Author: Anthony DeVivo, DO (@anthony_devivo, EM-Critical Care Fellow, Icahn School of Medicine- Mount Sinai Hospital) // Reviewed by: Alex Koyfman, MD (@EMHighAK), Brit Long, MD (@long_brit) and Manpreet Singh, MD (@MprizzleER)
Welcome back to Unlocking Common ED Procedures! Today, we focus on central venous lines in the ED.
Check out our new downloadable procedure card with QR code link to the article. Print them out and be ready to go over it with your learners!
Case 1:
A 55-year-old female with a past medical history of hypertension and diabetes was admitted for pneumonia 10 hours ago and is currently boarding in the Emergency Department (ED) whilst awaiting a telemetry bed. A nurse tells you the patient has become hypotensive and more confused. After evaluating the patient and providing an intravenous (IV) fluid bolus, the patient remains hypotensive, tachycardic, tachypneic, hypoxic, and febrile.
You request the nurse to start norepinephrine and are quickly reminded that as per hospital policy, peripheral vasopressors may only be utilized as a temporizing measure while definitive central venous access is obtained. While the peripheral norepinephrine drip is started, you begin to consider the ideal location of central venous access in this patient. Is there a particular site safer than others? Might one site have a lesser chance of infection over another? What if the ultrasound is in the shop for service over the holiday weekend?
Case 2:
A 22-year-old female with no past medical history presents to the ED via EMS after being struck while riding an electric scooter by a vehicle going approximately 30 mph. She presents awake, alert, and in severe painful and respiratory distress. Initial vitals include BP 80/40, HR 130, RR 30, and O2 Sat 91% on non-rebreather.
On secondary survey, the patient has obvious signs of blunt thoracic, abdominal, and pelvic trauma in addition to a right sided open femur fracture. Paramedics were only able to obtain intravenous access via a 22g IV in the right hand. As you rapidly run through ATLS in your head and organize the order of interventions necessary, the RN affirms she cannot get intravenous access on this patient. What is your next step? Will you pursue venous access through an ultrasound guided peripheral IV? Is this patient suitable for an IO? Is this a time to go straight to central venous access? If so, which location and why?
Introduction
The ability to obtain rapid IV access in any patient, regardless of the clinical scenario, is a quintessential skill practiced by emergency physicians. While most patients in the ED will simply require peripheral IV access, the vignettes above present two critically-ill patients in very different circumstances, both of whom will likely require central venous catheter (CVC) placement. While the location of insertion is vital in certain scenarios, it does not define a CVC. A CVC is actually defined as a catheter whose tip resides within the superior vena cava, inferior vena cava, or at the junction between the vena cava and the right atrium (cavoatrial junction). Central venous access is obtained via the readily utilized Seldinger technique, in which a catheter is placed over a guidewire that has been threaded into a vessel through an introducer needle. The Seldinger technique is the foundation of most intravascular procedures and can be utilized for both peripheral and central venous catheters. The methodology has evolved over the past several decades and is now performed with or without ultrasound guidance, both of which techniques we will discuss below. This article will focus on the placement of CVCs including indications, contraindications, site choice, and complications. In addition, we will discuss the current literature behind the insertion site options and when pathophysiology should play a role in this decision. Since there are a variety of approaches and subtypes of catheters that can be placed depending upon the clinical scenario, the conclusion of this article will specifically review the placement of an internal jugular triple lumen catheter. Further posts under this broad overview will continue to discuss alternative approaches and catheter subtypes.
Central Venous Catheters1-3
One of the most common type of central venous access used by emergency physicians are triple lumen catheters. These catheters provide multiple ports of access that coalesce into a single catheter that resides in a central vessel for the administration of anything from blood products to hypertonic solutions to medications that may potentially damage or sclerose peripheral vessels. In addition, CVCs mitigate the risk of the potentially deleterious effects that certain medications, such as vasopressors, may have in the setting of peripheral extravasation.

Aside from triple lumen catheters, emergency providers may also find themselves in clinical scenarios where patients without appropriate access require emergent dialysis that necessitates the placement of a central venous dialysis catheter. These catheters are wider in diameter and may come as a two-port catheter solely for dialysis or with a third port, through which medications may be administered.
Introducer sheath placement, also commonly referred to as a cordis, may be required in critically-ill patients while in the ED. These are wide-bore, single lumen catheters that end with a hub covered by a hemostatic valve and a laterally extending side port. The hub is utilized as a port of intravascular entry for the placement of additional catheters or a temporary transvenous pacer, while the side port can be used for the rapid administration of blood products, intravenous fluids, or other medications. These catheters are useful in the setting of hemorrhagic shock or unstable bradycardia that is unable to be temporized with transcutaneous pacing.

While we will specifically discuss the placement of triple lumen catheters below, the methodology and technique utilized for the placement of dialysis catheters, as well as introducer sheaths are the same. The nuances that differentiate the placement of these from triple lumen catheters will be discussed in separate reviews.
Central Access Site
Once the decision is made to obtain central venous access and the type of catheter is chosen based upon the clinical scenario, a site for insertion must also be determined. The insertion site may be based on the type of catheter indicated as well as factors related to patient history, anatomy, and the acuity of the situation. In the setting of triple lumen catheter placement there are 3 potential insertion sites; the internal jugular, subclavian, or femoral vein.1,3,4The debate over the safest insertion site is often broken down into the immediate mechanical complications, such as a pneumothorax or hematoma formation, and the more delayed complications of vessel stenosis and catheter-associated infection.3,4 While the deliberation over the preferred site has been ongoing for decades, there is still no finite answer. There is substantial conflicting data over which insertion site presents the lowest risk of catheter-related infection. Most recently, a multicenter RCT comparing the complications of the three insertion sites (placed under sterile conditions) showed subclavian access entry has a lower rate of catheter-related infections (0.6%) and venous thrombosis (2.0%) with a higher rate of iatrogenic pneumothorax requiring chest tube placement (1.5%).4 It should also be noted that in this study there was minimal difference in catheter-related infections (two) when comparing jugular and femoral vein access, which opposes the not uncommon sentiment that femoral central lines are “dirty” and should be quickly removed after stabilization.4 To add to the confusion and conflicting data on catheter-related infections, there is also substantial data suggesting all three insertion sites have similar rates of infection.5-6,8
Thus, emergency physicians are left with a dilemma – is a 1.5% risk of pneumothorax worth the greater than doubled risk of central line site infection and deep vein thrombosis formation with internal jugular and femoral vein access? Given the invasive nature of the procedure required for the resolution of those pneumothoraces (chest tube placement) it is generally accepted that the greater, though still small, risk of central line site infection and deep venous thrombosis is preferable. When it comes to deciding between femoral and internal jugular venous access, the traditional sentiment that femoral access bears a higher risk of infection seems to no longer be supported by recent literature and the procedure may have a lower rate of hematoma formation.9 Given the safety of the internal jugular approach under ultrasound guidance, combined with the still widely held sentiment that femoral central lines hold a higher risk of being contaminated, the internal jugular approach is generally the preferred central venous access site in the critically-ill, but not perimortem, situation.4-9 However, if ultrasound is not available, there is a high risk of procedure related bleeding, or if the central line must be placed rapidly without sterility due to critical acute decompensation, femoral access may be advantageous. While subclavian access is an invaluable skill and may be necessary in certain scenarios, the risk of pneumothorax tends to deter physicians from making this their access point of choice. However, with appropriate training and practice, the risk of pneumothorax can be mitigated, making this a viable option for central access. This is a decision every emergency physician will have to make for themselves utilizing recent data, in conjunction with the clinical scenario, patient history, body habitus, and personal comfort with each method of central line placement. That being said, this literature sheds light on the need for emergency physicians to be well versed in all three methods of central venous access as no access site is clearly superior and certain clinical scenarios may warrant the placement of one over the other.4-8
Emergent Indications1-4
- Large volume resuscitation (crystalloid or blood products)
- Emergent intravenous access in patients that present particular difficulty in obtaining peripheral access (i.e history of end stage renal disease, vasculopathy, obesity, intravenous drug abuse)
- Prolonged intravenous vasopressor administration
- Administration of highly concentrated electrolyte solutions such as hypertonic saline or large doses of potassium chloride
- Transvenous pacer placement
- Plasmapheresis
- Pulmonary artery catheter placement
This list summarizes the most common indications for central venous access in the ED. There are other non-emergent indications such as the administration of total parenteral nutrition that may require central venous access but is unlikely to be the indication for placement in the ED.
Contraindications
The establishment of central venous access in the ED generally implies a clinical scenario in which peripheral access is unobtainable or inadequate in a critically-ill patient. In light of this, there are no absolute contraindications to emergent central venous access. However, there are relative contraindications as well as risks that can be mitigated by appropriate site insertion choice which will be discussed further below.
- Coagulopathy9-10
- There is no literature to support that any strict degree of coagulopathy increases the likelihood of bleeding complications during central line placement. There are no data to support the use of prophylactic fresh frozen plasma or platelet transfusion in order to mitigate the risk of bleeding complications.However, there are data to support that central venous access placement in patients with an INR of up to 3, as well as platelet counts as low as 20,000/dl have no increased bleeding risk.9-10
- Signs of infection at insertion site
- Central venous access is a sterile procedure unless being performed under dire circumstances. As such, an insertion site with any overlying signs of infection should be avoided. As we have discussed, there are multiple site insertion options making placement through a skin infection likely unnecessary.
- Thrombus
- With the advent of ultrasound, physicians are now able to obtain central venous access while directly visualizing the vein. As such, the intended vein of insertion should be assessed for patency prior to placement. Any signs of thrombus, such as limited compressibility or direct visualization of a thrombus, should prompt an alternative insertion site.
- Distorted Anatomy or Trauma
- Distorted anatomy, whether acute from a traumatic injury or from prior surgical interventions, may complicate central line placement. If there are apparent anatomical barriers to the intended access site, an alternative procedure site should be considered if possible.
Technique
Central venous access was traditionally obtained via a landmark based approach for all three sites of entry. However, with the advent and ease of accessibility of ultrasonography in the ED, ultrasound guidance has become the standard of care, particularly for the internal jugular approach, as it has been shown to reduce procedural failure as well as complications.7 However, emergency physicians must be able to perform critical procedures under less than ideal circumstances making the knowledge and ability to perform landmark based central venous access an important skill. In addition, having a firm grasp of the landmark based approach will facilitate ultrasound usage.
- Internal Jugular Vein Landmarks
- Place patient in 15-30 degrees of trendelenburg with the neck turned to the contralateral side of the procedure site.
- Insertion should be between the medial and lateral heads of the sternocleidomastoid, just lateral to the carotid artery. This allows the formation of a triangle of relative safety with the clavicle acting as the base of the triangle and the medial and lateral heads of the sternocleidomastoid forming the two sides.
- In order to more clearly define these landmarks, the trachea can be palpated with the non-procedure hand at the level of the cricoid allowing the hand to both palpate and protect the trachea and carotid artery while assisting in defining the medial head of the sternocleidomastoid.
- The needle should be inserted at the apex of this triangle, where the 2 heads of the sternocleidomastoid proximally adjoin, at a 45 degree angle. Once the skin has been pierced, negative pressure should be applied to the syringe until blood return is achieved.
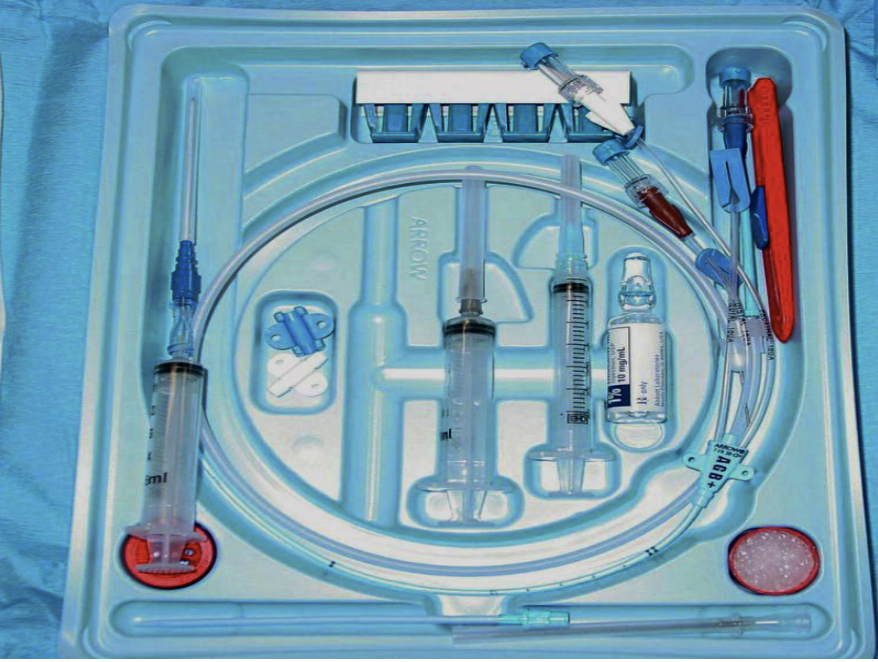
The Procedure
Below we will specifically discuss the performance of ultrasound guided internal jugular vein triple lumen catheter placement. In following articles, we will further discuss the approaches to the placement of femoral and subclavian vein central venous access.
- Ultrasound Guidance
- Place patient in 15-30 degrees of trendelenburg with the neck turned to the contralateral side of the procedure site.
- The trendelenburg position will facilitate venous congestion, making the internal jugular vein more prominent and easily visualized/cannulated.
- With the knowledge of the anatomical landmarks above, place the high-frequency linear ultrasound probe horizontally at the apex of the triangle formed by the 2 heads of the sternocleidomastoid.
- Scan the neck both caudad and cephalad to assess the patency, position, and trajectory of the internal jugular vein. It is key to have an understanding of the directionality of the vein as it tracks caudally to anticipate difficulty with puncture or cannulation.
- Both internal jugular veins should be assessed, however the right side is a more direct route to the cavoatrial junction and is preferable.
- While assessing the best point for puncture and insertion, keep in mind:
- Proximity and position of the internal jugular vein in relation to the carotid artery
- Proximity of the insertion site to the clavicle/apex of the lung
- Anatomic variations that may cause difficulty with cannulation.
- Once a site is chosen, preparation of the patient and procedure site may begin.
- Place patient in 15-30 degrees of trendelenburg with the neck turned to the contralateral side of the procedure site.

- Preparation
- Place extra sheet, towel, or “chuck” underneath patient’s head in order to avoid making a mess of the bed/sheets underneath the patient.
- Turn the patient’s head to the contralateral side and clean the intended entry site with chlorhexidine or other sterilizing solution.
- Apply sterile drape over patient with opening over the sterilized procedure site.
- Apply sterile ultrasound probe cover and place probe on field.
- This generally requires a non-sterile assistant to pass you the probe.
- Prior to applying the sterile cover, a small amount of ultrasound gel should be placed on the inside of the cover.
- The probe utilized should be a high-frequency linear probe. Before starting, a trick to staying oriented to midline on the screen is to turn on M-mode which will give you a midline marker.
- Prepare sterile table and open central line kit.
- If a sterile table or mayo stand is unavailable, the kit may be opened and prepared on-top of the patient sterile field.
- Assess and prepare the equipment within the central line kit
- Flush all 3 ports of the line with sterile saline, apply heparin locks to 2 out of the 3 ports and leave the third port (frequently brown colored) uncapped for the wire to be removed through.
- Check the wire to assure it advances easily through the sheath.
- Break the seal within the finder needle and assure it is not too tightly locked on the syringe. If the finder needle is fastened too tightly it will be difficult to remove from the syringe while maintaining the needle within the vessel.
- Arrange equipment within arm’s reach in the order they will be utilized for the procedure:
- Finder needle attached to syringe
- Guide Wire
- Gauze
- Scalpel
- Dilator
- Catheter
- Clean the entry site with alcohol-based solution an additional time before beginning the procedure.
- Procedure
- Utilize a small-bore needle to inject a wheal of lidocaine at the entry site followed by injecting lidocaine throughout the tissues down to the vessel under ultrasound guidance to form an anesthetic tract. Small-bore needles can be difficult to identify under ultrasound, but visualization of tissue displacement during lidocaine injection should be more easily appreciated.
- Using the finder needle, puncture the skin 0.5-1cm from the ultrasound probe at a 45-60 degree angle.
- While 45 degrees is the traditional teaching, a higher (60-70 degrees) angle may facilitate directly visualizing the needle tip entering the vessel. A more shallow angle may lead to the needle tip passing out of view when attempting to enter the vessel.
- Once through the skin, advance towards the vein while watching the needle tip displace the tissues under ultrasound guidance and applying negative pressure. Once you visualize the tip of the needle tenting the vessel wall into the lumen of the vessel, apply additional gentle pressure to puncture the vessel wall.
- Once smooth blood return is obtained, drop the ultrasound probe and gently decrease the angle of needle and syringe to 15-30 degrees. Once the angle has been decreased, apply additional negative pressure to assure the needle tip is still within the vessel.
- Gently remove the syringe from the needle tip with the dominant hand while stabilizing the needle tip with the non-dominant hand. Generally, once the syringe is removed there should be some blood dripping from the open end of the needle. However, in patients who are intravascularly depleted there may be minimal to nearly no blood return without negative pressure.
- If blood return ceases under negative pressure:
- Slightly withdraw the needle, as the needle may have punctured the posterior wall.
- Take another look with the ultrasound to assess the location of the needle tip and slightly adjust needle depth based on this.
- Small movements at this point are key to avoid vessel damage and hematoma formation.
- If blood return is brisk:
- Internal jugular blood return can be brisk, particularly in patients with elevated central venous pressures (cardiogenic shock).
- Blood return can also be brisk if the jugular vein is in close proximity to the carotid artery.
- If blood return is pulsatile:
- Even in the setting of cardiogenic shock or other pathologies that lead to significantly elevated central venous pressures, pulsatile blood return is highly suspicious for inadvertent carotid artery puncture. If there is any suspicion you have punctured the carotid artery, the needle should be removed and pressure should be held for several minutes. Do not apply too much pressure, which may cause a vagal response.
- As an alternative to using the isolated finder needle technique described above, an angiocatheter may also be utilized. In this technique, an angiocatheter is placed over the finder needle and advanced into the vessel in a similar manner as previously discussed. Once the needle tip is visualized within the blood vessel and blood return is readily obtained, the catheter may be advanced over the needle into the vessel. The advantage of using an angiocatheter is that it is less prone to being dislodged from the vessel while attempting to remove the syringe from the needle or advancing the wire. In addition, the angiocatheter can act as a “placeholder” within the vessel if the wire needs to be removed or redirected.
- Advance the wire through the needle to the 20cm mark. If there is any sense of resistance, stop advancing as this may be an indication of the needle being inappropriately positioned. Advancing the needle through resistance can lead to kinking of the wire and creation of a false tract through the posterior wall of the vessel into the deep tissues, as well as a non-functional central line.
- If resistance is met with wire placement:
- Withdraw the wire to the point at which there was no resistance, slightly decrease the angle of the needle, and attempt to reinsert the wire.
- If resistance persists:
- Withdraw the wire from the needle and reattach the syringe to assure there is still blood return with negative pressure. If there is no blood return, reassess needle placement with the ultrasound as previously discussed.
- If there is blood return, attempt reinsertion of the wire once again.
- If resistance persists after confirming needle location and blood return:
- This may indicate an intraluminal obstruction, and an alternative access site should be considered.
- Once the wire is advanced to the 20cm mark, the needle may be removed over the wire. The wire should still move smoothly in and out of the puncture site with minimal force.
- Confirm the wire is within the lumen of the internal jugular vein utilizing the ultrasound.
- The wire should be visualized within the internal jugular vein and then tracked down to ensure it does not pierce the posterior wall of the vessel.For documentation purposes, an image may be saved demonstrating the wire within the compressible internal jugular vein. Below is an example of a wire within a vessel in longitudinal view.
- If resistance is met with wire placement:
- If blood return ceases under negative pressure:

-
- Once the wire is confirmed within the vessel, a small nick with a #11 blade scalpel may be made to facilitate dilator entry. The nick should be superficial with the blade facing away from the wire and the patient’s neck.
- Place several 4×4 gauze underneath the wire as the next step will lead to more bleeding.
- Advance the dilator over the wire to the entry site and gently insert the dilator into the skin with a twisting motion. The dominant hand should be advancing the dilator with a firm drip on it just distal to the skin while the nondominant hand maintains control of the wire. The dilator should only be advanced approximately ⅓ the way into the skin as it is only meant for the tissues leading up to the vessel, not the vessel itself. Dilating the vessel will only lead to more bleeding. However, this often occurs due to difficulty anticipating where the vessel begins, hence the extra gauze.
- The gentle twisting motion is vital to successful dilation. Resistance or forceful dilation can lead to kinking of the wire which can make passing the catheter exceedingly difficult.
- Never let go of the wire.
- Pass the catheter over the wire until the tip of the catheter is near the skin. If the wire tip is not yet visible outside of the open brown port of the catheter, slowly withdraw the wire from the entry site into the catheter. Once the wire extrudes from the brown port it may be stabilized with the nondominant hand while the dominant hand advances the catheter into the vessel.
- If a 10-13cm long catheter is being utilized, the catheter may be advanced to the hub within the right internal jugular vein.
- If the left internal jugular vein is cannulated, a 16cm catheter can be utilized and advanced to the hub.
- Once the catheter is advanced into the vessel, the wire may be removed through the open port (often brown in color).
- Once the wire is out, a heparin lock should be attached to the brown port. The other ports should already have these locks attached.
- Each port should be checked to assess that they readily draw blood back and flush with ease, after which they should all be locked.
- Take a breath and prepare for the most difficult step: the dressing.
- Dry the insertion site with gauze, then if available, clean again with a chlorhexidine swab.
- Apply antibiotic patch. Applying this now is easier than doing so after the suturing.
- Secure line with suture. If the area is not anesthetized from prior injection, additional lidocaine should be injected.
- Apply sterile dressing.
- Order post-procedural chest x-ray.
Post-Procedural Complications
- Pneumothorax
- While the risk of iatrogenic pneumothorax during ultrasound-guided central line placement is low (0.5%), it is still a major complication to consider, particularly in a patient who has acute cardio-pulmonary decompensation post-procedurally.4 In order to mitigate this risk, the insertion site should be made more proximally if anatomy under ultrasound guidance allows. In addition, it may be possible to visualize the pleural line while assessing anatomy and its depth to avoid puncture. Finally, during the procedure, the needle tip should always be kept in plane with the ultrasound probe; if you know where your needle tip is at all times, you are less likely to inadvertently injure surrounding anatomical structures. If the patient does decompensate post-procedurally with signs of pneumothorax with or without tension physiology, ultrasound should be utilized to assess for lung sliding in conjunction with clinical assessment in order to quickly confirm the diagnosis and intervene appropriately. For a review of chest tube insertion, check out our emdocs post…
- Arterial Puncture
- If even under ultrasound guidance the carotid artery is inadvertently punctured, it’s ok! The important aspect of this is to recognize under ultrasound guidance or during aspiration that the artery has been punctured. Remove the needle from the skin, temporarily abort the procedure, and hold firm pressure for several minutes.
- Carotid Artery Cannulation
- If upon cannulation of the vessel and removal of the wire the blood flow appears arterial, this is a more serious issue than just a needle puncture as the vessel has been cannulated already and the catheter is theoretically acting as a means of tamponade — if it’s removed, the bleeding may not be controllable in the ED. Thus, the catheter should be left in place and either vascular surgery or interventional radiology should be consulted for appropriate removal.
- The Lost Wire
- This is one of the most commonly discussed and feared complications of central line placement. While it may theoretically seem difficult, losing the wire, particularly during the emergent placement of a central line, can happen. If the catheter has been completely inserted without removing the wire but the wire is not protruding from the brown central line port, there is still a chance of retrieval. First, inspect the port and line that the wire was inserted over. If the wire is able to be visualized, use a Kelly clamp to tightly stabilize the wire within the line and then remove the entire catheter with the wire while the clamp is attached. At this point, pressure will need to be held while a new access site is determined.
- If the wire is not able to be visualized within the port, there is a possibility that some of the wire still resides within the catheter and can be retrieved by tightly clamping the catheter with a Kelly clamp just distal to the skin insertion site and removing the entire catheter.
- If the wire is unable to be retrieved via these or any other method, further imaging should be obtained and either cardiothoracic surgery or interventional radiology should be emergently consulted for retrieval.
Central Venous Access Site Summary
- Subclavian access has an overall lower rate of infection and thrombosis with an increased risk of pneumothorax.
- Subclavian access may be preferable in the setting of blunt and/or penetrating thoraco-abdomino-pelvic trauma if there is concern for vascular injury at or above the femoral vein and the internal jugular vein is inaccessible due to cervical spine precautions.
- Femoral access was initially thought to have a higher infection rate with the advantages of being distant from the thorax and an easy site of compression if a hematoma develops. However, more recent data suggests there is no increased risk in catheter-related infections with femoral access, compared with other sites.
- Internal jugular access has a relatively low risk of infection as well as iatrogenic pneumothorax under ultrasound guidance. Thus in critically-ill patients who are not imminently dying, ultrasound-guided internal jugular access is often the preferred site of entry.
- There is no single best access site for a central venous catheter and emergency physicians may find themselves in situations where any of the 3 sites may be necessary and/or best for particular patient or clinical scenario.
Pearls and Pitfalls
- If time allows, consider and evaluate the anatomy of multiple insertion sites. The right internal jugular may be the go-to site for many, but the anatomy of the left internal jugular may be more appropriate when visualized under ultrasound.
- If there is concern for coagulopathy or bleeding, the femoral vein may be the more appropriate choice, given the compressibility of the site.
- Always keep the tip of the needle in view under ultrasound when advancing in order to avoid injury to underlying anatomic structures.
- Once the needle is within the vein and flow is obtained into the syringe, decrease the angle and recheck flow to assure the needle is still within the vessel.
- Brisk, even near pulsatile blood flow, can be seen when placing an internal jugular vein central line in the setting of certain pathologies such as heart failure or cardiogenic shock due to a severely elevated central venous pressure. In this situation, careful confirmation of the wire within the jugular vein should be performed with ultrasound.
- If resistance is felt while advancing the wire, cease advancement, decrease the needle angle and reattempt. If there is still resistance, remove the wire and reassess flow with the syringe.
- Never let go of the wire, but if it is lost, attempt to clamp the line and remove the wire and line as discussed above. If the wire is unable to be removed, obtain emergent imaging and consultation for removal.
- The emergency department can be a difficult place to maintain sterility. If there is any concern regarding the sterility of the central line, it is prudent to inform the admitting team for follow up care.
Rapid Procedure Review
- Position the patient in trendelenburg.
- Assess the anatomy under ultrasound bilaterally to assure the safest site is utilized.
- Sterile preparation of the procedure site and the proceduralist.
- Drape patient.
- Prepare sterile ultrasound probe cover.
- Prepare central line by flushing all lines and applying caps to 2 of the 3 ports.
- Anesthetize insertion site by first making a wheel of lidocaine at the site and then injecting lidocaine throughout the subcutaneous and deep tissues under ultrasound guidance.
- Under ultrasound guidance, insert introducer needle through the skin and advance towards the internal jugular vein via the tract of lidocaine previously made.
- Always keep the needle tip in view under ultrasound.
- Once flow is obtained within the syringe, drop the angle of the needle and reconfirm blood flow. See above for methods of managing loss of flow.
- Remove syringe from needle and advance wire. See above for methods of managing difficulty in wire advancement.
- Once the wire is at least 20cm within the vessel, remove the needle over the wire and confirm placement of the wire under ultrasound.
- Make a small nick of a #11 scalpel with the blade facing away from the midline.
- Advance dilator over the wire with the intention of dilating the soft tissue but not the vessel. This depth of dilation will be dependent on body habitus and anatomy which should be taken into account when assessing the anatomy under ultrasound.
- Advance the catheter over the wire and carefully feed the wire back through the catheter once at the skin to assure control of the wire before catheter advancement into the vessel.
- Once the catheter is within the vessel, remove the wire completely.
- Cap the third port from which the wire was removed.
- Flush all 3 ports.
- Secure line with suture.
- Apply antibiotic patch and sterile dressing.
- Sit patient up and order post-procedure chest x-ray.
References/Further Reading
- Wall Tintinalli, Judith E., et al. Tintinalli’s Emergency Medicine: a Comprehensive Study Guide. McGraw-Hill Education, 2016.
- Walls, Ron M., et al. Rosen’s Emergency Medicine: Concepts and Clinical Practice. Elsevier, 2018.
- Parrillo, Joseph E. Critical Care Medicine: Principles of Diagnosis and Management in the Adult. Elsevier, 2019.
- Parienti, Jean-Jacques, et al. “Intravascular Complications of Central Venous Catheterization by Insertion Site.” New England Journal of Medicine, vol. 373, no. 13, 2015, pp. 1220–1229., doi:10.1056/nejmoa1500964.
- Marik, Paul E., et al. “The Risk of Catheter-Related Bloodstream Infection with Femoral Venous Catheters as Compared to Subclavian and Internal Jugular Venous Catheters.” Critical Care Medicine, vol. 40, no. 8, 2012, pp. 2479–2485., doi:10.1097/ccm.0b013e318255d9bc.
- Ge, Xiaoli, et al. “Central Venous Access Sites for the Prevention of Venous Thrombosis, Stenosis and Infection.” Cochrane Database of Systematic Reviews, 2012, doi:10.1002/14651858.cd004084.pub3.
- Leung, Julie, et al. “Real-Time Ultrasonographically-Guided Internal Jugular Vein Catheterization in the Emergency Department Increases Success Rates and Reduces Complications: A Randomized, Prospective Study.” Annals of Emergency Medicine, vol. 48, no. 5, 2006, pp. 540–547., doi:10.1016/j.annemergmed.2006.01.011.
- Deshpande, Kedar S., et al. “The Incidence of Infectious Complications of Central Venous Catheters at the Subclavian, Internal Jugular, and Femoral Sites in an Intensive Care Unit Population*.” Critical Care Medicine, vol. 33, no. 1, 2005, pp. 13–20., doi:10.1097/01.ccm.0000149838.47048.60.
- Parienti, Jean-Jacques, et al. “Femoral vs Jugular Venous Catheterization and Risk of Nosocomial Events in Adults Requiring Acute Renal Replacement Therapy.” Jama, vol. 299, no. 20, 2008, p. 2413., doi:10.1001/jama.299.20.2413.
- Weerdt, Emma K. Van De, et al. “Central Venous Catheter Placement in Coagulopathic Patients: Risk Factors and Incidence of Bleeding Complications.” Transfusion, vol. 57, no. 10, 2017, pp. 2512–2525., doi:10.1111/trf.14248.
- Haas, Brian, et al. “Large-Bore Tunneled Central Venous Catheter Insertion in Patients with Coagulopathy.” Journal of Vascular and Interventional Radiology, vol. 21, no. 2, 2010, pp. 212–217., doi:10.1016/j.jvir.2009.10.032.









