pseudomonas in particular are not useful for selecting antibiotics and have no effect on patient outcomes [6, 11].
Of the aminoglycosides, tobramycin is most frequently used and has been extensively studied [12, 13]. Amikacin is also used but is used less often. Gentamicin is avoided in CF due to an increased risk of nephrotoxicity [12]. The CF Foundation recommends that aminoglycosides be administered in once daily doses rather than TID to maximize efficacy and minimize nephrotoxicity [3]. Of the β-lactams, ceftazidime is most commonly used among accredited CF centers, followed by cefepime, piperacillin-tazobactam, meropenem, ticarcillin-clavulanate, and aztreonam [14].
Note that patients with CF require larger doses of antibiotics (often larger doses than are FDA-approved) due to a larger volume of distribution, increased renal clearance, and the increased minimal inhibitory concentration (MIC) of Pseudomonas [12, 15]. Many providers, even in CF Foundation-accredited care centers, continue to prescribe inadequate doses of anti-pseudomonal antibiotics despite published dosing guidelines [16]. The best available evidence, based on the pharmacokinetics, pharmacodynamics, tolerability, and efficacy of different regimens, supports the dosing regimens listed below in Table 2 [15].
Table 2 – Antibiotic Choices in CF Exacerbation
| Aminoglycoside | Dose |
| Tobramycin | 10 mg/kg/day in one daily dose |
| Amikacin | 30-35 mg/kg/day in one daily dose |
| β-lactam | Dose |
| Ceftazidime | 200-400 mg/kg/day div every 6-8 hr (max 8-12 g/day) |
| Cefepime | 150-200 mg/kg/day div every 6-8 hr (max 6-8 g/day) |
| Pip-tazo | 350-600 mg/kg/day div Q4H (max 18-24 g/day piperacillin) |
| Meropenem | 120 mg/kg/day div Q8H |
| Ticarcillin-clavulanate | 450-750 mg/kg/day Q6H (max 24-30 g/day ticarcillin) |
| Aztreonam | 200-300 mg/kg/day div Q6H (max 8-12 g/day) |
For infections caused by bugs other than Pseudomonas, culture and susceptibility testing usually guide antibiotic choice. Staphylococcus aureus is a common pathogen in CF and can be treated according to local resistance patterns. Other typical bugs, notably B. cepacia complex, S. maltophilia, and A. xylosoxidans, tend to be very antibiotic-resistant, so treatment should be guided by culture and susceptibility testing [17].
Bronchodilators
Patients with CF have variable responses to bronchodilators. Approximately 50% of these patients will have some degree of bronchial hyperresponsiveness [9]. Given the relatively benign side effect profile of beta-agonists, nebulized treatments in the ED may assist in airway opening. Bronchodilators may not be the mainstay of treatment of pulmonary exacerbations, but it is helpful when administered prior to treatment with nebulized hypertonic saline, as discussed below.
Mucolytics (hypertonic saline)
Inhaled hypertonic saline (HS) has been shown to improve the properties of sputum and acutely increase mucociliary clearance in patients with CF. Unlike the other major mucolytic used for CF called DNase I (Dornase alpha), HS appears to be beneficial during an acute exacerbation, especially when followed by chest physiotherapy [9, 10]. A nebulized dose of 4mL of 7% saline has been shown to improve symptoms and lung function when compared to a control treatment with 0.12% saline. Patients treated with the 7% saline nebs during their hospitalization are more likely to return to their pre-exacerbation FEV1 with a number needed to treat of 6 [10]. It is generally well tolerated, but it does have the potential to cause a transient airflow obstruction. For this reason, patients should be treated with a bronchodilator immediately prior to hypertonic saline [18].
Positive pressure ventilation
As in COPD, non-invasive positive pressure ventilation is an attractive treatment option for severe pulmonary exacerbations. Several observational studies have looked at the use of NPPV in these situations and concluded that it may be useful, especially if used early during an acute exacerbation. It is at the very least preferable to invasive ventilation due to the high mortality rates seen in patients with CF who get intubated[19].
Steroids
Progression of lung disease in CF is largely mediated by chronic inflammation, so it makes sense that corticosteroids should provide some benefit [20]. Unfortunately, although treatment of CF with long-term corticosteroids has shown some improvement in lung function, the risks and significant side effect profile preclude their utility on a regular basis. Theoretically, short-term use of steroids during an acute pulmonary exacerbation should have a more acceptable risk-benefit profile. Only two small studies (n=44) have looked at the use of steroids during an acute exacerbation and showed a small trend towards improvement in lung function at follow up [20, 21]. In the larger of the studies, 3 of the 12 patients in the prednisone group had to be withdrawn from the study either from hypertension or hyperglycemia [20]. The CF Foundation concludes there is insufficient evidence to recommend routine use of steroids during an acute exacerbation [3]. Though they are not recommended for routine use, they may provide more benefit in patients with predominant asthma-type symptoms or suspected ABPA [9]. Close consultation with the patient’s pulmonologist if possible is needed to discuss steroid treatment.
Your patient reports growing Pseudomonas from multiple cultures in the past, so you decide to treat her with cefepime and tobramycin. You also give her an albuterol treatment immediately followed by a 7% saline neb. You had considered BiPAP, but her respiratory rate and work of breathing improve following treatment. She is admitted to the hospital for continued IV antibiotics and airway clearance therapy.
Case 2
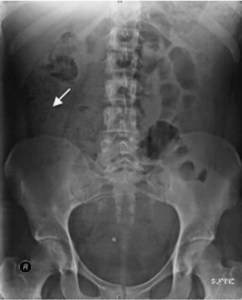
A 12 year old male with cystic fibrosis presents with 3 weeks of progressively worsening crampy right lower quadrant (RLQ) abdominal pain and now non-bilious vomiting and oral fluid intolerance. He had a few episodes of diarrhea yesterday but has since had no bowel movement. He takes pancreatic enzyme replacement therapy and has chronic constipation, for which he uses polyethylene glycol (PEG) 3350 daily. His vital signs are normal for his age except for a heart rate of 120 beats per minute (bpm). His examination is significant for dry mucous membranes and abdominal distention with RLQ tenderness, but no rigidity or guarding. You think you feel a small mass in the RLQ. His abdominal x-ray is shown below:
Distal Intestinal Obstruction Syndrome
Distal Intestinal Obstruction Syndrome (DIOS), previously known as “meconium ileus equivalent,” is an entity unique to CF that is caused by partial or complete obstruction of the ileocecum by inspissated fecal material [22]. It is seen in all age groups, though it is more common in adults and is almost always seen in patients with pancreatic insufficiency [23].
Presentation
DIOS commonly presents with progressive, cramping abdominal pain that usually located in the RLQ or peri-umbilical region. Pain may be acute, but it often precedes the actual obstruction by several weeks or even months. Patients may have abdominal distension first, however. Though DIOS is commonly seen with constipation, it may also be seen with diarrhea or even normal bowel movements [23]. On examination, the inspissated material can usually be felt in the RLQ, although a palpable mass may be present for years without causing obstructive symptoms [24].
Diagnosis
A plain abdominal radiograph is the first line imaging test that should be obtained. It will typically show an accumulation of “bubbly” or “granular” fecal material in the distal ileum [23]. The triad of characteristic abdominal pain, palpable RLQ mass, and distal ileal fecal material on x-ray is usually sufficient to diagnose DIOS. If symptoms or radiographic findings are atypical, or if there is no improvement with treatment, additional imaging such as ultrasound or CT scan should be obtained to evaluate mimics such as appendicitis or intussusception [24].
Differential Diagnosis
It may be difficult to distinguish impending DIOS from chronic constipation by history alone. Constipation usually has a more gradual onset of symptoms with fecal material distributed throughout the colon, as opposed to being localized to the right lower quadrant [22]. However, these entities often coexist and except in severe cases, initial treatment is the same.
Appendicitis should also be on the differential, but unfortunately is sometimes difficult to evaluate. Chronically inspissated mucoid contents may lead to a distended appendix that is difficult to distinguish from an acutely inflamed appendix. This can often lead to a delay in diagnosis of appendicitis, resulting in increased rates of appendiceal perforation and abscess formation in patients with CF. Other causes of bowel obstruction must also be considered such as adhesions, malignancy, or intussusception. Intussusception occurs in about 1% of CF patients and is a common mimic but can also be a complication of DIOS [24].
Management
First, correct any fluid or electrolyte abnormalities and treat any associated infections. After that, treatment is fairly straightforward and involves laxatives administered either orally, by nasogastric tube, or by enema. Patients with incomplete obstructions usually respond to oral therapy with any PEG bowel prep solution such as GoLytely®, Klean-Prep®, or Movicol®. Another option for oral therapy is Gastrografin diluted in water or juice (50mL in 200mL for kids under 6 years, and 100mL in 400mL for everyone else). Therapy is continued until symptoms resolve and bowel movements are clear. Thus, admission may be required [24].
For patients with complete obstruction, PO intolerance, or failure of oral therapy, a nasogastric tube should be placed for decompression, followed by a Gastrografin enema. Since Gastrografin is radiopaque, these enemas can be both diagnostic and therapeutic and may be used to monitor progress [23, 24, 25]. However, these enemas can cause significant fluid shifts as well as intestinal ischemia, perforation, and necrosis, so they should only be administered by an experienced radiologist. These patients all require admission with a low threshold for surgical consultation [24, 26].
You suspect an incomplete obstruction, but he remains PO intolerant. You place an IV to draw labs and give him a 20 cc/kg bolus of LR. After correcting his electrolytes, you place a nasogastric tube for decompression and to administer GoLytely for bowel irrigation. He has a small bowel movement and slight improvement in pain. He is admitted to pediatrics for continued bowel irrigation.
Summary
-CF is not limited to Caucasians and over 33% of cases are missed by newborn screening programs [2].
-Pulmonary exacerbations must be treated early and aggressively to slow the decline in lung function [10].
Treatment of pulmonary exacerbations:
- Antipseudomonal antibiotics (high doses): Aminoglycoside + β-lactam
- Bronchodilator – either MDI or nebulized albuterol
- Nebulized 7% saline, 4mL
- BiPAP if needed’
- Only use steroids if there are significant asthma-type symptoms (i.e. wheezing)
DIOS:
-Diagnosed by classic history, mass in the RLQ, and localized fecal material on plain abdominal film [22].
-Don’t miss appendicitis or intussusception. Get an ultrasound and/or CT scan if needed [24].
-Correct fluids and electrolytes first [24].
-If PO tolerant, may treat from above with PEG solution (e.g. GoLytely®) or Gastrografin diluted 1:4 in water or juice. Patients may require an NG tube in order to consume enough of the laxative [24].
-If PO intolerant or with bilious vomiting (i.e. complete obstruction), decompress the stomach with a nasogastric tube. Call radiology for a Gastrografin enema [24].
-Treat until bowel movements are clear and watery [24].
References / Further Reading
- Rowe SM, Miller S, Sorscher EJ. Cystic Fibrosis. N Engl J Med 2005; 352:1992-2001.
- Cystic Fibrosis Foundation Patient Registry: Annual Data Report to the Center Directors, 2014. https://www.cff.org/2014_CFF_Annual_Data_Report_to_the_Center_Directors.pdf/ (Accessed on June 30, 2016).
- Flume PA, Mogayzel PJ, Robinson KA, et al. Cystic fibrosis pulmonary guidelines: treatment of pulmonary exacerbations. Am J Respir Crit Care Med. 2009 Nov 1;180(9):802-8.
- Bilton D, Canny G, Conway S, et al. Pulmonary exacerbation: towards a definition for use in clinical trials. Report from the EuroCareCF Working Group on outcome parameters in clinical trials. J Cyst Fibros 2011; 10: Suppl. 2, S79-S81.
- Smyth A, Elborn JS. Exacerbations in cystic fibrosis: 3 – Management. Thorax 2008; 63: 180-184.
- Bhatt JM. Treatment of pulmonary exacerbations in cystic fibrosis. Eur Respir Rev. 2013 Sep 1;22(129):205-16. PMID 23997047.
- Flume PA. Pneumothorax in cystic fibrosis. Current Opinion in Pulmonary Medicine 2011. 17(4):220-225.
- Greene KE, Takasugi JE, Godwin JD, et al. Radiographic changes in acute exacerbations of cystic fibrosis in adults: a pilot study. Am J Roentgenol 1994; 163(3):557-62.
- Gibson RL, Burns JL, Ramsey BW. Pathophysiology and Management of Pulmonary Infections in Cystic Fibrosis. Am J Respir Crit Care Med 2003; 168:918-951.
- Dentice, RL, Elkins MR, Middleton PG, et al. A randomized trial of hypertonic saline during hospitalization for exacerbation of cystic fibrosis. Thorax 2016; 71:141-147.
- Hurley MN, Ariff AH, Bertenshaw C, et al. Results of antibiotic susceptibility testing do not influence clinical outcome in children with cystic fibrosis. J Cyst Fibros 2012; 11:288-292.
- Talwalkar JS, Murray TS. The Approach to Pseudomonas aeruginosa in Cystic Fibrosis. Clin Chest Med 2016; 37:69-81.
- Young DC, Zobell JT, Stockmann C, et al. Optimization of Anti-Pseudomonal Antibiotics for Cystic Fibrosis Pulmonary Exacerbations: V. Aminoglycosides. Pediatric Pulmonology 2013; 48:1047-1061.
- Fischer DR, Namanny H, Zobell JT. Follow-up survey of the utilization of anti-pseudomonal beta-lactam antibiotics at U.S. cystic fibrosis centers. Pediatr Pulmonol. 2016 Jul; 51(7):668-9.
- Zobell JT, Young DC, Waters CD, et al. Optimization of Anti-Pseudomonal Antibiotics for Cystic Fibrosis Pulmonary Exacerbations: VI. Executive Summary. Pediatric Pulmonology 2013; 48:525-537.
- Zobell JT, Waters CD, Young DC, et al. Optimization of Anti-Pseudomonal Antibiotics for Cystic Fibrosis Pulmonary Exacerbations: II. Cephalosporins and Penicillins. Pediatric Pulmonology 2013; 48:107-122.
- Doring G, Flume P, Heijerman H, et al. Treatment of lung infection in patients with cystic fibrosis: Current and future strategies. Journal of Cystic Fibrosis 11 (2012):461-479.
- Elkins MR, Robinson M, Rose BR, et al. A controlled trial of long-term inhaled hypertonic saline in patients with cystic fibrosis. N Engl J Med 2006; 354:229.
- Fauroux B. Why, when and how to propose noninvasive ventilation in cystic fibrosis? Minerva Anestesiol 2011; 77:1108-1114.
20. Dovey M, Aitken ML, Emerson J, McNamara S, Waltz DA, Gibson RL. Oral corticosteroid therapy in cystic fibrosis patients hospitalized for pulmonary exacerbations: a pilot study. Chest 2007;132:1212-1218. - Tepper RS, Eigen H, Stevens J, Angelicchio C, Kislin J, Ambrosius W, Heilman D. Lower respiratory illness in infants and young children with cystic fibrosis: evaluation of treatment with intravenous hydrocortisone. Pediatr Pulmonol 1997;24:48-51.
- Houwen RH, van der Doef HP, Sermet I, et al. Defining DIOS and constipation in cystic fibrosis with a multicenter study on the incidence, characteristics, and treatment of DIOS. J Pediatr Gastroenterol Nutr 2010; 50:38
- Houwen RH, van der Doef HP, Sermet I, et al. Defining DIOS and constipation in cystic fibrosis with a multicenter study on the incidence, characteristics, and treatment of DIOS. J Pediatr Gastroenterol Nutr 2010; 50:38.
- Khoshoo V, Udall JN Jr. Meconium ileus equivalent in children and adults. Am J Gastroenterol 1994; 89(2): 153.
- Colombo C, Ellemunter H, Houwen R, et al. Guidelines for the diagnosis and management of distal intestinal obstruction syndrome in cystic fibrosis patients. J Cyst Fibros 2011; 10 Suppl 2:S24.
- Nash EF, Ohri CM, Stephenson AL, and Durie PR. Abdominal pain in adults with cystic fibrosis. European Journal of Gastroenterology & Hepatology 2014, 26:129-136.
- Voynow JA, Mascarenhas M, Kelly A, Scanlin TF. Cystic Fibrosis. In: Grippi MA, Elias JA, Fishman JA, Kotloff RM, Pack AI, Senior RM, Siegel MD. Eds. Fishman’s Pulmonary Diseases and Disorders, Fifth Edition. New York, NY: McGraw-Hill; 2015. http://accessmedicine.mhmedical.com/content.aspx?bookid=1344&Sectionid=81189522. Accessed August 06, 2016.


Great post! Will reference for my upcoming lecture to the EM residents at my institution.