Today on the emDOCs cast with Brit Long, MD (@long_brit), we look at updates in Clostridioides difficile infection.
Episode 54: Clostridioides difficile infection updates
Background:
- Clostridioides difficile is an anaerobic Gram-positive, spore-forming, enterotoxin-producing bacterium. It’s transmitted by fecal or oral route and leads to tissue injury and death of intestinal cells, particularly epithelial and inflammatory cells of the GI system (resulting in diarrhea, GI symptoms).
- While CDI rates have decreased since 2010, domestic rates still average approximately 500,000 cases annually.
- Current data suggest 10% of patients in the ED with diarrhea are ultimately found to have CDI, and a recent 8-year search of the Nationwide Emergency Department Sample found over 900,000 emergency department (ED) visits with the primary diagnosis of CDI.
- In 2014, CDI replaced methicillin-resistant Staphylococcus aureus (MRSA) as the most common hospital-acquired infection in the U.S.
Risk Factors:
- Focus on 2 components: exposure and increased susceptibility to C. difficile.
- Exposure risks center on contact with contaminated individuals or clinical settings
- Susceptibility is the other risk factor. One of the most important risk factors is antibiotic exposure. Abx disrupt normal gut microbiota and enable C. difficile seeding in newly-exposed individuals as well as germination of pre-existing dormant C. difficile spores in carriers.
- Fluoroquinolones (OR 5.5-16.7), beta-lactam and beta-lactamase inhibitors (OR 2.7), third- and fourth-generation cephalosporins (OR 1.6-14.9), carbapenems (OR 1.5-5.7), and clindamycin (OR1.8-31.8).
- While 10 days of antibiotics classically associated, may occur in even single doses.
- Recent use includes those receiving antibiotics within 3 months of CDI presentation.
- Up to 40% of CDI patients lack a history of recent antibiotic use.
- Age > 65 years increased risk (5-fold).
- Recent healthcare exposure, IBD (3% risk in 5 years), and immunosuppression are risks.
- HIV, recent GI surgery, tube feedings, malnutrition, obesity, female sex, and low serum albumin.
- Proton pump inhibitor use likely increases risk, but many confounders in the available evidence.
Categories and Presentation
- CDI can cause a spectrum of manifestations ranging from an asymptomatic carriage to fulminant disease with toxic megacolon.
- Overall: abdominal pain (6-90%), fever (21-50%), bloody stools (5-21%), and watery diarrhea (55-84%).
- Classified into nonsevere, severe, and fulminant. Classification based on labs (Cr, WBC) and patient hemodynamics.
- Classic presentation: diffuse, watery stools and recent antibiotic use and/or hospitalization.
- Nonsevere disease: Watery diarrhea (≥3 loose stools in 24 hours) is the cardinal symptom of CDI. Other manifestations include lower abdominal pain and cramping, low-grade fever, nausea, and anorexia. Diarrhea may be present with mucus or occult blood, but melena or hematochezia are rare. Fever >38.5°C is associated with CDI in about 15% of cases.
- Diarrhea usually lasts weeks to months. Diarrhea may not be isolated. CDI may present with nausea and vomiting in 2-30% of patients.
- Cr < 1.5 mg/dL and WBC < 15,000.
- Severe colitis: Clinical manifestations include diarrhea, lower quadrant or diffuse abdominal pain, abdominal distention, fever, hypovolemia, lactic acidosis, hypoalbuminemia, elevated Cr, leukocytosis (WBC of 40,000 cells/mL or higher), elevated lactate.
- Defined by Cr > 1.5 mg/dL, WBC > 15,000.
- Fulminant colitis: Abdominal distention, shock or early vasopressor requirement.
- In 1-3% of CDI cases, patients can have a delayed presentation resulting in a fulminant colitis; these numbers may be increasing due to hypervirulent strains.
- Ileus can occur in severe forms. Present in 20% of CDI patients with fulminant colitis in a single 12-year study.
- Megacolon should be suspected in patients with severe systemic toxicity together with radiographic evidence of large bowel dilatation (>6 cm diameter in the colon and/or >12 cm diameter in the cecum).
- Radiographic findings of ileus and toxic megacolon should raise concern for CDI in at-risk patients, regardless of whether diarrhea is present.
- Recurrent disease: Defined by resolution of CDI symptoms while on appropriate therapy, followed by reappearance of symptoms within 2-8 weeks after treatment has been stopped.
Evaluation/Testing
- CBC, electrolytes, lactate, LFTs can assist in assessing disease severity and evaluate for other conditions/complications.
- IDSA guidelines recommend CDI testing in patients with 3 or more unformed stools in a 24-hour period.
- Use presentation and risk factors to determine need for testing, especially for diarrhea lasting over 1 week and there’s no other explanation.
- CDI testing centers on detection of the organism itself and the toxins (A and B) produced. We used to use the cell cytotoxicity neutralization assay (CCNA) for detecting toxin B as the diagnostic “gold standard,” but this is no longer used.
- Organism detection: nucleic acid amplification tests (NAAT) detect toxin genes.
- This includes PCR.
- Highly sensitive but cannot distinguish infection from asymptomatic colonization. NAATs may lead to overdiagnosis of CDI.
- Glutamate dehydrogenase (GDH) enzyme immunoassays (EIA): Detects the metabolic enzyme that is found in all strains of C. difficile, but this has low specificity. Can’t differentiate between toxigenic/nontoxigenic strains.
- Toxin production: Toxin EIA.
- Toxin EIA for A and B sensitivity ranges between 51-88%. Specificity is 99%.
- General consensus is lacking as to which laboratory test is best. IDSA emphasizes using a two or three step algorithm as agreed upon between individual institutions and their clinical laboratory (e.g., EIA for GDH plus EIA for toxin; EIA for GDH plus EIA for toxin plus NAAT; NAAT plus EIA for toxin).
- One sample algorithm: Start with NAAT/PCR. If NAAT/PCR negative, you can rule out disease. If NAAT/PCR positive, proceed with toxin EIA testing. If this is positive, treat patient. If negative, isolate patient but don’t treat. This is likely a carrier.
- Caveat: If immunocompromised and NAAT/PCR positive, treatment is warranted.
- If a patient can’t provide a stool specimen, send them home and have them bring back the sample.
Severity assessment
- Several scoring systems can be used to assess the severity of CDI and guide initial treatment.
- The ATLAS scoring system includes 5 parameters that are each scored as 0-2 points:
- Age (<60 y, 0 points; 60-79 y, 1 point; ≥80 y, 2 points)
- Treatment with systemic antibiotics – yes 2 points; no 0 points
- Leukocytosis (<16,000, 0 points; 16,000-25,000, 1 point; >25,000, 2 points)
- Albumin (>35 g/L, 0 points; 26-35 g/L, 1 point; <25 g/L, 2 points)
- Serum Creatinine (<1.3mg/dL 0 points; 1.4-2.0mg/dL 1 points;>2.1mg/dL 2 points)
- Ghosh identified several indicators of moderate to severe CDI:
- Leukocytosis (white blood cell count >20 × 109/L)
- Albumin level <30 g/L
- Creatinine level >50% baseline
- Vital sign instability (SBO <100 mm Hg, temperature >38°C)
- Abdominal pain/distension
- Ascites, colonic distension, or ileus on radiology studies
- Summary: Use hemodynamics, systemic toxicity, elevated WBC/Cr/lactate, presence of megacolon.
- The ATLAS scoring system includes 5 parameters that are each scored as 0-2 points:
Imaging:
- No clear recommendations on when diagnostic imaging should be used in CDI.
- The World Society of Emergency Surgery recommends CT “for patients with clinical manifestations of severe-complicated C. difficile colitis” without formerly defining “severe-complicated”.
- Patients with evidence of severe disease (severe abdominal pain, abdominal distention with apparent ileus, fever, hypovolemia, lactic acidosis, hypoalbuminemia, and/or marked leukocytosis), concern for another condition, history of abdominal surgery/obstruction, medical complications (such as diabetes, ulcerative colitis, or Crohn’s disease) should undergo CT imaging.
- CT may show pronounced colonic wall thickening and low-attenuation mural thickening (due to mucosal and submucosal edema, which may be visible as a “target sign” or “double halo sign” consisting of two or three concentric rings of different attenuation). Pericolonic stranding and ascites may occur but are not specific.
- Toxic megacolon: colonic dilatation (>6 cm in diameter) is diagnostic; other findings include small bowel dilatation, air-fluid levels (mimicking an intestinal obstruction or ischemia), and “thumb printing” (scalloping of the bowel wall) due to submucosal edema.
Therapy
- Empiric treatment without confirmation testing is not recommended.
- If confirmed positive, stop unnecessary antibiotics. First line used to be metronidazole. This is no longer the case due to increasing resistance (rates as high as 18%).
- First line is fidaxomicin per 2018 IDSA guidelines, or oral vancomycin if fidaxomicin not available.
- In 2011, fidaxomicin was FDA-approved for the treatment of CDI and is currently supported by IDSA guidelines in treatment of an initial episode of CDI.This recommendation is supported by two large-scale RCTs involving over 1100 patients and demonstrating near-equal efficacy of oral vancomycin vs. fidaxomicin on clinical resolution of symptoms at 10 days.
- Unfortunately, fidaxomicin is very expensive, and most use oral vancomycin.
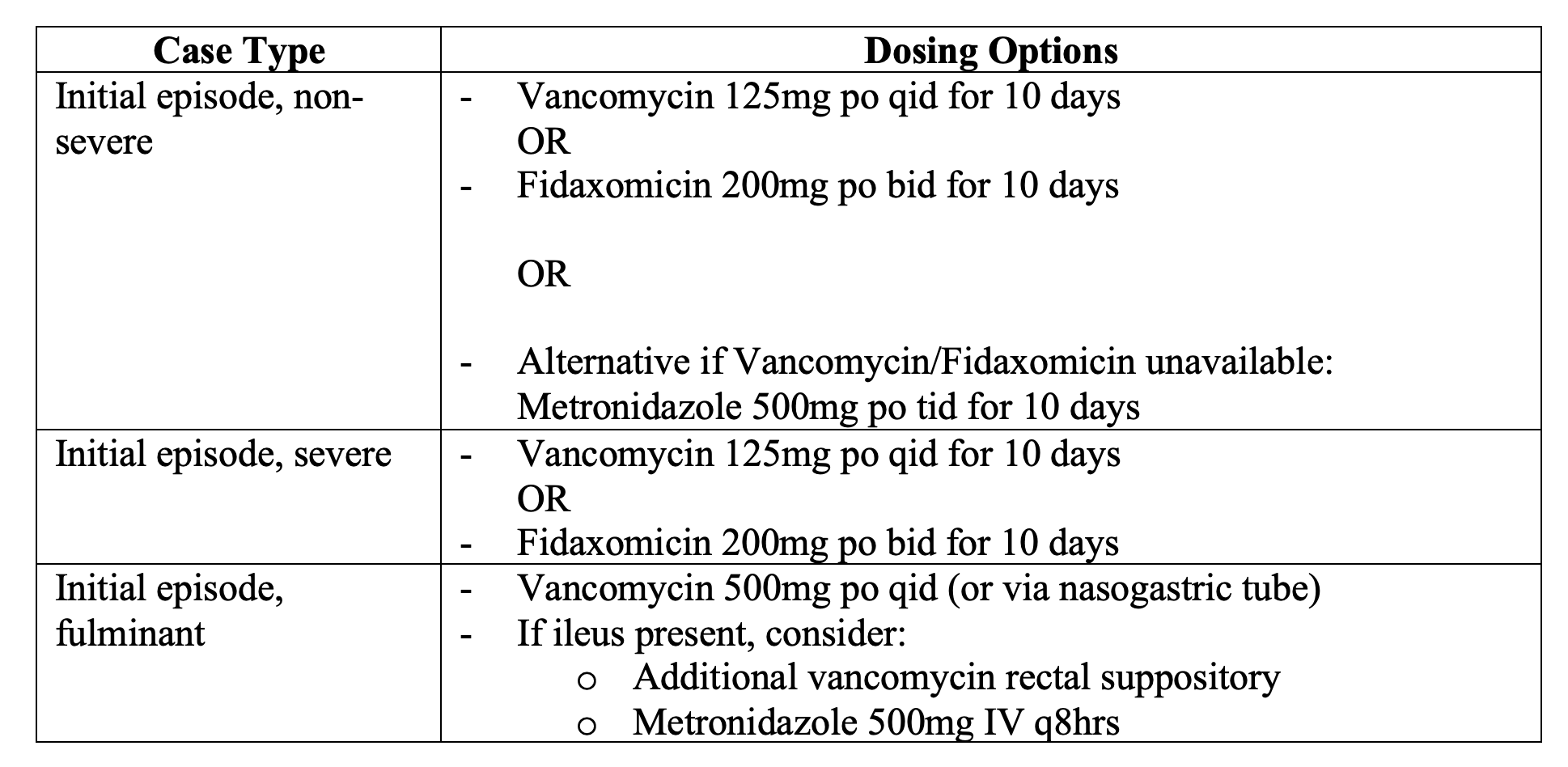
- Metronidazole is an option for initial treatment of CDI if vancomycin and fidaxomicin are unavailable.
- Oral antibiotic regimens for a duration of 10 days resolve symptoms in the majority of CDI patients but can be extended to 14 days if symptoms are slow to improve.
- Fulminant: Consult surgery due to concern perforation, peritonitis, and septic shock. Surgical treatment can include total abdominal colectomy or a diverting loop ileostomy with colonic lavage.
- High doses of oral/NG and rectal vancomycin should be administered in ED, contacting not only surgery but also GI. Endoscopic stool transplant having been shown to be surgery-sparing and life-saving and can be tried initially.
- IDSA – For fulminant CDI*, vancomycin administered orally is the regimen of choice (strong recommendation, moderate quality of evidence). Most experts recommend rectal administration as well as a suspension enema, and this will be the primary route tf ileus is present. The vancomycin dosage is 500 mg orally 4 times per day and500 mg in approximately 100 mL normal saline per rectum every 6 hours as a retention enema. Metronidazole should also be given IV in doses of 500 mg every 8 hours with the oral and rectal vancomycin.
Prevention/Isolation
- Alcohol-based hand sanitizers are less effective than handwashing with soap and water.
- C. difficile spores are resistant to alcohol solutions and have been shown in some instances to be equivalent to no hand hygiene at all.
- Gowns and gloves are required whenever entering the room of a patient in enteric precautions isolation to prevent contamination of garments/skin and the spread of disease.
- Hand washing with soap and water is the most effective strategy to prevent transmission; this is done after removing gown and gloves.
- Disinfect shared equipment such as stethoscopes, wrist band, ID scanners and others with bleach
- Frequently touched surfaces in the room and equipment removed from the room should be disinfected with bleach wipes. Terminal cleaning includes Ultraviolet light treatment.
Further Reading:








