Authors: Kyle Delbar, MD (EM Resident Physician, Department of Emergency Medicine, Advocate Christ Medical Center, Chicago, IL) and Michael Cirone, MD (@mcironeMD, Assistant Program Director, Department of Emergency Medicine, Advocate Christ Medical Center, Chicago, IL) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case 1
A 71-year-old male with a past medical history of cutaneous T-cell lymphoma who underwent chemotherapy 1 week ago presents to the ED (Emergency Department) today with fever. His vital signs are T 38 C, HR 143, BP 108/64, RR 18, 95% on room air. On exam he is alert, oriented, and diaphoretic. After walking out of the room, you run into the nurse who is grabbing vacutainers and asks, “So, blood cultures…or no?”
Case 2
A 56-year-old male with a history of hypertension and hyperlipidemia presents to the ED with a chief complaint of “toe pain.” His toe pain is the result of a stubbed toe a few days ago that is now red, swollen, and quite painful. His vital signs are T 38 C, HR 100, BP 131/82, RR 16, 95% on room air. On exam, he is thin, generally well-appearing, and mentating appropriately. His left great toe is erythematous and swollen, with similar findings extending to his midfoot. There is a poorly healing 1×1 cm superficial abrasion without blisters, crepitus, or evidence of necrosis. His CBC from triage reveals a WBC of 14,000 cells/mm3. The nurse walks in the room while you’re evaluating the patient with blood culture tubes and says, “He has a fever. So, blood cultures…or no?”
To culture blood or not to culture blood? This question can plague Emergency Physicians (EPs) who regularly treat a variety of infections. Bacteremia is associated with a 37% mortality rate, but falsely positive cultures are associated with increased length of stay, cost, and countless harmful side effecs.1 During the early management of septic shock, every hour delay in pathogen-specific or empirically-specified antibiotic treatment is associated with a 7.6% reduction in survival.2
Developing your gestalt
Clinical practice requires the co-evolution of both clinical acumen and evidence-based medicine. When considering blood cultures, recognizing at-risk patient populations in which cultures are indicated is a great starting point. After all, a diagnosis not considered, is a diagnosis never made.
During residency, EPs develop a gestalt in identifying bacteremic or septic patients even before their labs are resulted. The classical cases are easy to identify. On physical exam these patients are flush with abnormal vital signs and evidence of end-organ dysfunction (ex. altered mental status, respiratory failure, etc). While fever alone is not always indicative of bacteremia,7 fever in combination with rigors, is one of the most predictive physical exam findings, with a likelihood ratio of 4.7.8 Patients with severe sepsis or septic shock bear a pre-test probability for bacteremia of 38%3and 69%,4 respectively. Both the Surviving Sepsis Campaign (SSC) and Infectious Disease Society of America (IDSA) encourage ED utilization of protocols to identify and begin empiric treatment of sepsis and septic shock.5 In recent years, the US Centers for Medicaid and Medicare Services (CMS) have created core measures tying blood cultures to hospital reimbursement.
In addition to the more obvious presentations of bacteremia, certain patient populations should also prompt EPs to more strongly consider blood stream infection. Febrile patients who are immunocompromised or neutropenic deserve blood cultures. Among patients with suspected infections, the strongest predictors for bacteremia are patients with suspected endocarditis, suspected venous line infection, or suspected urinary source.10 Though not as strongly predictive, thrombocytopenia, indwelling catheters, respiratory failure, and vasopressor requirement should also raise consideration of bacteremia.10
When Gestalt Isn’t Enough
Perhaps the most reliable tool to predict bacteremia is one generated and validated by Shapiro et al.10,12Their criteria has a sensitivity 0.97 and strong negative predictive value of 0.99. The tool includes three major criteria: suspected endocarditis, fever >= 39.4° C, and an indwelling catheter. If any of these criteria are present then blood cultures are indicated. There are also minor criteria, the presence of two of which indicate obtaining blood cultures. These minor criteria include: chills, vomiting, age >65, fever 38.3-39.3° C, WBC count >18,000/µL, creatinine >2 mg/dL, and systolic pressures <90 mm Hg.
In an effort to avoid the prolonged use of unnecessary antibiotics, Pawlowicz et al have created the following evidenced based algorithm to guide the decision to obtain blood cultures based on the Shapiro Rules.
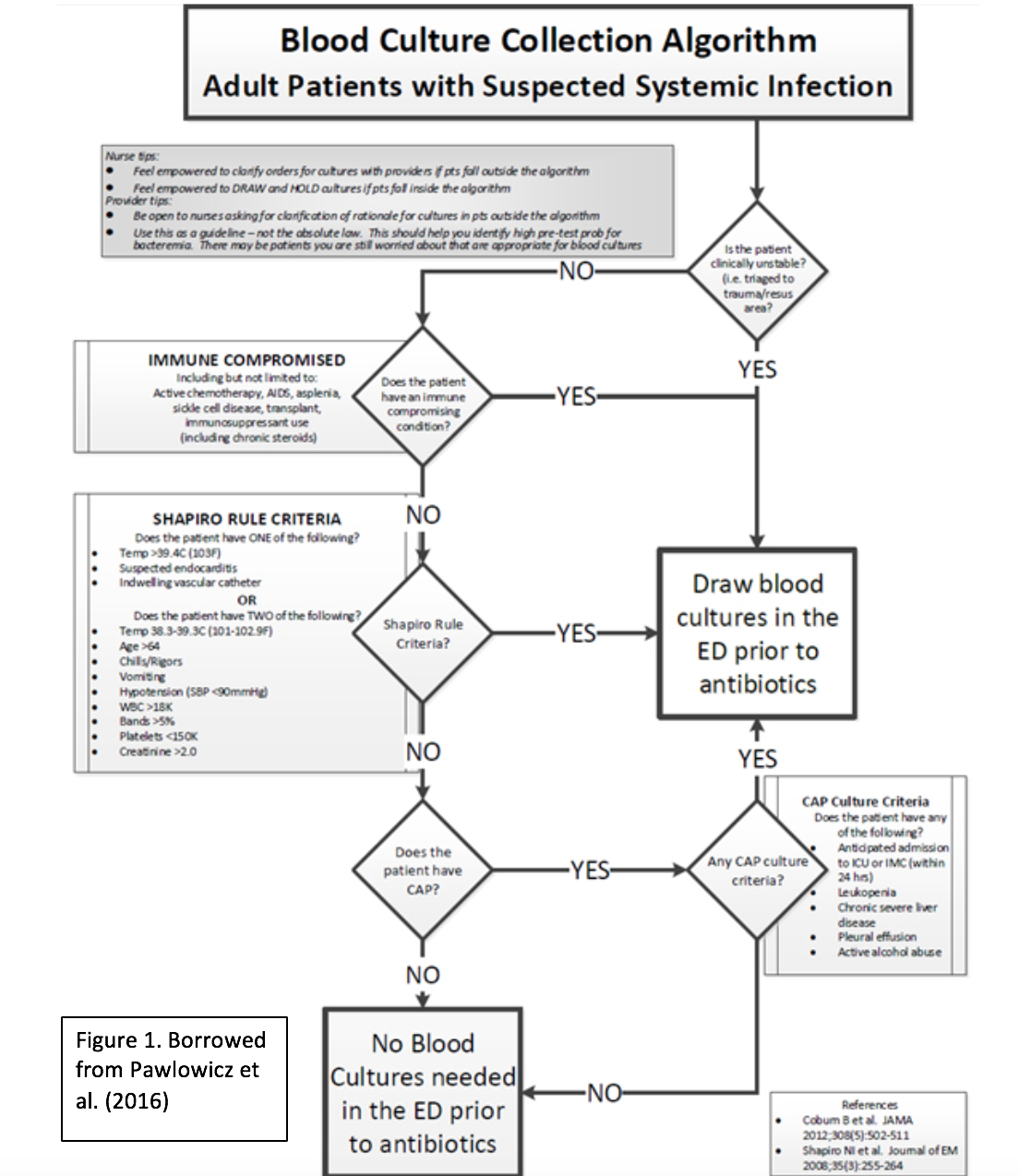
Translating to the Bedside
The following recommendations can be gleaned from the algorithms and guidelines discussed above:
- Patients who meet SIRS criteria and have unstable vital signs warrant blood cultures.
- Patients with suspected bacterial infections, whose clinical status appears sufficiently tenuous as to warrant an elevated level of care such as the intensive care unit or a stepdown unit warrant blood cultures, regardless of source.
- Patients with rigors and a suspected source of bacterial infection (ex. urinary tract infection, endocarditis, or pneumonia) warrant blood cultures.
- When EPs are concerned for an infection of indwelling catheters or lines, blood cultures should be obtained.
- Febrile neutropenic or immunocompromised patients warrant blood cultures.
- When feasible, blood cultures should be obtained prior to antibiotic administration.
Common Sources of Bacteremia
Bacteremia can originate from a variety of sources. Greater than 80% of blood stream infections are the result of urinary tract infections (UTI), followed by skin and soft tissue infections, lower respiratory infections, and biliary tract infections.14 In the ED, bacteremia secondary to lower respiratory infections has the highest mortality rate followed by intra-abdominal infections, primary bacteremia, and skin/soft tissue infections.14 Given that a simple cellulitis is very unlikely to cause bacteremia, these skin/soft tissue infections likely represent more complex infections such as necrotizing fasciitis.
The most commonly isolated pathogens from bacteremic patients in the ED include E. coli, Klebsiella spp, Staphylococcus spp., Streptococcus spp, and Pseudomonas spp. Together these five pathogens make up more than 80% of the culture specimens in the ED. These species correlate with the most prevalent sources of infection in the ED. In patients who are discharged from the ED and later found to have occult bacteremia, S. pneumoniae and Enterobacteriaceae are the most common isolates.14 Again, this correlates with the most frequent sources of infection in the ED- urinary tract infections, intra-abdominal, and respiratory sources.
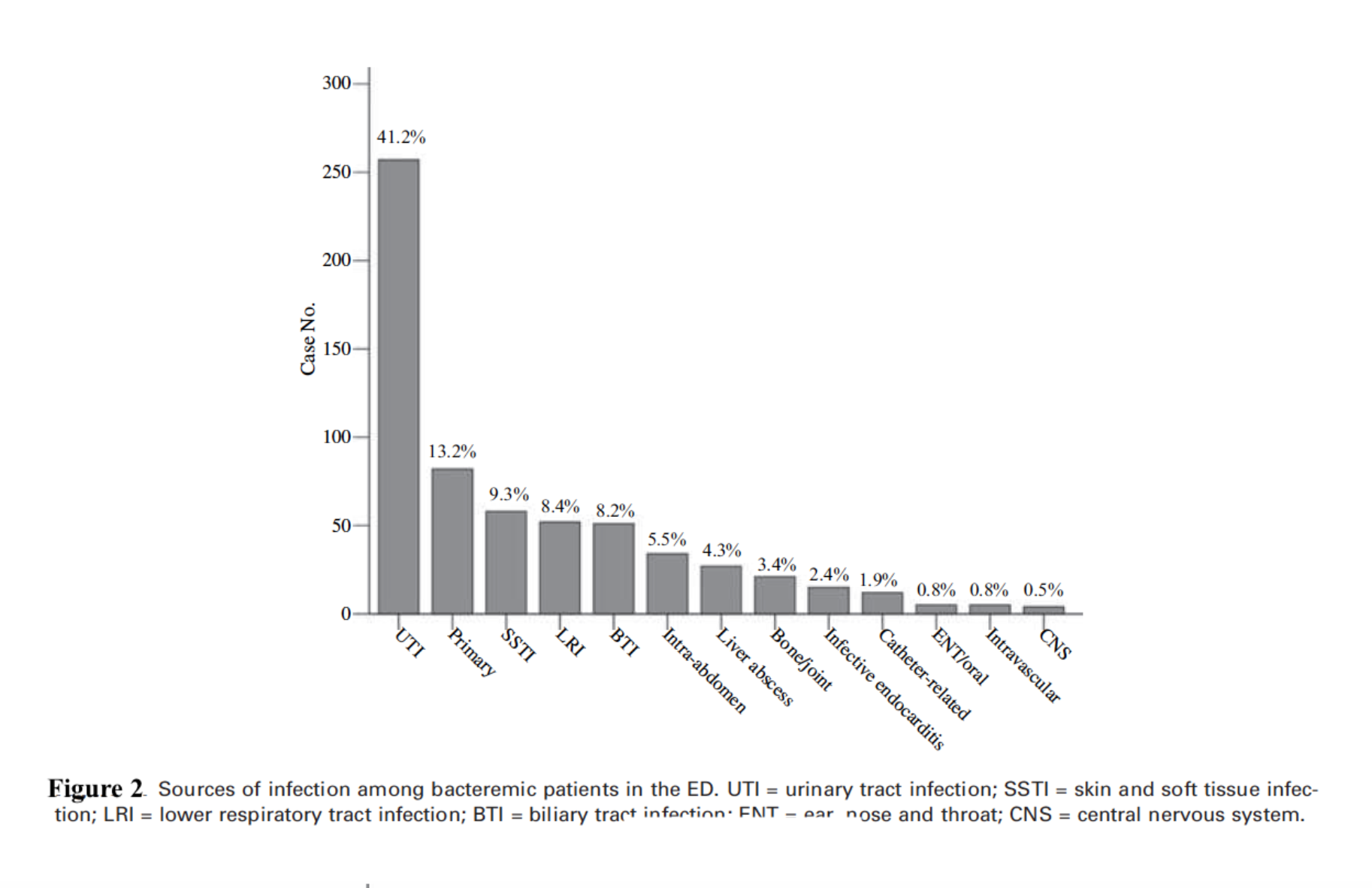

One study provided useful risk stratification of sources that placed patients at higher risk for bacteremia. Coburn et. al determined that patients with a diagnosis of community acquired pneumonia (CAP), simple cellulitis, and community acquired fever have a low pre-test probability for bacteremia.7 However, patients with pyelonephritis were considered moderate-risk with a pre-test probability for positive culture growth between 19-25%. Patients with bacterial meningitis were considered high-risk with a pre-test probability between 38%-68%.7
Now that bacteremia is suspected, how should it be managed?
Bacteremia in the ED should be managed much like any other infection in the ED: antibiotics and the age-old axiom “Ubi pus, ibi evacua,” also known as source control (“where [there is] pus, there evacuate [it]”).16,17 When infected, PICC lines and indwelling urinary catheters should be removed in the Emergency Department. Simple abscesses should be drained by EPs. Other sources of infection like orthopedic hardware, cardiac devices, and spinal stimulators should prompt specialist consultation for source control.
Appropriate antibiotic selection is important because source-specific therapy yields significant mortality benefit.2,18 In the ED, empiric antibiotics should be chosen based on the most likely source unless prior cultures are available for review. If a UTI or another classically gram-negative source is suspected, treatment should take place following the algorithm below.19 If there is concern for an intra-abdominal source, anaerobic coverage should be considered as well.

In the event that methicillin resistant staph aureus (MRSA) is a suspected source of bacteremia, a stat Echocardiogram should be ordered and patients should be started on Vancomycin (15 to 20 mg/kg/dose every 8 to 12 hours, not to exceed 2 g per dose).20
Pearls Before Swine
– Recognize that overall clinical impression trumps guidelines. Don’t let a suspicion of low-risk primary infection like cellulitis prevent you from ordering blood cultures in a frankly septic patient.
– Remember, when assessing a patient for bacteremia, one of the strongest physical exam predictors is rigors.
– If suspicion of bacteremia is low to moderate, evidence-based decision rules and algorithms (from Pawlowicz, Shapiro, etc) can be employed to help determine the need for cultures.10,12,13
– In addition to early antibiotics, the EP is responsible for performing source control or arranging for appropriate consultant intervention.
References / Further Reading
- Long B, Koyfman A. Best Clinical Practice: Blood Culture Utility in the Emergency Department. J Emerg Med. 2016 No;51(5):529-539. PubMed PMID: 27639424.
- Kumar A, Roberts D, Wood KE, Light B, Parrillo JE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med. 2006 Jun;34(6):1589-96. PubMed PMID: 16625125.
- Bates DW, Sands K, Miller E, Lanken PN, Hibberd PL, et al. Predicting bacteremia in patients with sepsis syndrome Academic Medical Center Consortium Sepsis Project Working Group. J Infect Dis. 1997 Dec;176(6):1538-51. PubMed PMID: 9395366.
- Rangel-Frausto MS, Pittet D, Costigan M, Hwang T, Davis CS, et al. The natural history of the systemic inflammatory response syndrome (SIRS) A prospective study. JAMA. 1995 Jan 11;273(2):117-23. PubMed PMID: 7799491.
- Howell MD, Davis AM. Management of Sepsis and Septic Shock. JAMA. 2017 Feb 28;317(8):847-848. PubMed PMID: 28114603.
- Centers for Medicare and Medicaid Services. Overview of specifications of measures displayed on hospital. Available at: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment Instruments/QualityMeasures/Core-Measures.html
- Coburn B, Morris AM, Tomlinson G, Detsky AS. Does this adult patient with suspected bacteremia require blood cultures? JAMA. 2012 Aug 1;308(5):502-11. PubMed PMID: 22851117
- Tokuda Y, Miyasato H, Stein GH, Kishaba T. The degree of chills for risk of bacteremia in acute febrile illness. Am J Med. 2005 Dec;118(12):1417. PubMed PMID: 16378800.
- Lee CC, Wu CJ, Chi CH, Lee NY, Chen PL, et al. Prediction of community-onset bacteremia among febrile adults visiting an emergency department: rigor matters. Diagn Microbiol Infect Dis. 2012 Jun;73(2):168-73. PubMed PMID: 22463870.
- Chase M, Klasco RS, Joyce NR, Donnino MW, Wolfe RE, et al. Predictors of bacteremia in emergency department patients with suspected infection. Am J Emerg Med. 2012 Nov;30(9):1691-7. PubMed PMID: 22626814.
- Pfitzenmeyer P, Decrey H, Auckenthaler R, Michel JP. Predicting bacteremia in older patients. J Am Geriatr Soc. 1995 Mar;43(3):230-5. PubMed PMID: 7884108.
- Jessen MK, Mackenhauer J, Hvass AM, Ellermann-Eriksen S, Skibsted S, et al. Prediction of bacteremia in the emergency department: an external validation of a clinical decision rule. Eur J Emerg Med. 2016 Feb;23(1):44-9. PubMed PMID: 25222426.
- Pawlowicz A, Holland C, Zou B, Payton T, Tyndall JA, et al. (2016). Implementation of an evidence- based algorithm reduces blood culture overuse in an adult emergency department. Gen Int Med ClinInnov1: doi:10.15761/GIMCI.1000108
- Lin JN, Tsai YS, Lai CH, Chen YH, Tsai SS, et al. Risk factors for mortality of bacteremic patients in the emergency department. Acad Emerg Med. 2009 Aug;16(8):749-55. PubMed PMID: 19594458.
- Epstein D, Raveh D, Schlesinger Y, Rudensky B, Gottehrer NP, et al. Adult patients with occult bacteremia discharged from the emergency department: epidemiological and clinical characteristics. Clin Infect Dis. 2001 Feb 15;32(4):559-65. PubMed PMID: 11181118.
- Eckmann C. The importance of source control in the management of severe skin and soft tissue infections. Curr Opin Infect Dis. 2016 Apr;29(2):139-44. PubMed PMID: 26779777.
- Tellor B, Skrupky LP, Symons W, High E, Micek ST, et al. Inadequate Source Control and Inappropriate Antibiotics are Key Determinants of Mortality in Patients with Intra-Abdominal Sepsis and Associated Bacteremia. Surg Infect (Larchmt). 2015 Dec;16(6):785-93. PubMed PMID: 26258265.
- Retamar P, Portillo MM, López-Prieto MD, Rodríguez-López F, de Cueto M, et al. Impact of inadequate empirical therapy on the mortality of patients with bloodstream infections: a propensity score-based analysis. Antimicrob Agents Chemother. 2012 Jan;56(1):472-8. PubMed PMID: 22005999; PubMed Central PMCID: PMC3256027.
- Moering R. Gram-negative bacillary bacteremia in adults. https://www.uptodate.com/contents/gram-negative-bacillary-bacteremia-in-adults
- Lowy F. Methicillin-resistant Staphylococcus aureus (MRSA) in adults: Treatment of bacteremia https://www.uptodate.com/contents/methicillin-resistant-staphylococcus-aureus-mrsa-in-adults-treatment-of-bacteremia









1 thought on ““So, Blood Cultures… or No?” – Use of Blood Cultures in the ED”
Nice review, especially the pearl to respect rigors!