Welcome back to the “52 in 52” series. This collection of posts features recently published must-know articles. Post #22 looks at vasopressin + methylprednisolone for in-hospital cardiac arrest.

Author: Brannon Inman (Chief Resident, Emergency Medicine Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Effect of Vasopressin and Methylprednisolone vs Placebo on Return
of Spontaneous Circulation in Patients With In-Hospital Cardiac Arrest:
A Randomized Clinical Trial
Question:
Does use of vasopressin + methylprednisolone (VAM) result in improved ROSC in patients with in-hospital cardiac arrest (IHCA)
Design: Multi-center, block randomized, double-blind, placebo-controlled trial
PICO
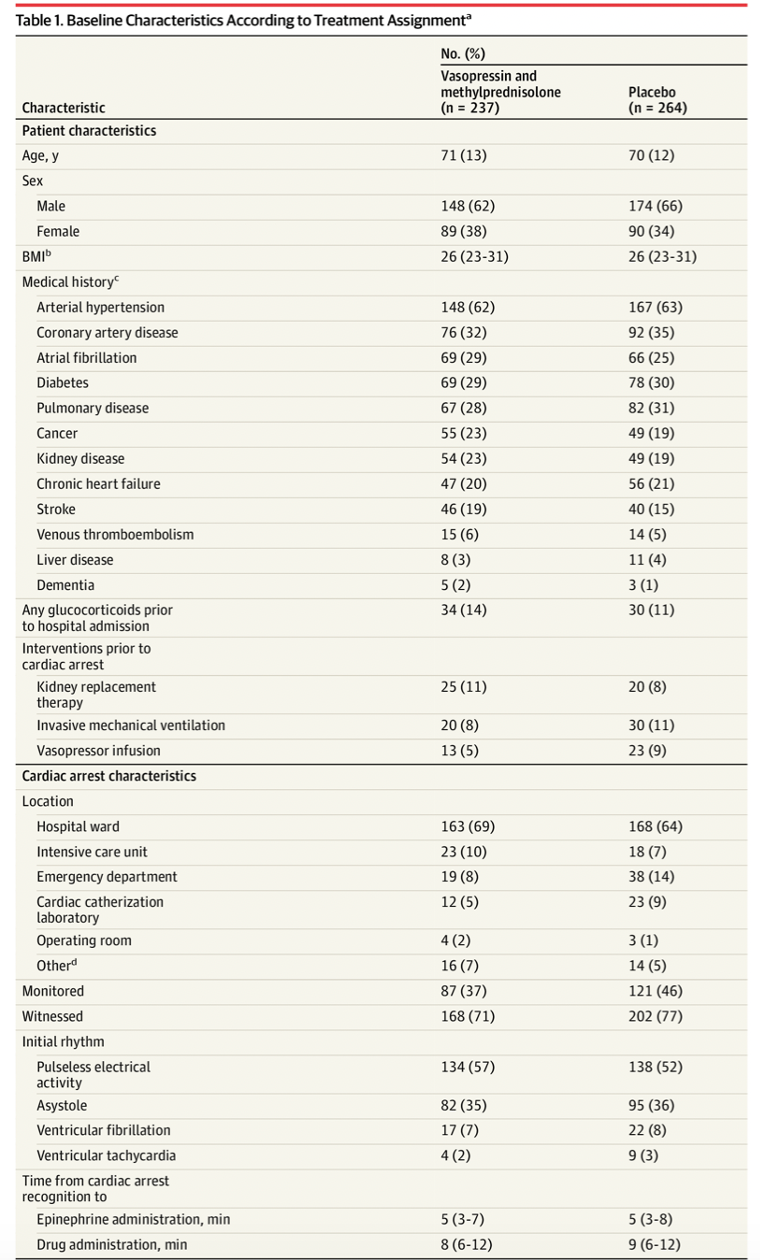
Population:
- Enrolled patients in 10 hospitals in Denmark
- Inclusion
- Age > 10 years
- Patients with in-hospital cardiac arrest (ER patients were included in this)
- Must have been given 1 dose of epinephrine
- Exclusion
- A valid DNR
- Was previously enrolled in this trial
- Invasive circulatory support (ECMO, Impella, etc.)
- Known/suspected pregnancy

Intervention:
- 40 mg methylprednisolone + 20 IU vasopressin immediately following the first dose of epinephrine
- Vasopressin repeated for up to 4 total doses
Comparator:
- An equal volume of normal saline
Outcome:
- Primary outcome ROSC
- 42% in the intervention arm vs 33% in the control arm
- RR 1.30 (95% CI 1.03-1.63)
- Fragility index (FI) = 3
- Secondary outcome: No difference in 30 or 90 day survival or favorable neurologic outcome
- No significant difference in safety outcomes on hyperglycemia, hypernatremia, GI bleeding.


Take Aways:
- This trial suggests that patients with IHCA who receive vasopressin and methylprednisolone have improved likelihood of ROSC.
- However, the rate of survivors with a good neurologic outcome was no different between the groups. When looking at other trials (i.e., PARAMEDIC 2) epinephrine alone demonstrated increased ROSC with increased rates of poor neurologic outcome.
- Two preceding trials suggest improved survival with good neurologic outcomes in those receiving vasopressin and methylprednisolone in the setting of IHCA.2,3
- There were several notable limitations to this 2021 study.
- A significant number of patients were excluded “for other reasons”. 193 patients were excluded because the “team forgot”. 170 patients were excluded due to “physician preference”.
- This study only included patients in one country (Denmark). This limits external validity.
- Higher rates of extracorporeal support were noted in the placebo group. This may suggest a failure of randomization as typically sicker patients receive this therapy.
- Overall there were low rates of TTM (27% in intervention, 26% in control).
- The study was powered for a ROSC rate of 45%, but they only obtained a ROSC rate of 33%. This can bias the data away from the null.
- These are fragile data (FI = 3) displaying a benefit of VAM in IHCA. These data add to a contingent of studies suggesting a benefit of VAM in IHCA.
My Take:
These are relatively fragile data supporting the use of VAM in resuscitation in IHCA. At the individual level, I will keep this in my back pocket when it comes to “pulling out all the stops”. Based on current guidelines I will not use this as a routine. However, at the big table, we need to be having discussions about the inclusion of VAM in standard resuscitation of IHCA.
References:
- Andersen LW, Isbye D, Kjærgaard J, et al. Effect of Vasopressin and Methylprednisolone vs Placebo on Return of Spontaneous Circulation in Patients With In-Hospital Cardiac Arrest: A Randomized Clinical Trial.JAMA. 2021;326(16):1586-1594. doi:10.1001/jama.2021.16628
- Mentzelopoulos SD, Malachias S, Chamos C, et al. Vasopressin, steroids, and epinephrine and neurologically favorable survival after in-hospital cardiac arrest: a randomized clinical trial.JAMA. 2013;310(3):270-279. doi:10.1001/jama.2013.7832
- Mentzelopoulos SD, Zakynthinos SG, Tzoufi M, et al. Vasopressin, epinephrine, and corticosteroids for in-hospital cardiac arrest.Arch Intern Med. 2009;169(1):15-24. doi:10.1001/archinternmed.2008.509








