Authors: Tony Spadaro, MD, MPH (EM Resident Physician at the Hospital of the University of Pennsylvania) and Kevin R. Scott, MD, MSEd (EM Attending Physician at the Hospital of the University of Pennsylvania) // Reviewed by: Richard Byyny, MD; Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case
A 43-year-old female with a history of migraines presents to the emergency department with a headache. She reports the headache came on much faster, reaching a maximal intensity in less than one minute. Her pain began while she was running. She had an episode like this last week and went to a local ED. She had a negative non-con head CT and was discharged with a prescription for metoclopramide. Prior to her initial ED visit, she has otherwise been in her usual state of health, although her seasonal allergies have been worse lately and she has been using a pseudoephedrine containing nasal decongestant prior to running or working out.
Overview
Reversible Cerebral Vasoconstriction Syndrome (RCVS) is a rare cause of thunderclap headache (TCH), estimated cause 8-45% of TCH, once aneurysmal subarachnoid hemorrhage (SAH) is ruled out.1 The true incidence of RCVS is not known. The incidence appears to be rising; however, it is unclear if that is due to increased physician awareness, improvements in imaging, or changing prevalence of risk factors.1 Reversible Cerebral Vasoconstriction Syndrome was first recognized in the 1960s.1 The International Headache Society defines RCVS as being characterized by severe acute headaches with or without neurologic signs and symptoms. The frequency and severity of symptoms usually gets worse after symptoms first onset, but then stabilizes or regresses with no new symptoms after one month of onset. Clinical evaluation of patient’s with RCVS reveals no evidence of aneurysmal subarachnoid hemorrhage, normal or near-normal CSF findings, and multifocal segmental cerebral artery vasoconstriction visible on angiography with reversibility within 12 weeks after onset.1 Initially RCVS was thought to be a diverse set of distinct clinical entities including benign angiopathy of the central nervous system (CNS), migrainous vasospasm, sexual headache, thunderclap headache with vasospasm, postpartum angiopathy, and drug-induced angiopathy. These various disease entities were eventually classified as RCVS due to their similar pathophysiology.1
Cerebral catheter angiography is the diagnostic gold standard, but this diagnostic tool is invasive and has significant risks including stroke. The risks of this diagnostic tool and subsequent higher threshold for use probably led to the initial under recognition of the disease.2 In 1988, advances in MRI and CT imaging technologies allowed for radiologists to describe the radiographic findings that became known as Call-Fleming syndrome.1 One study found that patients with RCVS encountered physicians 4.7 times before receiving an accurate diagnosis with a mean duration of symptoms until diagnosis of 9.3 days.3 As there is growing recognition of this syndrome and the possibility of missed diagnosis, it is important for the emergency medicine clinician to be familiar with the presentation, diagnosis, and therapy as it can lead to stroke, disability, and even death.
Pathophysiology
The exact pathophysiology of RCVS is not entirely understood.1 Leading theories include alterations in cerebral vascular tone induced by sympathetic overactivity, endothelial dysfunction, and oxidative stress.1,4,5 Cerebral blood vessels are densely innervated with sensory afferent neurons from the trigeminal nerve and it is speculated that rapid changes in cerebral blood vessel tone may cause nerve ischemia and contribute to the severe headache of RCVS; however, the headache in RCVS may precede radiographic evidence of vasoconstriction.1,5
Many of the risk factors for RCVS, discussed in more detail below, are thought to alter cerebral vasculature tone through adrenergic activation.1 Other risk factors are thought to increase risk for RCVS due to alterations in the balance of pro- and anti-angiogenic factors such as placental growth factor that lead to impaired vascular tone.1 In a study of patients with thunderclap headache without aneurysmal SAH, MRI evidence of blood brain barrier breakdown was more common among patients with RCVS.6 A related disease entity to RCVS, Posterior Reversible Encephalopathy Syndrome (PRES), shares a pathophysiological basis in impaired cerebral vascular tone.5 In contrast to the vasoconstriction seen in RCVS, in PRES there is an increase in cerebral blood flow that leads to endothelial dysfunction, impairment of the blood brain barrier, and ultimately cerebral hypoperfusion.5,6 Although PRES is a distinct clinical entity, it can overlap with RCVS. In fact, in observational studies of RCVS the rate of PRES ranges from 3.6-28%.6-13 There has also been noted an association between RCVS and carotid and vertebral artery dissection, thought to be due to impaired autoregulation of cerebral blood flow after dissection.14 In one study of RCVS patients 12% of patients had concomitant carotid or vertebral artery dissection.14
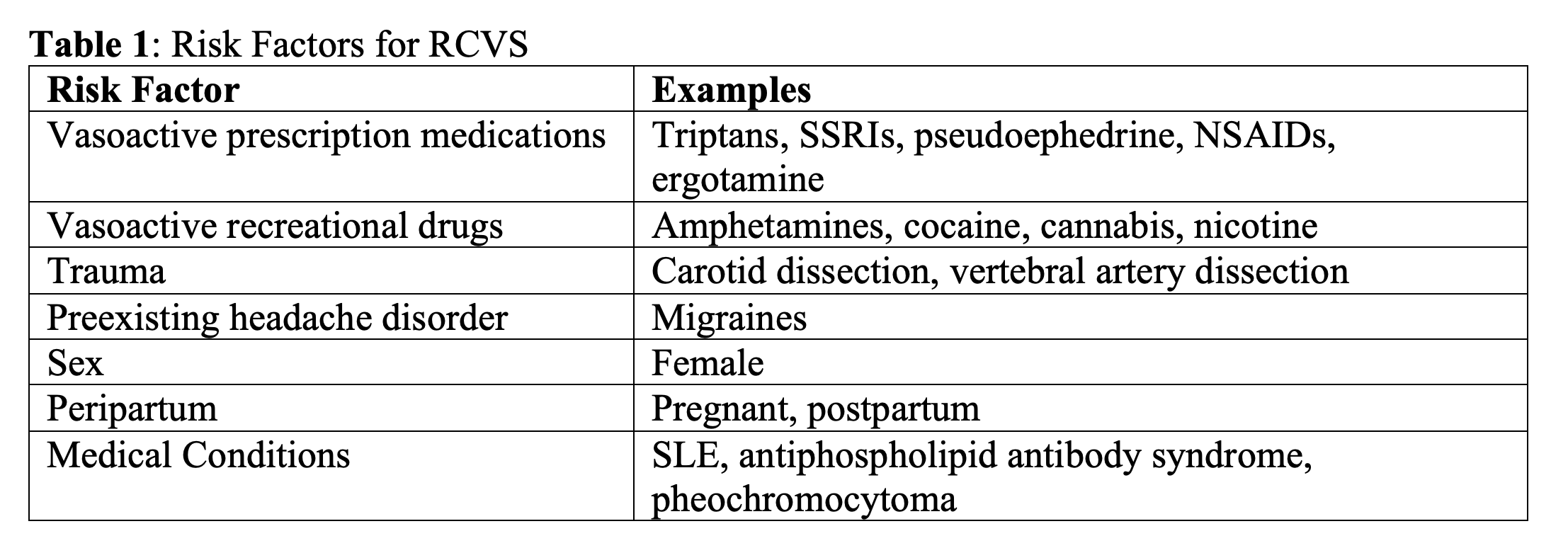
Risk Factors
RCVS predominately occurs in females with a predominance of approximately 70.7-89.6% and at a median age of 43.5-50.2 years. 6-13 Although RCVS is not as well described in the pediatric population, a 2017 study found that 11 of 13 (84.6%) reported cases of RCVS at that time were in males.15 An estimated 25-60% of patients with RCVS have an identifiable trigger.1 Common triggers for RCVS include vasoactive substances, exertion, coughing, showering, and sexual activity (Table 1).1,8,16,17 There is an association between migraines and RCVS, with 20-40% of cases of RCVS having a history of migraines.1 Further complicating matters is that triptans, which are commonly used to treat migraines, and other vasoactive prescription medications are also associated with RCVS.1,2,18
History and Physical Exam
The most common presenting symptom of RCVS is thunderclap headache (TCH).2 TCH is defined as a severe headache reaching peak intensity in less than one minute.2 Although there are no emergency department (ED) based studies of the prevalence of RCVS among TCH patients, a single study conducted at a headache clinic found that 11/24 patients (45.8%) with TCH had RCVS.19 However, caution should be used in generalizing these results to ED patients as the prevalence of SAH was 3.3% of clinic patients, compared to ED based studies that show rates of 6-25% among patients presenting with TCH.19 Among patients presenting with TCH for whom aneurysmal SAH has been ruled out, an ED-based prospective study of 34 patients with TCH, who had an initial head CT that was negative for SAH, all underwent MR angiography (MRA), lumbar puncture (LP), and serial transcranial dopplers. Three of thirty-four patients (8.8%) were ultimately diagnosed with RCVS.16
Although a single TCH is possible in RCVS, most patients usually have multiple episodes, which are typically more short lived, 1-3 hours, then the thunderclap headaches of SAH, which tend last for greater than 48 hours.4,20 A patient who has had multiple acute care visits for thunderclap headache within a brief period of time and ongoing recurrent symptoms should raise suspicion for RCVS.3 Although TCH is the most common described headache, not all RCVS patients will present with this classic description. An observational study in Korea found that 10.6% of patients presented with severe headache of more gradual onset, with the median time of maximal intensity of five minutes (range 2-120 minutes).11 Of note, in this study, 2.9% of patients with RCVS did not have any headache at all.11
Adding to the difficulty of this diagnosis is that there is an association between RCVS and migraines with 9.8-42% of RCVS patients having a history of migraines. 6-13 One study reported that migraineurs with RCVS often identify the TCH of RCVS as different than their usual headache.21 Patients with RCVS may also present with seizures or other nonspecific complaints such as neck pain or focal neurologic deficits. In prospective observational studies, the rate of seizures in RCVS has been between 1.2-5% of patients, while retrospective studies have demonstrated rates as high as 16.7%.6-13
Carotid and vertebral artery dissection are associated with RCVS, and neck pain in a headache patient, particularly after trauma or neck manipulation, should also lead to the consideration of RCVS.1,14 In the largest case series exploring the association between RCVS and cervical artery dissection there were cases of both spontaneous and traumatic dissection.14Typically the complaint of TCH, seizure, or a focal neurological deficit will trigger further workup in the ED. Particular attention to the history and any risk factors for RCVS will be important for consideration in directing the evaluation and management of these patients.
The physical exam in patients with RCVS is often unrevealing. Vital signs are also usually unremarkable, 67-89.2% will have normal blood pressure.6-13 Focal neurologic deficits from transient cerebral ischemia, ischemic infarct, and cerebral hemorrhage can occur in RCVS.20 One retrospective study of 139 RCVS patients found that 43% had focal neuro deficits including aphasia, hemiparesis, ataxia, or visual field deficits.22 Other studies estimate the range of focal neurologic deficits in RCVS to be between 8-43%.20
Differential Diagnosis
The differential diagnosis of TCH is broad, including aneurysmal SAH, cerebral venous thrombosis (CVT), PRES, intracranial hypotension, cerebral or vertebral artery dissection, pituitary apoplexy, third ventricle colloid cyst, migraine, and RCVS.23 Table 2 lists clinical features of diseases included in the differential diagnosis of RCVS.

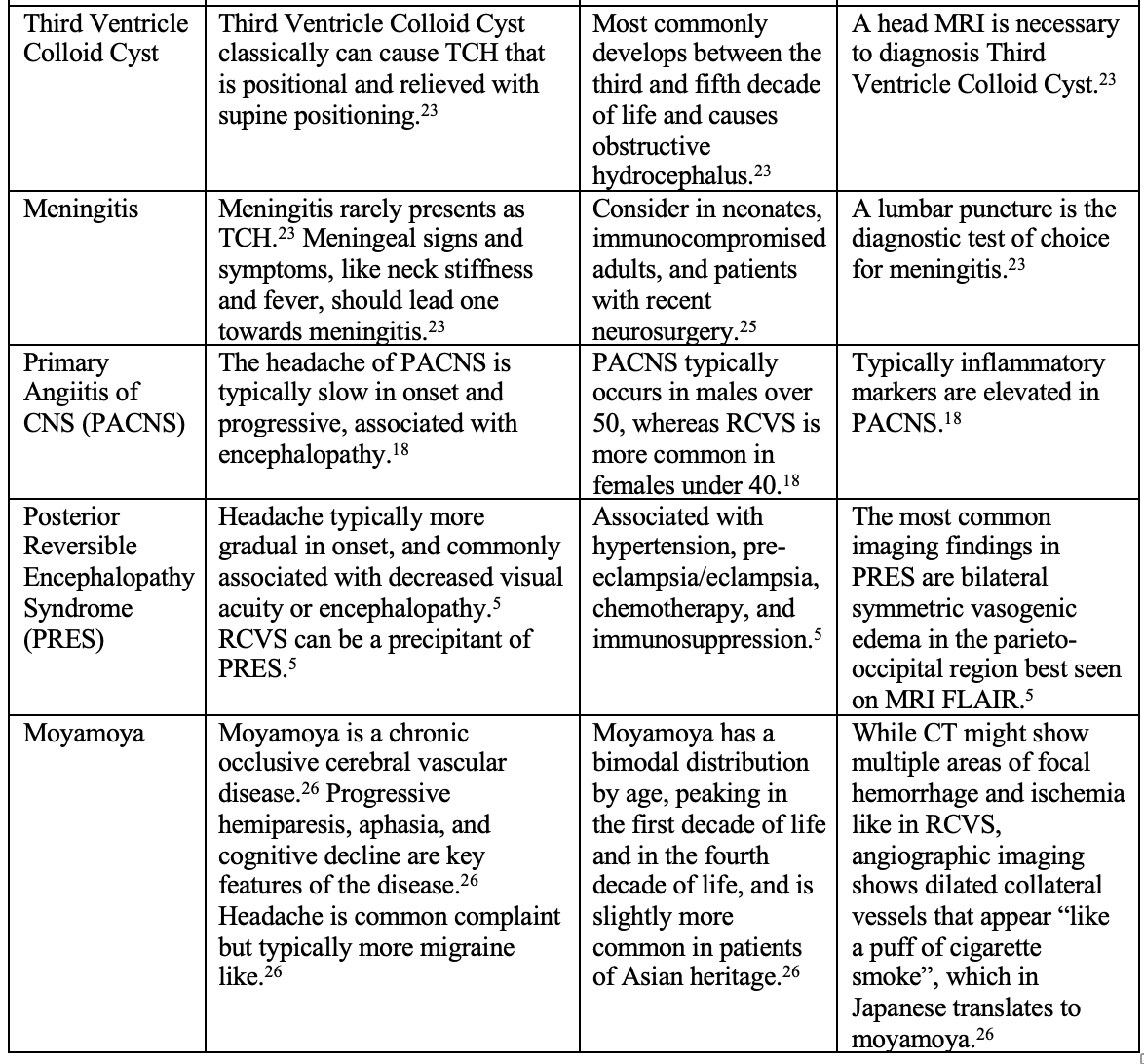
PACNS is a challenging clinical entity for neurologists and neuroradiologists to distinguish from RCVS, and in ambiguous case can lead to brain biopsy as the treatment is different.22 Although less likely to present with TCH, PACNS shares many radiographic features with RCVS.22 PACNS is an autoimmune vasculopathy associated with many rheumatologic conditions such as systemic lupus erythematosus (SLE) and rheumatoid arthritis.8 PACNS typically presents as a slow, insidious onset headache associated with encephalopathy and cognitive dysfunction.18 One study found a single TCH has a sensitivity of 15% and specificity of 96% for RCVS over PACNS and recurrent TCH had a sensitivity of 74% and specificity of 98% for RCVS over PACNS.10 Additionally one study found that upon hospital admission, head CT or MRI was abnormal in all patients with PACNS and abnormal in only 31% of patients with RCVS (p < 0.0001).8 This distinction is important as steroids are the treatment of choice in PACNS, and one retrospective cohort study found that of 159 RCVS cases, the four cases that resulted in death were misdiagnosed as PACNS and treated with steroids.18 An additional study found that use of glucocorticoids in RCVS was associated with a trend for poorer outcome.22
As discussed above the differential diagnosis for RCVS includes some common and some less common diseases with clinical and radiologic overlap. Important to making the diagnosis of RCVS is first considering it, see table 3 for scenarios that should raise the consideration of RCVS.

Diagnostics
The radiographic finding of multifocal cerebral artery vasoconstriction is a key diagnostic feature in RCVS.1 Cerebral catheter digital subtraction angiography (DSA) is considered the gold standard.2 While DSA can also be therapeutic with intra-arterial injection of vasodilators, it is rarely performed due to the invasive nature and a complication rate of 1-5%.2 CTA and MRA are both potential non-invasive modalities for making the diagnosis. However, CTA and MRA only have estimated sensitivities of approximately 80%.2 Findings on initial CTA and MRA have been estimated to be normal in 33%-55% of patients.22,27 Maximal vasoconstriction of cerebral vessels is typically seen approximately 16 days after symptom onset.20
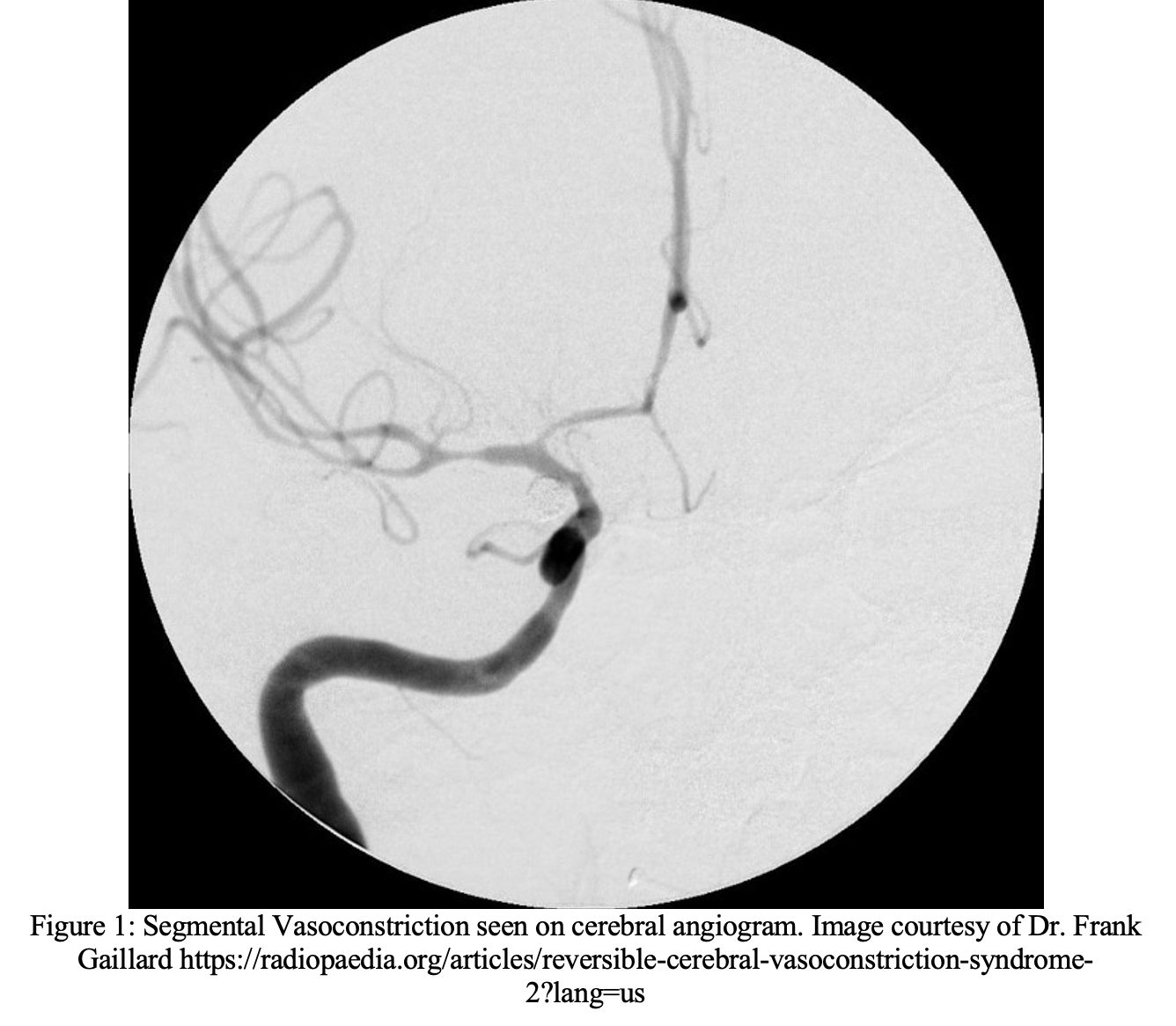
The classic radiographic findings of RCVS are the “sausage on a string” or “string and beads” pattern of focal segmental vasoconstriction (Figure 1).26 In RCVS these findings will have resolved after 3 months and the segmental vasoconstriction will no longer be detectable.1,28 Transcranial doppler can be used to diagnose and monitor RCVS; however, the sensitivity varies depending on the arteries involved with sensitivities as high as 67% if involving the middle cerebral artery and as low as 42% if involving the anterior cerebral arterty.2 In the ED, CTA or MRA should be the initial diagnostic test of choice.
The classic radiographic findings of RCVS lag behind the clinical course. Other findings on imaging suggestive of RCVS include focal cortical SAH, intracerebral hemorrhage, and PRES-like bilateral posterior cerebral edema.1,2 In RCVS patients, intracerebral hemorrhage (ICH) is present in 1.5-10.1% of cases. 6-13 In contrast to the diffuse SAH associated with aneurysmal rupture, SAH in RCVS follows a characteristic pattern of focal cortical subarachnoid blood in high convexity sulci.2 Focal SAH is found in 15.7-34% of RCVS patients.9,22 This number should be interpreted with caution as many studies include populations of patients from headache clinics, emergency departments, and stroke services and thus focal SAH might not be present as commonly among RCVS patients upon their initial ED presentation. One observational study found that in patients with RCVS and concomitant hemorrhage, those without a focal neurologic deficit experienced a mean delay in diagnosis of four days compared to 1.1 days in patients with a deficit.29 This likely reflects the severity of disease leading to further evaluation and diagnosis.
Other radiographic findings that may be discovered in the course of a workup for TCH that should raise suspicion for RCVS include focal ischemia and cerebral edema.20 There are wide ranges for rates of associated ischemic stroke in RCVS patients from as low as 1.5% to as high as 39%, likely related to selection bias, as some studies recruited less severe cases from a headache clinic versus studies that more severe cases from an inpatient stroke service.7,22 Cerebral edema affecting the cerebral cortex of the occipital and parietal lobes, the classic radiographic findings of PRES, occur in anywhere between 9-38% of RCVS.1 The cerebral edema associated with RCVS is typically reversible within one month, usually resolving before the vasoconstriction.20
In patients with TCH a lumbar puncture (LP) might be performed as part of the work-up for aneurysmal SAH. In one study of patients with RCVS, 84% of patients had completely normal CSF studies. The most common abnormality was elevated protein levels, present in 16% of the patients, while 3% of patients had WBC counts>10. No patients had xanthochromia.22 Erythrocyte sedimentation rate (ESR) and C-reactive protein (CRP) are sometimes obtained in order to exclude rheumatologic causes, however, these are frequently normal in RCVS and can be nonspecific.22 Some experts in RCVS have advocated for testing for Lyme disease and measuring rheumatoid factor, antinuclear antibody, and urinary concentrations of vanillylmandelic acid and 5-hydroxy indoleacetic acid.20 These tests are unlikely to change emergency department management and should be left for inpatient or outpatient neurologist consideration.
One study has sought to develop a clinical score to aid in the diagnosis of RCVS (Table 4).30 The RCVS2 score was developed from a retrospective review of 110 patients with cerebral arteriopathy, 30 with RCVS and 80 with non-RCVS.30 This study then applied the score to a validation cohort and found a score of five or higher to have a sensitivity of 86% (95% CI 80-91) and a specificity of 94% (95% CI 82-99) for RCVS.30 They also found a score of two or lower to have a sensitivity of 77% (95% CI 62-88) and specificity of 96% (95% CI 92-99) for a non-RCVS diagnosis.30 This score should be used with caution given the wide confidence intervals of the sensitivity and specificity, and it has yet to be externally validated. It is important to note in order to calculate this score a patient needs a CTA to assess for carotid artery stenosis, which they found to be less likely associated with RCVS, compared to carotid artery dissection.30 Although potentially helpful in ruling in RCVS, it has limited utility in the emergency department to rule out RCVS.

Management
There are no RCTs to guide the management of RCVS. Calcium channel blockers such as nimodipine are frequently used to treat vasospasm.1,13,15,22 One study reported treating all 77 of RCVS patients with nimodipine at doses of 30-60mg every four hours, adjusted according to the severity of vasoconstriction on initial MRA.13 Nimodipine therapy has been associated with a 64-83% resolution of headache in RCVS, although there is no evidence that calcium channel blocker therapy reduces complications from RCVS.4 For patients who develop severe manifestations such as ischemic infarct or PRES, nimodipine can be infused at a rate of 0.5-2mg/hr titrated to blood pressure.4,13 One observational study reported the use of magnesium in 16% of cases, however there was no reporting on any outcomes from this treatment.7 Other vasodilators such as phosphodiesterase inhibitors have been used anecdotally.1 Some case reports also describe using intra-arterial injection of vasodilators.2 Vasodilators, including calcium channel blockers, must be used with caution because drops in systolic blood pressure may impair cerebral perfusion in patients with RCVS with severe cerebral vasoconstriction. Although no guidelines exist for how much to lower blood pressure, providers should be judicious in avoiding hypotension.1
As with the care used in the management of aneurysmal SAH, patients with RCVS associated hemorrhage should have their antithrombotics and anticoagulation reversed or withheld.1 There are no guidelines with regards to seizure prophylaxis in RCVS.
Any medications suspected as the underlying etiology or associated with RCVS should be withheld. As discussed previously, triptans should not be used to treat the headache of RCVS as they may potentially be a trigger.1,18 Steroids are associated with worse outcomes and should not be used.18,22
Disposition and Prognosis
Although most patients with RCVS have good neurologic outcomes and experience complete recovery, the rates of disability and death range from 5-13%.1,7 By definition, the segmental vasoconstriction underlying the disease should resolve within three months. One study looking at long-term outcomes of RCVS followed 173 patients for a mean of nine years and found a total of 10 recurrent cases (5.8%) of RCVS.31
RCVS patients with SAH, stroke, or seizure should be admitted to a neurology service. An undifferentiated patient with a thunderclap headache for whom aneurysmal SAH has been ruled out can be a challenging disposition and should be managed in accordance to patient preferences, local practices, and in consideration of availability of outpatient neurology versus in-hospital neurology consultation.
Conclusion
- Consider RCVS in patients with Thunderclap headache, particularly if they have had multiple episodes
- Physical exertion, triptans, and stimulants are common provoking causes of RCVS
- The diagnostic test of choice is CTA or MRA, which will classically show multiple segmental arterial narrowing; however, be aware that there can be a delay in imaging findings from symptom onset
- RCVS can be associated with focal ischemia, intracerebral hemorrhage, PRES, and cervical artery dissection
- Treatment includes calcium channel blockers such as nimodipine 30-60mg every 4 hours or a continuous infusion for severe cases. Avoid glucocorticoids, reverse anticoagulants if hemorrhage is present, and stop offending agents.
Case Conclusion:
A non-con head CT is ordered on the patient which showed a small focal SAH. CTA is obtained without identification of cerebral aneurysm. She is admitted to the neurology service and started on nimodipine. An MRI/MRA ordered on hospital day seven showed segmental narrowing in multiple cerebral vessels concerning for RCVS.
References:
- Miller TR, Shivashankar R, Mossa-Basha M, Gandhi D. Reversible Cerebral Vasoconstriction Syndrome, Part 1: Epidemiology, Pathogenesis, and Clinical Course. AJNR Am J Neuroradiol. 2015;36(8):1392-1399. doi:10.3174/ajnr.A4214
- Burton TM, Bushnell CD. Reversible Cerebral Vasoconstriction Syndrome. Stroke. 2019;50(8):2253-2258. doi:10.1161/STROKEAHA.119.024416
- Kim T, Ahn S, Sohn CH, Seo DW, Kim WY. Reversible cerebral vasoconstriction syndrome at the emergency department. Clin Exp Emerg Med. 2015;2(4):203-209. Published 2015 Dec 28. doi:10.15441/ceem.15.099
- Cappelen-Smith C, Calic Z, Cordato D. Reversible Cerebral Vasoconstriction Syndrome: Recognition and Treatment. Curr Treat Options Neurol. 2017;19(6):21. doi:10.1007/s11940-017-0460-7
- Pilato F, Distefano M, Calandrelli R. Posterior Reversible Encephalopathy Syndrome and Reversible Cerebral Vasoconstriction Syndrome: Clinical and Radiological Considerations. Front Neurol. 2020;11:34. Published 2020 Feb 14. doi:10.3389/fneur.2020.00034
- Lee MJ, Cha J, Choi HA, et al. Blood-brain barrier breakdown in reversible cerebral vasoconstriction syndrome: Implications for pathophysiology and diagnosis. Ann Neurol. 2017;81(3):454-466. doi:10.1002/ana.24891
- Katz BS, Fugate JE, Ameriso SF, et al. Clinical worsening in reversible cerebral vasoconstriction syndrome. JAMA Neurol. 2014;71(1):68-73. doi:10.1001/jamaneurol.2013.4639
- de Boysson H, Parienti JJ, Mawet J, et al. Primary angiitis of the CNS and reversible cerebral vasoconstriction syndrome: A comparative study. Neurology. 2018;91(16):e1468-e1478. doi:10.1212/WNL.0000000000006367
- Caria F, Zedde M, Gamba M, et al. The clinical spectrum of reversible cerebral vasoconstriction syndrome: The Italian Project on Stroke at Young Age (IPSYS). Cephalalgia. 2019;39(10):1267-1276. doi:10.1177/0333102419849013
- Singhal AB, Topcuoglu MA, Fok JW, et al. Reversible cerebral vasoconstriction syndromes and primary angiitis of the central nervous system: clinical, imaging, and angiographic comparison. Ann Neurol. 2016;79(6):882-894. doi:10.1002/ana.24652
- Choi HA, Lee MJ, Choi H, Chung CS. Characteristics and demographics of reversible cerebral vasoconstriction syndrome: A large prospective series of Korean patients. Cephalalgia. 2018;38(4):765-775. doi:10.1177/0333102417715223
- Chen SP, Chou KH, Fuh JL, et al. Dynamic Changes in White Matter Hyperintensities in Reversible Cerebral Vasoconstriction Syndrome. JAMA Neurol. 2018;75(9):1106-1113. doi:10.1001/jamaneurol.2018.1321
- Chen SP, Fuh JL, Wang SJ, et al. Magnetic resonance angiography in reversible cerebral vasoconstriction syndromes. Ann Neurol. 2010;67(5):648-656. doi:10.1002/ana.21951
- Mawet J, Boukobza M, Franc J, et al. Reversible cerebral vasoconstriction syndrome and cervical artery dissection in 20 patients. Neurology. 2013;81(9):821-824. doi:10.1212/WNL.0b013e3182a2cbe2
- Coffino SW, Fryer RH. Reversible Cerebral Vasoconstriction Syndrome in Pediatrics: A Case Series and Review. J Child Neurol. 2017;32(7):614-623. doi:10.1177/0883073817696817
- Grooters GS, Sluzewski M, Tijssen CC. How often is thunderclap headache caused by the reversible cerebral vasoconstriction syndrome? Headache. 2014;54(4):732-735. doi:10.1111/head.12256
- Wolff V, Lauer V, Rouyer O, et al. Cannabis use, ischemic stroke, and multifocal intracranial vasoconstriction: a prospective study in 48 consecutive young patients. Stroke. 2011;42(6):1778-1780. doi:10.1161/STROKEAHA
- Kunchok A, Castley HC, Aldous L, et al. Fatal reversible cerebral vasoconstriction syndrome. J Neurol Sci. 2018;385:146-150. doi:10.1016/j.jns.2017.12.009
- Cheng YC, Kuo KH, Lai TH. A common cause of sudden and thunderclap headaches: reversible cerebral vasoconstriction syndrome. J Headache Pain. 2014;15(1):13. Published 2014 Mar 1. doi:10.1186/1129-2377-15-13
- Ducros A. Reversible cerebral vasoconstriction syndrome. Lancet Neurol. 2012;11(10):906-917. doi:10.1016/S1474-4422(12)70135-7
- Ducros A, Wolff V. The Typical Thunderclap Headache of Reversible Cerebral Vasoconstriction Syndrome and its Various Triggers. Headache. 2016;56(4):657-673. doi:10.1111/head.12797
- Singhal AB, Hajj-Ali RA, Topcuoglu MA, et al. Reversible cerebral vasoconstriction syndromes: analysis of 139 cases. Arch Neurol. 2011;68(8):1005-1012. doi:10.1001/archneurol.2011.68:
- Long D, Koyfman A, Long B. The Thunderclap Headache: Approach and Management in the Emergency Department. J Emerg Med. 2019;56(6):633-641. doi:10.1016/j.jemermed.2019.01.026
- Bian L, Liu Y, Nichols LT, et al. Epidemiology of subarachnoid hemorrhage, patterns of management, and outcomes in china: A Hospital‐Based multicenter prospective study. CNS Neuroscience & Therapeutics. 2012;18(11):895-902. https://onlinelibrary.wiley.com/doi/abs/10.1111/cns.12001. doi: 10.1111/cns.12001.
- Richie MB, Josephson SA. A Practical Approach to Meningitis and Encephalitis. Semin Neurol. 2015;35(6):611-620. doi:10.1055/s-0035-1564686
- Scott RM, Smith ER. Moyamoya disease and moyamoya syndrome. N Engl J Med. 2009;360(12):1226-1237. doi:10.1056/NEJMra0804622
- Calic Z, Cappelen-Smith C, Zagami AS. Reversible cerebral vasoconstriction syndrome. Intern Med J. 2015;45(6):599-608. doi:10.1111/imj.12669
- Wolff V, Ducros A. Reversible Cerebral Vasoconstriction Syndrome Without Typical Thunderclap Headache. Headache. 2016;56(4):674-687. doi:10.1111/head.12794
- Ducros A, Fiedler U, Porcher R, Boukobza M, Stapf C, Bousser MG. Hemorrhagic manifestations of reversible cerebral vasoconstriction syndrome: frequency, features, and risk factors. Stroke. 2010;41(11):2505-2511. doi:10.1161/STROKEAHA.109.572313
- Rocha EA, Topcuoglu MA, Silva GS, Singhal AB. RCVS2 score and diagnostic approach for reversible cerebral vasoconstriction syndrome. Neurology. 2019;92(7):e639-e647. doi:10.1212/WNL.0000000000006917
- Boitet R, de Gaalon S, Duflos C, et al. Long-Term Outcomes After Reversible Cerebral Vasoconstriction Syndrome. Stroke. 2020;51(2):670-673. doi:10.1161/STROKEAHA.119.027703








