Author: Brit Long, MD (@long_brit; EM Resident at SAUSHEC; USAF) // Editor: Alex Koyfman, MD (@EMHighAK) & Justin Bright, MD (@JBright2021)
A 42 year-old female with poorly controlled type II diabetes presents with several days of fatigue, headache, fever to 102F, and sinus pressure. She does not regularly check her blood glucose, with her last check at 320. Initial vital signs show a blood pressure of 155/92, heart rate of 92, oxygen saturation of 97% on room air, temperature of 102.2, and respiratory rate of 24. Initial D-stick is 330. You order some labs, which reveal an anion gap of 22, bicarbonate of 11, with glucose of 322 and potassium of 4.2. Your ECG obtained is normal. You begin your standard treatment for diabetic ketoacidosis. As you begin to go through your algorithm for management/treatment of DKA, you question why the patient is in DKA. You remember that she has had several days of headache, fever, and sinus pressure. You go back into the room after the 1L bolus to complete your exam, and what you see on your exam surprises you:
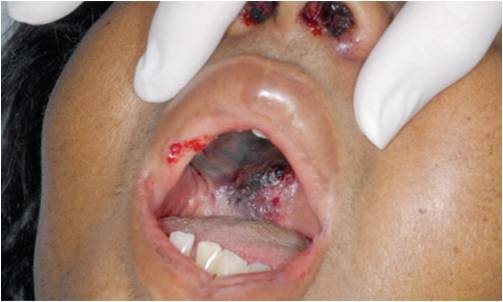
You immediately call your ICU to update them, as you feel this is consistent with Rhinocerebral mucormycosis!
Introduction: Mucormycosis can present in a variety of ways, primarily in immunocompromised patients. Rhino-orbital-cerebral and pulmonary infections are most common.1 These fungi are found naturally on decaying vegetation and soil. They grow rapidly and release large numbers of airborne spores. All humans have frequent exposure to the fungal species in the environment, but a normal immune system is quickly able to destroy the fungi. Genera include Rhizopus and Mucor most commonly. For those of you who love the study of fungi, the hyphae are broad with irregular branching and rare septations.1-3
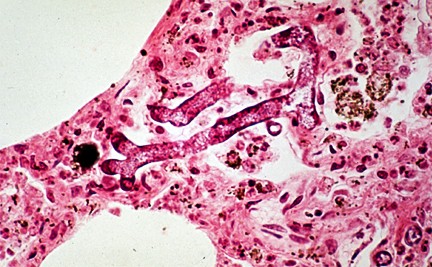
These fungi contain ketone reductase, with allows the organisms to grow rapidly in environments with high sugar and low pH.4 The fungi also contain a siderophore that increases iron uptake, stimulating growth.5 Once an individual inhales the spores, cilia transport them to the pharynx, lungs, and gastrointestinal tract. Most individuals clear the organisms. Susceptible patients suffer infections that begin in the nasal turbinates and alveoli. The organism can then spread. The fungi invade blood vessels, allowing rapid spread but can cause infarction of infected tissues.1,3
Epidemiology: By far the most common risk factor is diabetes, especially with ketoacidosis. Chronic steroid use, hematologic malignancies, stem cell/solid organ transplant patients, deferoxamine treatment (other iron chelating agents do not increase risk), iron overload, injection drug use, and trauma all predispose to infection. The fungi grow well in acidotic, hyperglycemic, and iron-containing environments.1-8
Presentation: Mucormycosis results from vascular invasion by hyphae, causing infarction and necrosis of tissues. For the rhino-orbital-cerebral form, which is most common, inhalation of spores allows bacteria to invade. Sinusitis (26%), fever (44%), congestion, nasal discharge, and headache (25%) are all initial symptoms. Unfortunately, most cases will progress rapidly. The classic case is palatal necrosis with eschar formation (38% of cases), turbinate destruction, nasal swelling (34%), and erythema of the face. If the orbits are involved, edema, proptosis, and decreased vision (30%) or blindness can result.6,7 If intracranial spread occurs, mental status changes are prevalent. Cranial nerve palsies are also common. The most common underlying comorbidity is diabetes in this form (up to 70% of this form is correlated with diabetics).1,8
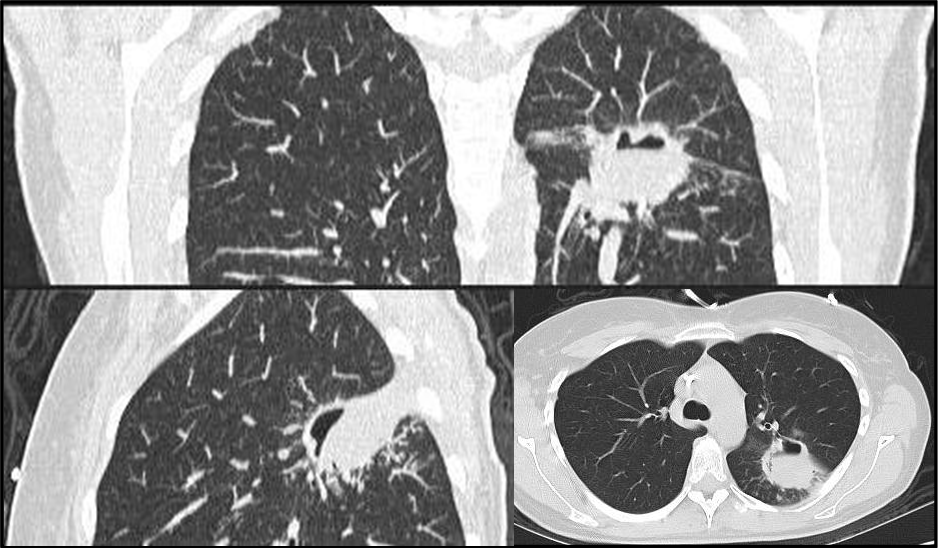
The pulmonary form is rapidly progressive and occurs after spores reach alveoli and bronchioles. Pneumonia and infarction set in, often resulting in hemoptysis. From the lungs the organisms can spread to the mediastinum and heart. In this form, iron supplementation, glucocorticoid treatment, and malignancies are the most common underlying conditions (diabetes is less common).1,3,9
The gastrointestinal tract can also be involved, though much rarer, with the stomach most commonly affected, followed by the colon. Hematemesis and pain are presenting symptoms. Perforation, peritonitis, and bleeding can result from ulcer formation.1,3,10
Cutaneous mucormycosis usually is due to wound inoculation. The lesion starts as a small bite-like lesion that develops into cellulitis.
Renal, isolated CNS (IV drug use, HIV patients), and disseminated forms are also present.1-3
Diagnosis: One important key is that if a patient is high risk and has symptoms/signs of this disease (diabetic with sinusitis, fever, infarcted palatal/nasal tissue), then treat first! The ultimate diagnosis requires culture confirmation with slide preparation to identify the organism, which shows nonseptate hyphae. In patients with rhino-orbital-cerebral infection, endoscopic imaging is needed. CT head/orbits is also useful. For pulmonary infection, chest Xray and/or CT often demonstrates focal consolidation, mass, pleural effusion, or nodules. Both the pulmonary and GI forms require tissue sampling. PCR is currently being investigated for use as definitive diagnosis. Ultimately, it comes down to your exam and history. If the patient is immunocompromised/has risk factors and your exam findings support the diagnosis, begin treatment.1-3,11,12
Treatment: Like in severe sepsis, treatment revolves around source control with surgical debridement and antifungal therapy. Predisposing factors such as acidosis, hyperglycemia, deferoxamine, and immunosuppression should be addressed. For example, patients in the setting of DKA with mucormycosis do show improved outcomes with better hydration and blood glucose control.
The antifungal most widely used is IV amphotericin B (lipid) at 5mg/kg daily. Posaconazole is used as step-down therapy or as salvage therapy. One study demonstrated that patients with delayed antifungal treatment (greater than six days) had double the mortality! Debridement is also required as soon as possible to remove the source of infection. 1-3,12
Prognosis: Outcomes vary based on time to treatment. Mortality rate for the pulmonary form is up to 87%, with the rhino-orbital-cerebral form possessing a mortality of 25-62%.3
Summary: Mucormycosis has a variety of forms: rhino-orbital-cerebral, pulmonary, and GI. Immunosuppressed patients, particularly diabetics and patients with hematologic malignancies, make up the majority of patients. The organisms are found all over, and normal hosts are able to fight off infection. If infection occurs, fungi invade tissue, causing infarction and necrosis with vascular invasion. Diagnosis requires organism identification by histopathology, but if suspected, begin treatment with lipid formulation of amphotericin B. Surgical debridement is the second necessary arm of treatment.
References/Further Reading:
- 1. Kauffman CA, Malani AN. Zygomycosis: an emerging fungal infection with new options for management. Curr Infect Dis Rep 2007; 9:435.
- Spellberg B, Walsh TJ, Kontoyiannis DP, et al. Recent advances in the management of mucormycosis: from bench to bedside. Clin Infect Dis 2009; 48:1743.
- Roden MM, Zaoutis TE, Buchanan WL, et al. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis 2005; 41:634.
- Gale GR, Welch AM. Studies of opportunistic fungi. I. Inhibition of Rhizopus oryzae by human serum. Am J Med Sci 1961; 241:604.
- Boelaert JR, de Locht M, Van Cutsem J, et al. Mucormycosis during deferoxamine therapy is a siderophore-mediated infection. In vitro and in vivo animal studies. J Clin Invest 1993; 91:1979.
- Ferguson BJ. Mucormycosis of the nose and paranasal sinuses. Otolaryngol Clin North Am 2000; 33:349.
- Yohai RA, Bullock JD, Aziz AA, Markert RJ. Survival factors in rhino-orbital-cerebral mucormycosis. Surv Ophthalmol 1994; 39:3.
- McNulty JS. Rhinocerebral mucormycosis: predisposing factors. Laryngoscope 1982; 92:1140.
- Lee FY, Mossad SB, Adal KA. Pulmonary mucormycosis: the last 30 years. Arch Intern Med 1999; 159:1301.
- Agha FP, Lee HH, Boland CR, Bradley SF. Mucormycoma of the colon: early diagnosis and successful management. AJR Am J Roentgenol 1985; 145:739.
- Hammond SP, Bialek R, Milner DA, et al. Molecular methods to improve diagnosis and identification of mucormycosis. J Clin Microbiol 2011; 49:2151.
- Walsh TJ, Gamaletsou MN, McGinnis MR, et al. Early clinical and laboratory diagnosis of invasive pulmonary, extrapulmonary, and disseminated mucormycosis (zygomycosis). Clin Infect Dis 2012; 54 Suppl 1:S55.
- http://www.ncbi.nlm.nih.gov/pubmed/10958140
- http://www.ncbi.nlm.nih.gov/pubmed/10431954




