Author: Anthony DeVivo, DO (@anthony_devivo, EM Resident Physician, Mount Sinai St. Luke’s-West) // Edited by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit); and Manpreet Singh, MD (@MprizzleER)
Welcome back to Unlocking Common ED Procedures! Today, we look at paracentesis.
Check out our new downloadable procedure card with QR code link to the article. Print them out and be ready to go over it with your learners!
Case
A 58-year-old male with a past medical history of alcohol-induced cirrhosis presents to the Emergency Department (ED) for 3 days of progressively worsening confusion, myalgias, and fatigue. Upon arrival to the ED, the patient is normotensive, tachycardic, and febrile. He is awake but confused with a distended and diffusely tender abdomen. His urinalysis indicates he has a urinary tract infection, and his chest x-ray displays a new infiltrate. Should you still consider Spontaneous Bacterial Peritonitis (SBP)? Do the alternative sources for this patient’s condition change whether or not a paracentesis should be performed in the ED?
Background
A paracentesis is the procedure by which clinicians gain access to and assess ascitic fluid that has collected within the confines of the peritoneal space. It is performed under sterile conditions by introducing an 18-20g needle into the peritoneal space where a large pocket of fluid has formed.1-2 The fluid collection may be solely for diagnostic purposes or also for therapeutic purposes in order to relieve symptoms caused by large volumes of intraperitoneal fluid.3 The remainder of this article will focus on traditional and up to date, evidence-based indications, contraindications, and the performance of paracentesis in the ED.
Indications:1-3
- Diagnostic
- Assessment for SBP
- Assessment of undifferentiated or new-onset ascites including etiologies such as malignancy or newly diagnosed cirrhosis
- Therapeutic
- Relief of symptoms secondary to increased intraabdominal pressure from ascites such as abdominal pain, early satiety, and respiratory compromise
Considerations and Contraindications:1,3,5-8
While the only absolute contraindication to a paracentesis is the presence of an “acute abdomen” such that would require surgical intervention, there are several scenarios and confounding conditions which should be considered before performing a paracentesis.1,3These considerations or relative contraindications will be discussed below in conjunction with the literature that supports or negates them.
- Thrombocytopenia and Coagulopathy
- One of the most significant concerns in performing any invasive procedure is the risk of peri- or post- procedural bleeding. This risk is theoretically confounded in the setting of patients who are “auto-anticoagulated” due to baseline coagulopathy and/or thrombocytopenia secondary to chronic conditions (cirrhosis).1,3 This theoretical risk has been evaluated in multiple trials, which suggest no significant increased peri or post-procedural bleeding risk.5,7-8 Prospective literature supports no increased risk of bleeding after the performance of a paracentesis in the setting of an INR<8.0 or Platelet count >20,000/ml.5 Thus, while there is understandable concern that patients who are coagulopathic and thrombocytopenic are at risk for bleeding, there is extensive literature to support that these lab abnormalities are not a reason to defer the performance of a paracentesis when clinically indicated.6-7 In addition, there is no evidence to support the prophylactic administration of blood products such as platelets or FFP in patients with no clinical signs of bleeding.8
- Disseminated Intravascular Coagulation
- While coagulopathy and thrombocytopenia have not been shown to be independent risk factors for bleeding complications during a paracentesis, clinical suspicion for disseminated intravascular coagulation (DIC) is a contradiction to the procedure. In scenarios with clinically apparent DIC or a hyperfibrinolytic state, deferment of paracentesis and assessment of the need for pre-procedure blood products may be considered. Due to the uncommon nature of these clinical scenarios, literature is limited, and further discussion with the admitting team, as well as the intensive care team, is necessary.2,5,7
- Large Bowel Ileus
- In patients with clinically apparent or diagnosed large bowel ileus, caution is warranted when performing a paracentesis. As we will further discuss later, ultrasound guidance should be utilized in most paracenteses, but it is particularly vital if performing this procedure on a patient with significantly distended bowel due to the risk of bowel perforation.
- Overlying abdominal wall cellulitis and/or suspected abdominal wall infection
- As with any invasive procedure, skin entry through an area clinically suspected to be cellulitic is contraindicated due to risk of bacterial translocation.
- Pregnancy
- While there is no literature to support pregnancy as a true contraindication to a paracentesis, caution is needed to avoid important anatomic structures.
Anatomy and the age of Ultrasound2,4,5,9-11
Traditionally, the performance of a paracentesis was done without imaging guidance, and a fluid pocket was located solely by percussing the abdomen until a significant area of dullness was isolated. However, ultrasound guidance has become routine to perform a paracentesis.9This does not, however, mitigate the importance of percussion as a physical exam technique when assessing patients with clinically apparent ascites. In addition, it is still important to know the anatomical landmarks and considerations for a paracentesis.
Utilizing the ultrasound, identify a pocket in the right or left lower quadrant that is lateral in the rectus abdominis. This is avoids the liver and spleen in the upper quadrants, as well as the inferior epigastric vessels in the medial lower quadrants.2,4,11It is also important to take into consideration more superficial abdominal anatomy such as engorged veins that may obstruct needle insertion during the procedure. Once a substantial pocket has been identified that does not contain superficial viscera at risk for perforation, the site should be marked, with anatomical landmarks taken into consideration. If it has not already be done, consent for the procedure should be obtained, after which you will be ready to set up and begin.
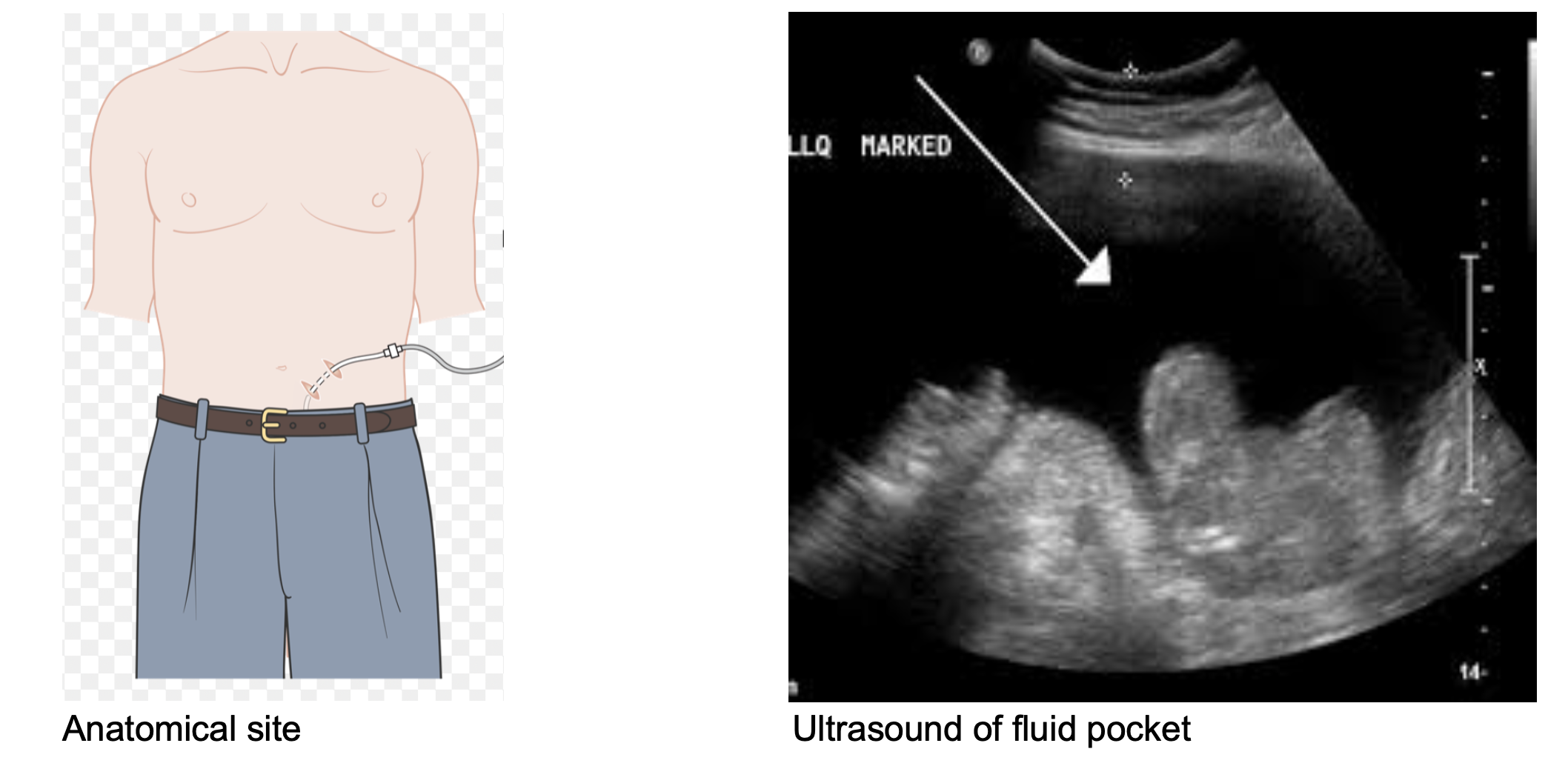
The Procedure2,4,5,9-10
- Positioning
- The patient should be positioned supine with the head of the bed angled at approximately 45 degrees allow fluid to dependently accumulate in the lower quadrants.
- Site Marking
- Utilizing the ultrasound, locate a fluid pocket and mark the procedure site with the anatomical considerations previously discussed kept in mind. You may utilize a marking pen or just make a light indentation at the intended site. The site is not yet sterile, and so depending on what type of marking pen and sterilization is utilized, the mark may be incidentally erased. In order to avoid losing the intended site, a semi-permanent marker that can withstand sterilization can be used. In addition, it is possible to use a blunt object such as the back of a marking pen to make a slight skin indentation at the site instead of an actual marker. In the setting of significant ascites and abdominal distention, the skin indentation should still be visible in the time it takes to sterilize and begin the procedure.
- Static vs Dynamic Ultrasound Guidance9
- A paracentesis may be performed with static ultrasound guidance, as discussed above, or dynamically during which you visualize the needle entering the fluid pocket within the peritoneal space. While it is not necessary to perform this procedure under dynamic guidance, it may be preferred in patients who only have a small fluid pocket for aspiration. If dynamic ultrasound guidance is utilized, a sterile probe cover is necessary to maintain procedure sterility.
- Preparation
- Once the patient is positioned supine with the head of the bed at 45 degrees and the site is marked, be sure to confirm that the patient is comfortable. Patients may experience worsening dyspnea in the setting of large peritoneal fluid volumes when placed supine.
- At this point, sterile gloves, mask, and cap should be worn. If dynamic ultrasound guidance is being utilized, a sterile sheath should be placed over the probe.
- A 10cm circumference around the marked site of insertion should be sterilized with chlorhexidine or betadine.
- Apply a drape over the sterilized area.
- If performing a therapeutic tap, be sure to have vacuum containers prepared at bedside before advancing any further.
The procedure described next will be for the performance of a static diagnostic and therapeutic paracentesis. At this point in preparation, a pre-packaged paracentesis kit should be sterilely opened at the bedside. We will discuss later a faster means of obtaining ascitic fluid for an isolated diagnostic tap that does not require a pre-made kit.

- Using 1% or 2% lidocaine with epinephrine, first make a small anesthetic wheal at the marked site. Then insert the needle, making sure to aspirate before injecting lidocaine along the track until you have anesthetized down to the peritoneum. This may be apparent, as you will aspirate ascitic fluid once you have reached the peritoneal cavity.
- Utilizing a #11 blade scalpel, make a small nick at the center of the intended procedure entry site. This is to allow easier insertion of the catheter after needle insertion.
- Displace the nicked skin either caudal or cephalad, and using your dominant hand, insert needle with attached catheter and syringe. Once you begin to advance the needle into the abdominal wall, the retracted skin may be released. The purpose of this step is to form a “Z track” which is a track of displaced subcutaneous tissue that will collapse on itself when the needle or catheter is removed in order to decrease post-procedural fluid leakage. This method may also be used when injecting anesthetic. There is no evidence to show this method of injection significantly decreases fluid leakage, but it is a generally accepted textbook practice.10
- Proceed with advancing the needle. Once the peritoneal cavity has been punctured, ascitic fluid should be easily aspirated while applying negative pressure to the syringe.
- Advance the catheter over the needle and withdraw the needle. In order to facilitate the transition between diagnostic and therapeutic fluid removal, a three-way stopcock can be attached to the catheter.
- Attach a 50cc syringe and aspirate fluid for diagnostic analysis.
- Some kits may come with sterile collecting tubes for fluid analysis. If there are not designated tubes within the procedure kit, a sterile urinalysis container can be utilized. If there is concern for SBP, the fluid should be placed in blood cultures bottles to increase the chance of detecting an organism. In addition, the following labs should be sent in order to assess for SBP:
- Cell count with differential analysis
- Gram stain
- Aerobic and anaerobic cultures
- Protein
- Glucose
- Albumin
- LDH
- Cytology if concern for malignant ascites
- In order to proceed with a therapeutic tap, lock the stopcock attached to the catheter, remove the 50cc syringe, and attached drainage tubing. You may now attach the tubing to either a drainage bag or 1-2L evacuated bottles. When using evacuated bottles for large volume drainage, the drainage tubing will have a spike at the end that should be inserted into the top of the bottles.
- Whether using a drainage bag or evacuated bottles, gravity is your friend. Arrange the bottles or bag so that gravity assists as much as possible with fluid flow.
- Caution should be taken only to puncture the evacuated bottle once, as more than one puncture may impede drainage.
- Another option in place of gravity and evacuated bottles is to utilize wall suction for drainage. Multiple wall suction canisters can be linked together with suction tubing. The first canister in this series can then be attached to the paracentesis drainage tubing. Once the 3-way valve is open, the wall suction can be turned on, and drainage should ensue. Once the first canister has filled, the fluid will immediately begin draining into the second and so on.
- Some kits may come with sterile collecting tubes for fluid analysis. If there are not designated tubes within the procedure kit, a sterile urinalysis container can be utilized. If there is concern for SBP, the fluid should be placed in blood cultures bottles to increase the chance of detecting an organism. In addition, the following labs should be sent in order to assess for SBP:

- In the setting of large volume paracentesis, the patient should have frequent hemodynamic reassessment as will be discussed further.
- If there is decreased or cessation of fluid flow:
- Recheck your stopcock position.
- Remove tubing, reattach syringe, and attempt to aspirate using negative pressure.
- Flush a small amount of sterile normal saline through the catheter. This may dislodge bowel or omentum stuck to the catheter tip.
- Utilizing the ultrasound, reassess fluid pocket.
- Once satisfactory fluid has been drained, removal the catheter, apply pressure, and bandage to the procedure site.
Quick Diagnostic Paracentesis
ED stock room short on paracentesis kits? There’s a way to perform a diagnostic paracentesis without the use of a fancy premade kit!
- Assess a fluid pocket, mark, anesthetize, and sterilize the procedure site as discussed above.
- Without a premade kit, a separate sterile drape will need to be obtained. If there are no separately packaged drapes stocked in the department, the drape from a laceration kit may be utilized. Also, do not forget to have tubes or a sterile urinalysis cup for specimen collection ready at the bedside.
- Instead of the paracentesis kit catheter, the procedure will be performed with a long (1.8-2”) angiocatheter with a gauge of 20 or less. The smaller the gauge, the more quickly the ascitic fluid will flow.
- Using the “Z-track” method described above, displace the skin and insert the angiocatheter. Once ascitic fluid begins to backflow, similar to when an angiocatheter is successfully placed into a vein, advance the catheter while retracting the needle and begin collecting fluid for diagnostic analysis.
- Once adequate fluid is collected, remove the catheter, apply a bandage, and the procedure is complete.
Post-Procedural Complications4,7,12-13
Post-paracentesis complications are uncommon, but when they do occur should be readily recognized and either managed immediately in the ED or further management should be discussed with the admitting team.
- Post-procedural fluid leak: An ascitic fluid leak is the most common post-paracentesis complication, and while is not immediately life threatening, it can lead to further complications. For this reason, the procedure site should be frequently reassessed, and bandages should be changed as necessary. If saturated bandages are left unchanged, skin breakdown may ensue, leading to ulcerations and an increased risk of cellulitis.7 Fluid leaks most often resolve as patients are further diuresed as an inpatient, although in extreme cases patients may require another paracentesis to resolve the leak. While it is not literature supported, a leak may be ceased by drying the procedure site and quickly applying tissue adhesive. This adhesive does not function well when placed on wet surfaces, and in order to improve success, a nasal cannula can be placed over the site to improve drying.
- Post-Paracentesis Circulatory Dysfunction: While not the most common complication, paracentesis induced circulatory is a potential product of large volume paracentesis (>5L).4,7 To understand why this theoretically happens, one must first review why ascites forms. The leading theory of ascitic fluid formation within the intraperitoneal space is due to an imbalance in oncotic and hydrostatic forces between the intravascular and interstitial spaces. This may occur due to a number of different reasons, but it is frequently seen in the setting of cirrhosis due to decrease protein production from the liver. Thus, when a large amount of intraperitoneal fluid is removed for symptomatic relief, the resulting fluid shifts, in conjunction with physiologic responses to those shifts, can lead to rapid fluid redistribution which in turn can result in accumulation of ascites and intravascular depletion. Rapid intravascular fluid shifts can cause a number of different electrolyte abnormalities, hypovolemic shock, and even flash pulmonary edema.4,7 So while this is an uncommon complication overall, how can it be avoided? The simplest solution is to remove <5L of ascitic fluid during a therapeutic tap, which has been shown to drastically reduce the risk of post-paracentesis circulatory dysfunction.7,12,13 In the exceedingly uncommon scenario where more than 5L needs to be removed in the ED, studies have shown that the prophylactic administration of 6-8g of IV 25% albumin per liter of ascites removed as a volume expander significantly decreases the risk of circulatory dysfunction.12,13 However, this is a rare circumstance in the emergency department and the average therapeutic paracentesis being performed by ED providers does not require albumin or any other volume expanders.
Case Query- Who needs a paracentesis?
As posed in the case above, does this patient require a diagnostic paracentesis, even though his symptoms might be explained by other etiologies? A large study in 2015 assessed over 17,000 patients with cirrhosis admitted for ascites and found in-hospital mortality was reduced by 24% when a paracentesis was performed.14It should be noted that this study labels early paracentesis as <1 day but does not differentiate paracentesis done by emergency providers versus the admitting physicians. While this study shows a clear benefit to patients with ascites having an early paracentesis done, it does not directly ascribe this benefit to it being done by emergency providers. This literature, in conjunction with a paracentesis being a simple, minimally invasive, relatively low-risk procedure, supports its performance and assessment of ascitic fluid in the ED upon admission of patient with ascites.14
Pearls and Pitfalls
- Make sure the patient is optimally positioned and comfortable prior to confirming the location of the fluid pocket and surrounding structures. If the patient moves significantly after this, the fluid pocket may shift, and the location of bowel relative to the fluid pocket may change.
- In order to assure adequate pain control, lidocaine should be injected down to the peritoneum. This can be confirmed by aspirating and injecting slowly until ascitic fluid is able to be aspirated at the end of the track created.
- If ascitic fluid ceases flowing prematurely, you can aspirate using negative pressure or attempt to redirect the catheter in case it is stuck against the omentum or abdominal wall.
- When performing a therapeutic paracentesis, no more than 5L should be drained without considering prophylactic albumin administration in order to avoid circulatory dysfunction. Removing >5L of fluid is rarely indicated in the ED.
- There are few true contraindications to performing a paracentesis. Unless one of these is present, it is generally best for any patient being admitted with ascites to have a paracentesis done upon admission unless it is completely unrelated to the ascites.
Rapid Procedure Review – Therapeutic Paracentesis
- Position patient supine with the bed elevated to 45 degrees
- Assess anatomical landmarks, locate a fluid pocket using ultrasound, and mark the skin
- Prepare paracentesis kit at bedside, ensuring all necessary equipment including collecting tubes and lidocaine are at the bedside
- Anesthetize down to the peritoneum
- Using the “Z-track” method, insert needle and catheter into fluid pocket
- Once fluid is aspirated, advance catheter and retract needle.
- Aspirate fluid for laboratory analysis
- Attach three-way stopcock and drainage tubing, and begin therapeutic fluid removal
- Once adequate fluid has been drained, remove catheter, apply pressure, and bandage the procedure site
Rapid Procedure Review – Rapid Diagnostic Paracentesis
- Position patient supine with the bed elevated to 45 degrees
- Assess anatomical landmarks, locate a fluid pocket using ultrasound, and mark the skin
- Anesthetize down to the peritoneum
- Using the “Z-track” method, insert a 1.8-2” angiocatheter (20g or less) into the fluid pocket
- Once ascitic fluid “flash” is obtained, advance the angiocatheter and then remove needle
- Collect ascitic fluid specimen
- Once adequate specimen has been obtained, remove the angiocatheter, apply pressure, and bandage the procedure site
References/Further Reading
- Ginès, Pere, et al. “Management of Cirrhosis and Ascites.” New England Journal of Medicine, vol. 350, no. 16, 2004, pp. 1646–1654., doi:10.1056/nejmra035021.
- Walls, Ron M., et al. Rosen’s Emergency Medicine: Concepts and Clinical Practice. Elsevier, 2018.
- Runyon, Bruce A. “Management of Adult Patients with Ascites Due to Cirrhosis.” Hepatology, vol. 39, no. 3, 2004, pp. 841–856., doi:10.1002/hep.20066.
- Wilkerson, R. Gentry, and Richard Sinert. “The Use of Paracentesis in the Assessment of the Patient With Ascites.” Annals of Emergency Medicine, vol. 54, no. 3, 2009, pp. 465–468., doi:10.1016/j.annemergmed.2008.09.005.
- Grabau, Catherine M., et al. “Performance Standards for Therapeutic Abdominal Paracentesis.” Hepatology, vol. 40, no. 2, 2004, pp. 484–488., doi:10.1002/hep.20317.
- Gaetano, John N, et al. “The Benefit of Paracentesis on Hospitalized Adults with Cirrhosis and Ascites.” Journal of Gastroenterology and Hepatology, vol. 31, no. 5, 2016, pp.
- Gottardi, Andrea De, et al. “Risk of Complications After Abdominal Paracentesis in Cirrhotic Patients: A Prospective Study.” Clinical Gastroenterology and Hepatology, vol. 7, no. 8, 2009, pp. 906–909., doi:10.1016/j.cgh.2009.05.004.
- Sharzehi, Kaveh, et al. “Hemorrhagic Complications of Paracentesis: A Systematic Review of the Literature.” Gastroenterology Research and Practice, vol. 2014, 2014, pp. 1–6., doi:10.1155/2014/985141.
- Nazeer, Shameem R., et al. “Ultrasound-Assisted Paracentesis Performed by Emergency Physicians vs the Traditional Technique: a Prospective, Randomized Study.” The American Journal of Emergency Medicine, vol. 23, no. 3, 2005, pp. 363–367., doi:10.1016/j.ajem.2004.11.001.
- Shriver, Amy, et al. “A Randomized Controlled Trial of Procedural Techniques for Large Volume Paracentesis.” Annals of Hepatology, vol. 16, no. 2, 2017.
- Sakai, Hideki, et al. “Choosing the Location for Non-Image Guided Abdominal Paracentesis.” Liver International, vol. 25, no. 5, 2005, pp. 984–986., doi:10.1111/j.1478-3231.2005.01149.x.
- Tan, Hiang Keat, et al. “Albumin May Prevent the Morbidity of Paracentesis-Induced Circulatory Dysfunction in Cirrhosis and Refractory Ascites: A Pilot Study.” Digestive Diseases and Sciences, vol. 61, no. 10, 2016, pp. 3084–3092., doi:10.1007/s10620-016-4140-3.
- Bernardi, Mauro, et al. “Does the Evidence Support a Survival Benefit of Albumin Infusion in Patients with Cirrhosis Undergoing Large-Volume Paracentesis?” Expert Review of Gastroenterology & Hepatology, 2016, pp. 1–2., doi:10.1080/17474124.2017.1275961.
- Kim, John J, et al. “Delayed Paracentesis Is Associated With Increased In-Hospital Mortality in Patients With Spontaneous Bacterial Peritonitis.” The American Journal of Gastroenterology, vol. 109, no. 9, 2014, pp. 1436–1442., doi:10.1038/ajg.2014.212.







3 thoughts on “Unlocking Common ED Procedures – Pocket Full of Sunshine: Paracentesis in the ED”
Pingback: Ascites – Översikt – Mind palace of an ER doc
Pingback: October ’19 Asynchronous Learning – Lakeland Health EM Blog
Pingback: Liver Emergencies: Thrombosis, Bleeding, SBP, portal vein thrombosis, paracentesis | EM Cases