Author: Erica M. Simon, DO, MHA (EM Resident Physician, SAUSHEC) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW Medical Center / Parkland Memorial Hospital) & Justin Bright, MD (@JBright2021)
Case Presentation
A 34-year-old female, without significant past medical history, presents with the acute onset of chest pain. The patient endorses intermittent palpitations of two days duration. ROS is remarkable for six months of progressively worsening dyspnea on exertion.
HR 175, BP 82/Palp (Manual), T 99.8, RR 21, SpO2 88% RA
Two large bore IVs are placed and the patient is started on supplemental oxygen. Physical exam reveals an increased pulmonary component of the second heart sound, JVD, and +3 pitting edema of the bilateral lower extremities. Initial EKG reveals an irregularly irregular rhythm.
Your patient is crashing – she is in Afib w RVR, she is hypotensive and hypoxic with signs of right-sided heart failure.
As your crash cart wheels into the room, you proceed to cardiovert. Repeat EKG reveals NSR with a rate of 92 bpm, RAD, RVH, and signs of right heart strain.
BP 101/74, RR 16, SpO2 90% on 4L NC
Bedside echo reveals significant RVH, a shift of the septum towards the LV during diastole, and an IVC collapse >50% with respirations. The lung fields are without Kerley B-lines.
What is going on? What do you do? Fluids? Inotropes?
Background
Pulmonary hypertension (PH) is as an elevated pulmonary arterial pressure (≥ 25mmHg) as diagnosed by right heart cath. The prevalence of PH in the US is between 15-26 million persons. 1 The majority of patients with PH present with dyspnea, fatigue, palpitations, lightheadedness, chest pain, near-syncope, and syncope. Given these non-specific complaints, 20% of patients go undiagnosed for more than 2 years.2
The World Health Organization classifies PH according to etiology, pathophysiology, and treatment:
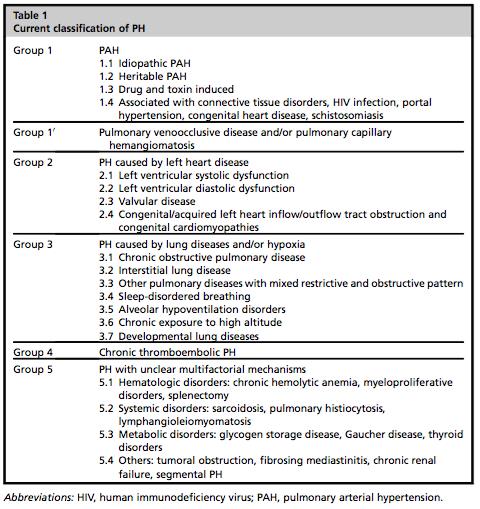
Group 1
- Idiopathic PH affects women much more commonly than men (2:1), with the mean age of onset at 37 years.
- Heritable PH is often secondary to mutations in bone morphogenic protein receptors.
- Drug-and toxin-induced PH has been associated with weight loss medications (fenfluramine, dexfenfluramine) and amphetamines.3
- PH is known to occur in the setting of connective tissue diseases such as scleroderma, and is a rare side effect of HIV.4
- A number of individuals with portal hypertension develop PH, although the physiologic reason is not clear.3
- Individuals suffering from untreated, congenital systemic-to-pulmonary shunts often experience PH.
Group 2
This group by far represents the most common etiology of PH. In these individuals, left-sided ventricular or valvular disease increases left atrial pressure, which results in increased pulmonary vascular resistance (PVR).
Group 3
Group 3 patients experience PH secondary to lung diseases or chronic hypoxia. As a general rule, the more advanced the parenchymal lung disease and the hypoxia, the more symptomatic the PH.
Group 4
Individuals in Group 4 suffer from thromboembolic diseases. Up to 4% of patients experience PH after an acute PE.5 As you might expect, treatment consists of systemic anticoagulation.
Group 5
Finally, Group 5 includes a hodgepodge of diseases/pathological processes linked to PH through unexplained mechanisms.
Pathophysiology
End Game: All roads lead to ventricular dysfunction.
The result of continuous pulmonary arterial hypertension is right ventricular hypertrophy (RVH). Over time, RVH transitions to RV dilation to maintain preload and stroke volume. Many patients experience tricuspid regurgitation as deficits in leaflet coaptation ensue. As the RV plateaus on its Starling curve, contractile force progressively weakens; evidenced by outward manifestations of right heart failure. Further complicating cardiac function, the previously mentioned RV dilatation impairs LV ventricular filling, subsequently decreasing cardiac output.
Individuals with long-standing PH are also predisposed to ischemia and dysrhythmias:
Perfusion of the right coronary artery (RCA) is significantly impaired in patients with advanced PH. Under normal circumstances, the RCA is perfused during both systole and diastole. As the RV remodels secondary to chronic PH, elevation in trasmural pressure impairs RCA perfusion to the point that flow occurs almost exclusively during diastole. This perfusion failure often leads to ischemia and increased risk of arrhythmias.17 In this setting, tachyarrhythmias can rapidly precipitate RV failure as the heart becomes increasingly dependent upon the atrial kick to maintain cardiac output. 6
Evaluation
ED evaluation of suspected PH should include:
EKG
Findings commonly associated with PH include RAD, RVH, RBBB, rsR’ in V1, qR in V1, large inferior p waves, and ST depression or T-wave inversions in V1 or the inferior leads (right heart strain).
CXR
AP findings include enlarged hilar pulmonary arterial shadow, pulmonary venous congestion, prominent right heart border, and right atrial enlargement. A common radiologic finding is vascular pruning, or enlarged pulmonary arterial shadows that have an early taper toward the periphery.17
Echo
Bedside echo is the ED physician’s best tool in the evaluation of PH. Assessment of the RV is the single most important component of the exam. Patients with PH commonly exhibit RA and RV dilation, RV free wall thickening, and an RV: LV ratio >1 (normal <0.6) on apical four-chamber view. RV pressure overload is often signified by the “D Sign,” or a paradoxical septal shift toward the LV during early diastole.6,17
(Note: These findings differ from PH secondary to an acute PE in which the RV is dilated, and the RV free wall thin.)

Troponin
As we mentioned earlier, individuals with PH are pre-disposed to myocardial ischemia secondary to poor RCA perfusion. An elevated Troponin T has been identified as an independent predictor of mortality in patients with PH. 9-10
BNP
Although there is little evidence correlating BNP levels with adverse outcomes, it is likely that this value reflects the degree of myocardial stretch encountered by the after-loaded RV.10 As you would expect, this marker is of increasing utility as compared to a baseline value – often unavailable in the previously undiagnosed pulmonary hypertensive.
CTPA
CTPA can be helpful when evaluating previously undiagnosed PH as it may demonstrate structural and vascular abnormalities of the heart and lungs. Of note, Tan et al. demonstrated a 96% PPV for PH in individuals in which the maximum transverse diameter of the pulmonary artery was greater than the diameter of the proximal ascending aorta.11,17
Management
No current consensus guidelines exist for the management of PH, thus management goals are based on what we know of the pathophysiology of PH:
Reverse Underlying Causes
Although not applicable in all cases, this has the potential to be addressed in:
Group I – Drug and Toxin-induced PH and Congenital Defects (shunts)
Group II – LV dysfunction
Group III – COPD
Group IV – Chronic thromboembolic disease
Group V – Anemia, Thyroid disorders
The list goes on…
Optimize RV Preload
Adequate, NOT excessive preload should be your mantra. Patients with PH can augment their cardiac output with IVF, but caution is key as excessive intravascular volume ultimately leads to increased cardiac septal bowing and worsening LV diastolic dysfunction (decreased cardiac output).
USE YOUR BEDSIDE ECHO FOR IVC ASSESSMENT. RV failure and hypotension are MORE COMMONLY the result of increased RV afterload and hypervolemia.2,17
If the patient is stable and presents with volume overload, diuretics can be considered (furosemide and bumetanide).12 Diuretic dosing should be titrated to the patient’s hemodynamic response.17
If the patient appears intravascularly volume depleted – START LOW AND GO SLOW. Repeated, small volume, crystalloid boluses are the way to go. DO NOT start continuous fluids; you WILL cause cardiac compromise.
Support RV Systolic function
Individuals with undiagnosed PH often present with obstructive or cardiogenic shock. Inotropic therapy is recommended in the case of poor peripheral perfusion (elevated lactate, AKI, etc.) or hypervolemia unresponsive to controlled diuresis.
Dobutamine is FIRST LINE.12-13 Dobutamine is a selective β1-agonist that increases cardiac contractility and reduces pulmonary vascular and systemic vascular resistances.17 A dobutamine drip should be started at 2mcg/kg/min-10mcg/kg/min. Doses >10mcg/kg/min predispose to reflex tachycardia and are associated with increased mortality.13 It is important to be aware that dobutamine can cause hypotension secondary to β2-mediated systemic vasodilation. If the patient is persistently hypotensive or becomes hypotensive in the setting of dobutamine therapy, start NE. Why NE? We’ll get to that below.
Milrinone is SECOND LINE. Milrinone has been shown to reduce pulmonary pressures and improve RV function in the setting of LV failure.14 Milrinone is a PDE-3 inhibitor that increases cardiac contractility by increasing cAMP, therefore increasing intracellular calcium. A milrinone drip can be started at 0.375 mcg/kg/min and titrated to 0.75 mcg/kg/min. Similar to dobutamine, milrinone also results in hypotension at higher doses. Again, if the patient is hypotensive despite milrinone therapy, or becomes hypotensive in conjunction with the administrative of milrinone – start NE.
Support RCA Perfusion
In order to adequately perfuse the RCA, mean arterial BP must be > pulmonary arterial pressure.
AVOIDING HYPOTENSION IS A MUST.
IF PVR exceeds SVR, coronary blood flow suffers and the patient is pre-disposed to ischemia.
Norepi is the PRESSOR OF CHOICE. The α1 and α2 agonist properties of NE result in increased SVR, thus augmenting RV function and CO. As compared to dopamine, Kerbaul et al. demonstrated that the utilization of NE in PH reduced 28-day mortality in individuals suffering from cardiogenic shock. Individuals receiving NE therapy also experienced decreased rates of tachyarrhythmias when NE was employed.13 A NE drip should be initiated at 0.05 mcg/kg/min. Although titration is often necessary, be wary of high doses which may result in pulmonary vasoconstriction (α1 activity).12
Avoid Phenylephrine. PE is a strong α1 agonist which constricts arterioles => increased pulmonary arterial pressure and PVR leading to worsening RV systolic function.15
Reduce RV Afterload
Excessive pulmonary arterial pressure = increased RV afterload.
To reduce RV afterload avoid hypoxemia and hypercapnia which increase PVR => use supplemental O2 to titrate SpO2 to >90%.2,17
What about BiPAP or CPAP? These may be useful in patients presenting with known Group 2 PH (secondary to LV failure) or Group 3 PH (secondary to underlying pulmonary disease).
AVOID NON-INVASIVE POSITIVE PRESSURE VENTILATION IN THE SETTING OF HYPOTENSION.
Increased intrathoracic pressure secondary to positive pressure ventilation will only worsen RV preload.
What about intubation and mechanical ventilation? Hopefully your resuscitation does not go this way. As above, PPV in a patient with PH will increase PVR and reduce SVR => decreased coronary perfusion and ischemia, and decreased RV preload and subsequent CO. IF you need to intubate:
1) Use etomidate (has minimal effect on SVR, PVR and cardiac contractility).
2) Use lung-protective ventilator settings to avoid pulmonary injury resulting in increased PVR.
3) Avoid permissive hypercapnea which increases pulmonary arterial pressure.2
Dysrhythmias
Atrial fibrillation and atrial flutter are the most common tachydysrhythmias seen in patients suffering from PH.17 This is a DANGEROUS situation. If left untreated, tachydysrhythmias will precipitate acute decompensation.16 (As discussed earlier, PH patients are dependent upon the atrial kick to maintain CO.) Aggressively manage unstable patients with electrical cardioversion. Use EXTREME caution with B-blockers and CCB, which impair cardiac contractility and can lead to cardiogenic shock.17
Take-Home Messages
- Patients with PH quickly decompensate – worry about obstructive and cardiogenic shock.
- Bedside echo is your #1 tool in undiagnosed PH – Assess the RV, is there hypertrophy? What is the RV:LV ratio? Is it >1? Is there septal bowing? What does the IVC tell you about the patient’s intravascular volume status?
- Get a troponin – patients with severe PH often suffer from coronary ischemia secondary to RCA perfusion deficits.
- Your goals as an EM physician should be to:
- Reverse Underlying Causes When Possible
- Optimize RV Preload
- Use your knowledge gained from the bedside echo to help you. REMEMBER: RV failure and hypotension are MORE COMMONLY the result of increased RV afterload and hypervolemia.
- Intravascular hypovolemia: 250-500cc crystalloid boluses
- Intravascular hypervolemia: controlled diuresis
- Support Systolic Function
- Dobutamine is FIRST LINE: 2mcg/kg/min-10mcg/kg/min
- Milrinone is SECOND LINE: 0.375mcg/kg/min-0.75mcg/kg/min
- HYPOTENSION = Norepi
- Maintain RCA Perfusion
- Norepi: Start at 0.05mcg/kg/min but caution with high doses = worsens pulmonary vasoconstriction
- No Phenylephrine: worsens RV systolic function
- Reduce RV Afterload
- SpO2 > 90%
- AVOID PPV in hypotensive patients
- Try to avoid intubation and mechanical ventilation at all costs, but if you need to intubate: etomidate, lung-protective ventilator settings, and avoid permissive hypercapnea
- Dysrhythmias
- Rhythm control is much more important than rate control – PH patients are dependent upon the atrial kick to maintain CO
- Cardiovert when indicated
References / Further Reading
- George M, Schieb L, Ayala C, Talwalkar A, Levant S. Pulmonary hypertension surveillance 2001 to 2010. Chest 2014; 146 (2): 467-495.
- Hoeper M, Ganton J. Intensive care unit management of patients with severe pulmonary hypertension and right heart failure. Am J Resp Crit Care 2011; 184 (10): 1114-1124.
- Farber H, Loscalso J. Pulmonary arterial hypertension. NEJM 2004; 351 (16): 1655-1665.
- Simmonneau G, Robbins I, Beghetti M, et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 2009;54:S43–54.
- Matthews J, McLaughlin V. Acute right ventricular failure in the setting of acute pulmonary embolism of chronic pulmonary hypertension: a detailed review of the pathophysiology, diagnosis, and management. Curr Cardiol Rev 2008; 4(1): 49-59.
- Voelkel N, Quaife R, Leinwand L, et al. Right ventricular function and failure. Circulation 2006; 114: 1883-1891.
- Rich S, Dantzker D, Ayres S, et al. Primary pulmonary hypertension: a national prospective study. Ann Intern Med 198;107:216–23.
- Goldman, Lee. Chapter 68 – Pulmonary Hypertension. Goldman-Cecil Medicine. Philadelphia, PA: Elsevier Saunders, 2015.
- Foris V, Kovacs G, Tscherner M, et al. Biomarkers in pulmonary hypertension: what do we know? Chest 2013; 144 (1): 275-283.
- Benza RL, Miller DP, Gomberg-Maitland M, et al. Predicting survival in pulmonary arterial hypertension: insights from the Registry to Evaluate Early and Long-term Pulmonary Arterial Hypertension Disease Management (REVEAL). Circulation 2010;122:164–72.
- Tan RT, Kuzo R, Goodman LR, et al. Utility of CT scan evaluation for predicting pulmonary hypertension in patients with parenchymal lung disease. Medical College of Wisconsin Lung Transplant Group. Chest 1998;113:1250-1256.
- Zamanian RT, Haddad F, Doyle RL, et al. Management strategies for patients with pulmonary hypertension in the intensive care unit. Crit Care Med 2007; 35:2037–50.
- Kerbaul F, Rondelet B, Motte S, et al. Effects of norepinephrine and dobutamine on pressure load-induced right ventricular failure. Crit Care Med 2004;32:1035–40.
- Oztekin I, Yazici S, Oztekin DS, et al. Effects of low-dose milrinone on weaning from cardiopulmonary bypass and after in patients with mitral stenosis and pulmonary hypertension. Yakugaku Zasshi 2007;127:375–83.
- Kwak YL, Lee CS, Park YH, et al. The effect of phenylephrine and norepinephrine in patients with chronic pulmonary hypertension. Anaesthesia 2002;57: 9–14.
- Tongers J, Schwerdtfeger B, Klein G, et al. Incidence and clinical relevance of supraventricular tachyarrhythmias in pulmonary hypertension. Am Heart J 2007; 153:127–32.
- Greenwood J, Spangler R. Management of crashing patients with pulmonary hypertension. Emerg Med Clin N Am 2015; 33: 623-643.
- http://www.ncbi.nlm.nih.gov/pubmed/25986329






5 thoughts on “The Crashing Pulmonary Hypertension Patient”
Great read, thanks JB
Thank you for your nice and concise update on this subject. I have one remark about the last section; if the atrial kick is of utmost importance, then rythm control not rate control is the priority; or is it not? It is therefore that one should cardiovert early in these cases.
That would be my thought too. DC cardiovert, and optimise Mg and K.
Great review for very complex patients! The pulmonary vasculature has alpha 1 receptors as well which norepinephrine would constrict. Consider vasopressin as your pressor of choice to spare this. Also, in severe RV dysfunction the right sided CO is very dependent on HR so choice of your ionotrope may be guided by the underlying HR (high choose milrinone, low choose epi or dobutamine). The higher heart rates will decrease your diastolic filling time and help put the RV dilatation back on the Starling curve. In places that have it, I judiciously use iNO. No real side effects, well tolerated, and gives after load reduction for the RV.
Pingback: What’s the Diagnosis? By Dr. Julie Calabrese -