Authors: William Laborde, MD, Brandon Mong, MD, and Joel Mosley, MD (Louisiana State University Health Sciences Center-New Orleans School of Medicine, Emergency Medicine Residency Program, Baton Rouge Campus, Baton Rouge, Louisiana) // Edited by Alex Koyfman, MD (@EMHighAK), Brit Long, MD (@long_brit), and Tim Montrief, MD (@EMinMiami).
Case #1
A patient comes into your ED one morning with the chief complaint of altered mental status. You check his vitals and find a temperature of 103.2F, heart rate of 119, respiratory rate of 25, and a blood pressure of 96/50. He is an 18-year-old college student that recently moved into his new dorm room. His roommate tells you that he has been acting differently for the past eight hours. After performing a lumbar puncture, you finally get a cerebrospinal fluid (CSF) sample, and send it to the lab. The CSF has a pink hue, and your initial results show 8,300 red blood cells (RBCs) and 112 white blood cells (WBCs) in your sample. You are now faced with a diagnostic dilemma. Does this patient actually have meningitis, or are the WBCs from the peripheral blood?
Case #2
You see a patient check in with the chief complaint of “headache”. She tells you that she is well accustomed to having bad headaches, but says that this one is different. The pain rapidly reached a 10/10 within seconds of onset. A non-contrast head computerized tomography (CT) scan is ordered. Seven hours and six minutes after the onset of the headache, the CT is performed. There is no evidence of subarachnoid hemorrhage (SAH) on the CT. Knowing that the sensitivity of the CT will decrease after the first six hours after onset; you make the decision to perform a lumbar puncture. No xanthochromia is present in the CSF. CSF analysis shows that there are 7,552 RBC/mm3 in tube #1 and 1,306 in tube #4. Does this patient need to be admitted for subarachnoid hemorrhage, or was this the unfortunate result of a “bloody tap”?
Background
“Bloody tap” and “traumatic tap” are both terms that have been used whenever accidental trauma is caused to a blood vessel during a lumbar puncture (LP). While they are often used interchangeably, previous authors have chosen to differentiate between these two terms. One such paper that looks at incidence in children with acute lymphoblastic leukemia (ALL) defines traumatic LP as one where the CSF sample has >10 RBCs/microL and a bloody LP as >500 RBCs/microL. These authors found that, of the 5,609 LPs performed, 29% were traumatic and 10% were bloody [1]. Other authors, however, tend to use both terms to refer to the same issue. For the scope of this article, we will not differentiate between the two terms. A separate study that looked at patients of all ages had similar numbers, with 15% having greater than 400 RBCs [2]. This number was chosen because this was found to be the visual threshold for bloody fluid. In the absence of any consensus on this topic, we will also use 400 RBCs/microL as the definition for bloody tap. Because this occurs so frequently, we must understand the impact of a bloody tap on the analysis of the obtained CSF.
Bloody taps most commonly occur when small capillaries or venules are inadvertently punctured. While this can affect the analysis of CSF in multiple ways, the increase in RBCs and WBCs are most relevant for this discussion. The most important disease processes in our practice that can be affected by this are meningitis and subarachnoid hemorrhage. In a bloody tap, both the numbers of RBCs and WBCs will be increased when measured in the CSF. Misinterpreting a bloody tap can have implications that lead to diagnostic uncertainty in the case of meningitis, as we rely on the number of WBCs in a CSF sample for diagnosis. When evaluating for meningitis, in the case of an equivocal tap, it is difficult to distinguish between WBCs in the CSF from WBCs that may simply be a contaminant from a bloody tap. A similar question arises when an LP is done to evaluate for subarachnoid hemorrhage (SAH). Are these RBCs in the collected sample due to an actual SAH, or is this just due to a bloody tap? We will investigate each of these questions individually and will discuss some of the literature surrounding each topic before providing specific recommendations for CSF interpretation.
Meningitis
CSF Basics
Bloody taps will cause both the number of CSF WBCs and CSF RBCs to increase. You may remember the charts from medical school that take not only CSF WBCs into account when assessing for meningitis, but also look at CSF protein, CSF glucose, opening pressure, and overall CSF appearance. Therefore, while it is not the only parameter that should be used in meningitis evaluation, CSF WBCs do have an integral role in the diagnosis of meningitis. Below is a chart that shows not only the normal CSF findings, but also the two pathological conditions that we are most interested in [3].
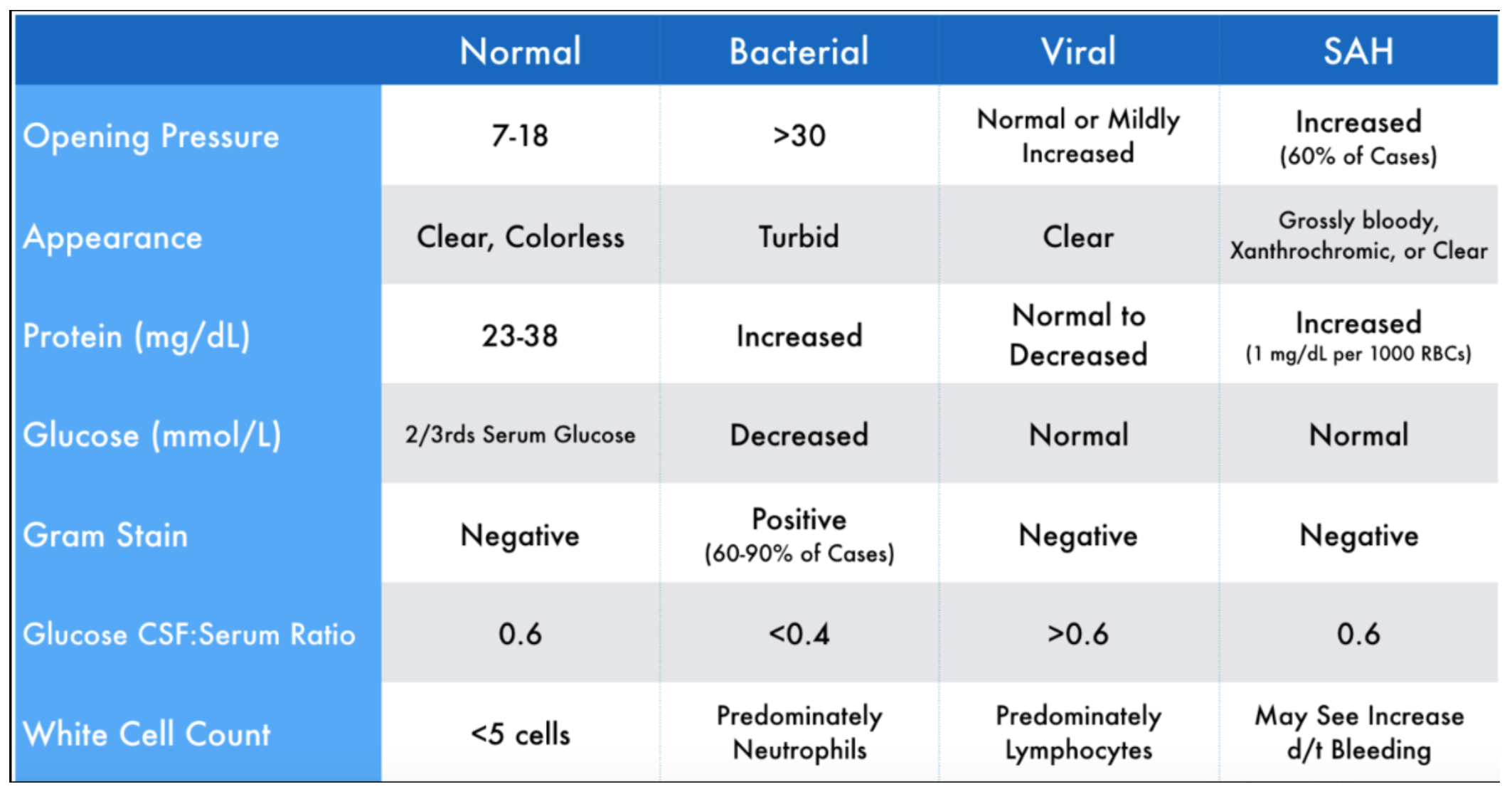
Many of us have heard of “correction formulas” that can allow us to compare the number of CSF WBCs that were actually visualized to the number of WBCs that would be expected due to blood contamination. Whenever we incidentally puncture a blood vessel in a bloody tap, the peripheral blood will mix with our CSF sample. Through analysis of the proportion of RBCs/WBCs in peripheral blood, we can in theory “assign” WBCs either as originating from the periphery or from the CSF.
CSF is typically acellular. Any more than 5 WBCs/microL or 3 polymorphonuclear leukocytes/microL is considered abnormal in adults. The data is not as clear in neonates, with some studies finding that an average CSF WBC count of 6-7 cell/mm3, while others found that up to 28 WBC/mm3 could be normal. For our purposes, we will use an upper limit of 20 WBC/mm3 in neonates [4]. Greater than 9 WBC/mm3is considered abnormal for ages 29-90 days [5] and greater than 6 WBC/mm3 is abnormal for children older than three months [6]. Additionally, any more than 5 RBCs can be considered abnormal, and should raise concern for the diagnosis of subarachnoid hemorrhage. One should never simply assume that a bloody tap was caused by trauma and use this assumption to justify the number of RBCs in your CSF. Blood doesn’t necessarily equal traumatic tap. When the pretest probability or index of suspicion for meningitis is high, there are other CSF findings that will steer you towards the diagnosis. Discussing the subtleties of CSF interpretation is outside the scope of this article, but it is important to remember that the analysis of WBCs in the CSF should not occur in a vacuum. There are many other considerations that become relevant, especially when the concern for traumatic tap muddies our diagnostic picture.
Correction Formulas
There are two main correction formulas that are employed when evaluating CSF in the context of a bloody tap. The first (and easiest) is a simple ratio. Remember, with a traumatic tap, you will see both increases in WBC and RBC in your CSF. Knowledge of normal ratios can (in theory) aid the clinician in distinguishing WBCs in the setting of suspected meningitis from simple peripheral contamination. Unfortunately, even in this “simple” ratio method, the ratio is not as simple as we promised. Some sources recommend using 1 WBC:500 RBC ratio, whereas others recommend 1 WBC:1,000 RBC [7]. Let us focus on this formula initially (and set the correction ratio at 1:1,000 to even further simplify the math). For each 1,000 RBC that are present in your CSF, you are allowed a single WBC. Therefore, according to this formula, if you have a sample with 25 WBCs and 13,400 RBCs, you are able to have up to 13 WBCs (when correcting for the number of RBCs in your specimen). You would simply subtract the predicted number of WBCs from the observed number (25 – 13 = 12). You are left with an elevated number of WBCs even after taking into account the traumatic tap [8].
The second correction formula takes into consideration the ratio of WBCs to RBCs noted in the bloodstream. The first step is calculating our expected number of CSF WBCs based off of what’s happening in the peripheral blood sample. The equation is (WBCs in blood * RBCs in CSF)/(RBCs in blood) [8]. It will become readily apparent if you are using different units improperly (e.g. comparing #/microL to #*10^6/microL). Therefore, make sure that your units match prior to utilizing this formula. Once you have obtained an expected number of WBCs in the CSF, you simply have to subtract this number from the measured number of CSF WBCs that you have obtained in your LP (observed CSF WBC – expected CSF WBC). Alternatively, an observed:predicted (O:P) ratio can be calculated by (observed CSF WBC) / (predicted CSF WBC) [8].

One study looked at neonatal 6,374 LPs at 150 different neonatal intensive care units (NICUs) over a period of seven years. The authors chose an upper limit of 20 WBC/mm3 [4]. They used multiple methods of correction factors to attempt to determine if they had a clinical use. The take-home message is that while the correction formulas increased the specificity, they simultaneously decreased the sensitivity. With uncorrected values, the sensitivity was 82% and the specificity was 57%. In this particular study, this resulted in a substantial number of patients that received treatment that didn’t have meningitis, with a relatively small number of patients with missed meningitis. Using both the 500:1 correction and the peripheral RBC:WBC correction both increased the specificity (to 75% and 82%, respectively), but the sensitivity fell (to 74% and 73%, respectively). In the study, for both correction factors, this would increase the number of false negatives to 5 from a baseline of 3 (without corrected values). Obviously, this is problematic on a “screening exam” for a disease with significant morbidity and mortality. The authors conclude, “The results of this study indicate that adjustment of WBC counts in the setting of a traumatic LP does not aid in the diagnosis of bacterial and fungal meningitis in neonates” [8]. It is worth noting, however, that this study was done on neonates ≤ 30 days of age, and therefore, the generalizability is called into question.
An additional study focuses on children ≤ 60–days-old. They found a ratio of CSF RBCs to CSF WBCs to be 877:1 amongst 2,880 infants. This fits relatively well with both the 500:1 and 1,000:1 correction factors that are commonly employed. They found that the 877:1 correction will increase the specificity from 21.8% to 67.4%, but decreases the sensitivity from 87.9% to 66.7%. This resulted in an additional 7 infants with bacterial meningitis being misclassified. However, there was an interesting subgroup analysis performed that looked at the infants from age 29-60 days. While this also showed a decrease in the sensitivity (100% to 85.7%), only 1 infant with positive cultures was misclassified. This infant grew Staphylococcus aureus from CSF, and this was determined to be a contaminant, leading to 0 infants with missed meningitis in the 29-60 day-old subgroup. The authors here explicitly reference the study that we previously discussed: “Similar to our study, correction for a traumatic LP using a 1000:1 correction substantially reduced the sensitivity of CSF WBC for bacterial meningitis. However, this study did not include infants in the second month of life where our correction formula performed with greater accuracy” [9].
Similar studies have reached conclusions that do not differ substantially. One study that looked at children age 1 month to 18 years used the areas under respective receiver operator characteristic curves to make their determination (AUC). They found that there was no difference between the AUC values of observed and predicted CSF leukocytes, lending no advantage to corrected CSF counts [7]. A separate study found that an observed number of WBCs 10 times greater than the predicted values from the correction formulas are both sensitive and specific for meningitis, but the authors caution against using the correction formulas as they currently stand [10].
During our search of the literature, it became apparent that there were more data on children than on adults. While it is impossible to know that any adult data would map onto that of the pediatric population, the trend seems unambiguous. Even the paper that paints the correction formulas in the most charitable light states, “Because adjustment of CSF WBC count for the presence of CSF RBCs reduces the corrected CSF WBC count, any approach to correction will reduce sensitivity and increase specificity for bacterial meningitis” [9]. Other than the single subgroup analysis that we detailed above, all of the literature we reviewed shows an increase in cases of missed meningitis when using correction formulas.
These correction factors have permeated through the discussion when it comes to bloody taps. We believe that it is important that the reader at least be familiar with their use. However, if we know that using corrected levels of WBCs reliably decreases the sensitivity of a meningitis diagnosis, we will eventually miss cases of meningitis if we employ these calculations. The Gram Stain and culture will eventually return and confirm or disconfirm the diagnosis. Prior to this, we believe that it is unwise to utilize a correction method with proven decreased sensitivity in a condition with this much morbidity and mortality.
Take-away Points
– While “correction formulas” may increase specificity, they decrease sensitivity.
– Due to the decrease in sensitivity, corrected levels of CSF WBCs should not be used to withhold therapy in the ED.
Subarachnoid hemorrhage
As we know, the mainstay for diagnosing subarachnoid hemorrhage will be non-contrast CT scan. Within the first 6 hours, CT will reliably identify SAH, and there will be no need to perform any additional diagnostic steps. But what happens when your suspicion of a SAH is high, and the CT shows no abnormalities? This all depends on the time from onset of symptoms. A 2011 study has shown that if the CT was obtained within 6 hours of symptom onset, the scan will have 100% sensitivity (as well as 100% specificity, 100% negative predictive value, and 100% positive predictive value) [11]. However, the sensitivity begins to fall after the 6-hour cutoff. If there is concern for SAH in the context of a negative CT, you should strongly consider an LP, especially when the presentation is delayed. After the 12-hour mark has elapsed, while sensitivity of CT remains generally strong, the risks of missing a deadly pathology become far too great to definitively exclude an SAH with a non-contrast CT. In this patient, an LP should be performed. Potential confusion arises when blood is found in the CSF fluid and a diagnostic challenge arises.
Xanthochromia
Xanthochromia (pictured on the left) [12] is the yellow or pink discoloration that can sometimes be seen in your CSF sample. This is due to the breakdown of hemoglobin into oxyhemoglobin and later into bilirubin. Oxyhemoglobin has a pink tint to it, whereas bilirubin gives the characteristic yellow tint; these degradation products often do not appear  until 12 hours after blood enters the CSF. Xanthochromia can either interpreted by the use of CSF analysis via spectrophotometry or by visual inspection of the CSF against a white background. However, <1% of hospitals in North America use spectrophotometry, and the vast majority simply use visual inspection [13]. While outside of the scope of this article, it is important to briefly note that it is unclear whether visual inspection or spectrophotometry is the superior assessment tool. While spectrophotometry had a higher sensitivity in a systematic review of the literature, the same review found that its specificity was significantly lower than that of visual assessment. The authors of this review conclude that it is “impossible to provide a definite conclusion about the diagnostic accuracy of spectrophotometry versus visual inspection” [14].
until 12 hours after blood enters the CSF. Xanthochromia can either interpreted by the use of CSF analysis via spectrophotometry or by visual inspection of the CSF against a white background. However, <1% of hospitals in North America use spectrophotometry, and the vast majority simply use visual inspection [13]. While outside of the scope of this article, it is important to briefly note that it is unclear whether visual inspection or spectrophotometry is the superior assessment tool. While spectrophotometry had a higher sensitivity in a systematic review of the literature, the same review found that its specificity was significantly lower than that of visual assessment. The authors of this review conclude that it is “impossible to provide a definite conclusion about the diagnostic accuracy of spectrophotometry versus visual inspection” [14].
Xanthochromia is highly suggestive of SAH, but can be found in rare cases with severe systemic hyperbilirubinemia, traumatic tap, or in the context of highly elevated CSF protein [15]. Owing to several older studies, such as one performed by Vermeulen et al. in 1989, it has long been the belief that a normal CT scan and negative xanthochromia (provided that the LP was done between 12 hours and 2 weeks) was sufficient to rule out SAH with a sensitivity of 100% (as all 111 patients with CT confirmed SAH displayed xanthochromia)[16]. In this study, spectrophotometry was the sole method used for evaluation of CSF. However, a more recent systematic review in 2014 by Chu K., et al. analyzes a related, but distinct, question. Because this is a systematic review, there is heterogeneity in the times from symptom onset to LP. Not every study included in this analysis waited 12 hours after symptom onset to perform the LP. As we would expect, this decreases the overall sensitivity. In this study, pooled sensitivity and specificity of spectrophotometry was 86.5% and 85.8%, respectively. Visual inspection for xanthochromia was also imperfect, with a sensitivity and specificity of 83.3% and 95.7%, respectively [14]. At this time, further research likely needs to be conducted before relying exclusively on the presence or absence of xanthochromia. While a sensitivity of 83.3% is high, this provides an unacceptably high miss rate of a deadly disease.
While we do not necessarily recommend that we ensure that 12 hours elapse prior to our LP, we do strongly recommend that the physician understand the limitations of the testing that is employed. If a sample does not have xanthochromia, but was collected <12 hours after symptom onset, the lack of xanthochromia cannot be used to definitively exclude SAH. However, it can be used in conjunction with certain other findings (physical exam, history, CT findings, RBC absolute count, and RBC clearing) to bolster an already strong argument for the likelihood of a bloody tap.
Clearing of RBCs
In addition to xanthochromia, analysis of the number of RBCs in separate tubes can provide useful information. However, (similar to xanthochromia) there is no universally accepted method of evaluating RBC clearance to establish the likelihood of traumatic tap vs. SAH. On the surface, a commonly held belief has held that if there are fewer RBCs in tube 4 than tube 1, the likelihood of a traumatic tap is high, and the probability of SAH is lower. Theoretically, in SAH, the RBC count will remain relatively constant from tube 1 to tube 4. Multiple studies have evaluated and attempted to quantify this methodology; however, most have been limited in size and scale. There does not appear to be consensus in the literature regarding either RBC clearing or an absolute number of RBCs that rules in or out the diagnosis. However, we will provide a discussion of the relevant recent literature. It is incumbent upon the physician (reader) to understand the limitations and make ultimate treatment decisions in coordination with subspecialists and shared decision making with patients.
Some early studies suggested that LPs could likely be considered traumatic if there was RBC clearance of >30% [17]. A more recent study published in 2007 by Gorchynski et al. attempted to help clarify this commonly held theory. In this study, the authors found that a count in tube #4 of <500 RBC/mm3 had a negative predictive value of 100% for SAH in this study. This number was independent of RBC clearing. The ultimate conclusion of this study is that a CSF RBC count of less than 500, coupled with an RBC decrease from tube 1 to 4 of at least 70%, may exclude patients with a radiographically detectable SAH [18]. While potentially practice changing, the authors of this study do note that this study was limited by sample size, as there were only 15 total people diagnosed with SAH. This data was referenced in an article published in Annals of Emergency Medicine in 2016, by Chin and Sarko entitled “Tried and True and Still the Best”. The authors of this article reiterated these above findings, suggesting (as hinted in the title) that a CT then LP approach is superior to a CT then CTA approach [19]. While there may be a role for CTA, that is beyond this discussion of RBC clearing.
In 2015, a prospective cohort study by Perry JJ, et al. attempted to provide what could be the most recent evidence yet. The authors of this study enrolled 1,739 patients, of which 36.5% had abnormal results upon analysis of CSF fluid in the final tube. Ultimately, 15 patients were diagnosed with aneurysmal subarachnoid hemorrhage. Of the patients diagnosed, the median number of RBCs in the CSF was 20,000, compared to patients with a traumatic tap that had a median of 20 RBCs. The authors concluded that no xanthochromia (in any tube) and less than 2,000 red blood cells (in the final tube) is a low-risk finding. [20] While the recommendations set forth by Perry are backed by a study that appears to be methodologically sound, it is less conservative than the recommendations of Gorchynski, Until more data comes out to further clarify this topic, we will choose the conservative approach of Gorchynski out of an abundance of caution. We will additionally add that this conclusion must be interpreted integrating the whole context of the patient’s presentation (history, physical exam, time of onset, etc.) and the pretest probability of SAH.
Take-away Points
– A non-contrast head CT has 100% sensitivity within the first 6 hours; after this time window, a CT by itself cannot be used to exclude SAH.
– Xanthochromia doesn’t reliably appear in the CSF until 12 hours after symptom onset. If your LP is performed prior to this, the absence of xanthochromia cannot be used to exclude SAH.
– While there is no formal consensus on absolute RBC count or RBC clearing to definitively exclude SAH, on our review of the literature, we find that an absolute count of less than 500 in the final tube coupled with a clearing of >70% is reassuring. While this is conservative, this is the cutoff that we recommend.
– In situations where the diagnosis is unclear, you should consult neurosurgery.
Back to the Vignettes
If we go back to the first vignette, the question was “should we treat this patient as if he has meningitis”. The answer is clear: yes. In the ED, we should give him broad-spectrum antibiotics to cover him for bacterial meningitis. If you recall, he had 8,300 RBCs and 112 WBCs in his CSF sample. With a high suspicion, the conversion formulas will only decrease the sensitivity of your testing and make it more likely that you will miss a case of meningitis. Therefore, we honestly should not be employing these calculations in the ED in the absence of culture data, and they should be left for the inpatient team once more information has been gathered.
The second vignette asks whether the RBCs found in the CSF are due to a SAH or a traumatic tap. There is no xanthochromia noted. The number decreases from 7,552 to 1,306 from tubes #1 to #4. This is an 83% decrease. According to one of the studies quoted above [18], an absolute count of 500 RBCs in tube #4 was safely assumed to be due to a traumatic tap. However, this example clearly does not meet that standard. On the other hand, the RBCs did clear >70%. According to Gorchynski’s analysis, both conditions (absolute count of <500 and >70% decrease) must be satisfied in order to rule out SAH. If we were to instead consider this in the context of Perry’s recommendations, we can take this to be a low risk finding. We argue that rather than clinging to the suggestions set forth by Perry, we recommend shared decision making and neurosurgical consultation in situations where the answer is unclear. While is it likely safe to assume that these RBCs are due to a bloody tap, both the heterogeneity of the recommendations set forth in the literature and the extreme morbidity/mortality of the underlying disease process causes us to approach the situation with significant caution.
References/Further Reading
[1] Howard SC, Gajjar AJ, Cheng C, et al. Risk factors for traumatic and bloody lumbar puncture in children with acute lymphoblastic leukemia. Jama. 2002;288(16):2001–7.
[2] Shah KH, Richard KM, Nicholas S, Edlow JA. Incidence of traumatic lumbar puncture. Acad Emerg Med. 2003 Feb; 10(2):151-4.
[3] Hill, J. (2017, April 19). Taming the SRU. What’s in a Tap: CSF Analysis. Retrieved from http://www.tamingthesru.com/blog/diagnostics/whats-in-a-tap-csf-analysis
[4] Gunn VL, Nechyba C Johns Hopkins Hospital. Children’s Medical and Surgical Center. The Harriet Lane handbook: a manual for pediatric house officers. 16. xiv. Philadelphia: Mosby; 2002. p. 1036.
[5] Kim KS. Bacterial meningitis beyond the neonatal period. In: Feigin and Cherry’s Textbook of Pediatric Infectious Diseases, 7th, Cherry JD, Harrison GJ, Kaplan SL, et al (Eds), Elsevier Saunders, Philadelphia 2014. p.425.
[6] Kestenbaum LA, Ebberson J, Zorc JJ, et al. Defining cerebrospinal fluid white blood cell count reference values in neonates and young infants. Pediatrics 2010; 125:257.
[7] Bonsu BK, Harper MB. Corrections for leukocytes and percent of neutrophils do not match observations in blood-contaminated cerebrospinal fluid and have no value over uncorrected cells for diagnosis. Pediatr Infect Dis J. 2006;25(1):8–11.
[8] Greenberg RG, et al. Traumatic Lumbar Puncture in Neonates. Pediatr Infect Dis J. 2008; 27(12): 1047-51.
[9] Lyons TW, Cruz AT, Freedman SB, et al. Interpretation of Cerebrospinal Fluid White Blood Cell Counts in Young Infants With a Traumatic Lumbar Puncture. Ann Emerg Med. 2016;69(5):622-631.
[10] Mayefsky JH, Roghmann KJ. Determination of leukocytosis in traumatic spinal tap specimens. Am J Med. 1987 Jun;82(6):1175-81.
[11] Perry JJ, et al. Sensitivity of computed tomography performed within six hours of onset of headache for diagnosis of subarachnoid haemorrhage: prospective cohort study. BMJ. 2011; 343:d4277.
[12] Williams, A. Xanthochromia in the cerebrospinal fluid. Practical Neurology, 2004;4:174-175.
[13] Edlow J, Bruner K, Horowitz G. Xanthochromia. Arch Pathol Lab Med. 2002;126(4):413-5.
[14] Chu K et al. Spectrophotometry or visual inspection to most reliably detect xanthochromia in subarachnoid hemorrhage: systematic review. Ann Emerg Med. 2014;64(3)256-264.
[15] O’Glasser A, Mansoor, A. “If You Prick Us, Do We Not Bleed?”: An Uncommon Cause of Xanthochromia. J Gen Intern Med. 2015;30(6)853-854.
[16] Vermeulen M et al. Xanthochromia after subarachnoid haemorrhage needs no revisitation. J Neurol Neurosurg Psychiatry. 1989;52(7):826-8.
[17] Rinkel GJ, van Gijn J, Wijdicks EF. Subarachnoid hemorrhage without detectable aneurysm. A review of the causes. Stroke. 1993;24:1403–9.
[18] Gorchynski J, Oman J, Newton T. Interpretation of traumatic lumbar punctures in the setting of possible subarachnoid hemorrhage: who can be safely discharged? Cal J Emerg Med. 2007;8(1):3-7.
[19] Chin, N et al. Tried and True and Still the Best: Lumbar Puncture, Not Computed Tomography Angiogram, for the Diagnosis of Subarachnoid Hemorrhage. Ann Emerg Med. 2016;67(6):774-775.
[20] Perry JJ, et al. Differentiation between traumatic tap and aneurysmal subarachnoid hemorrhage: prospective cohort study. BMJ. 2015 Feb 18;350:h568







1 thought on “The Bloody CSF Tap – Pearls and Pitfalls”
Pingback: An Ache in the Head - Chief Medical Resident Blog