Welcome back to the “52 in 52” series. This collection of posts features recently published must-know articles. Post #25 looks at risk of major ischemic or hemorrhagic event with rivaroxaban versus apixaban.

Author: Brannon Inman (Chief Resident, Emergency Medicine Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); and Brit Long, MD (@long_brit)
Association of Rivaroxaban vs. Apixaban With Major Ischemic or Hemorrhagic Events in Patients With Atrial Fibrillation
Question:
Does rivaroxaban compared to apixaban have a stronger association with major ischemic and hemorrhagic events?
Study design: Retrospective registry cohort study
PICO
Population:
- Inclusion criteria
- Age ≥ 65 years old
- Medicare Parts A, B and D insurance coverage
- Participants with complete demographic information
- Patients had to have a prescription for apixaban or rivaroxaban at either:
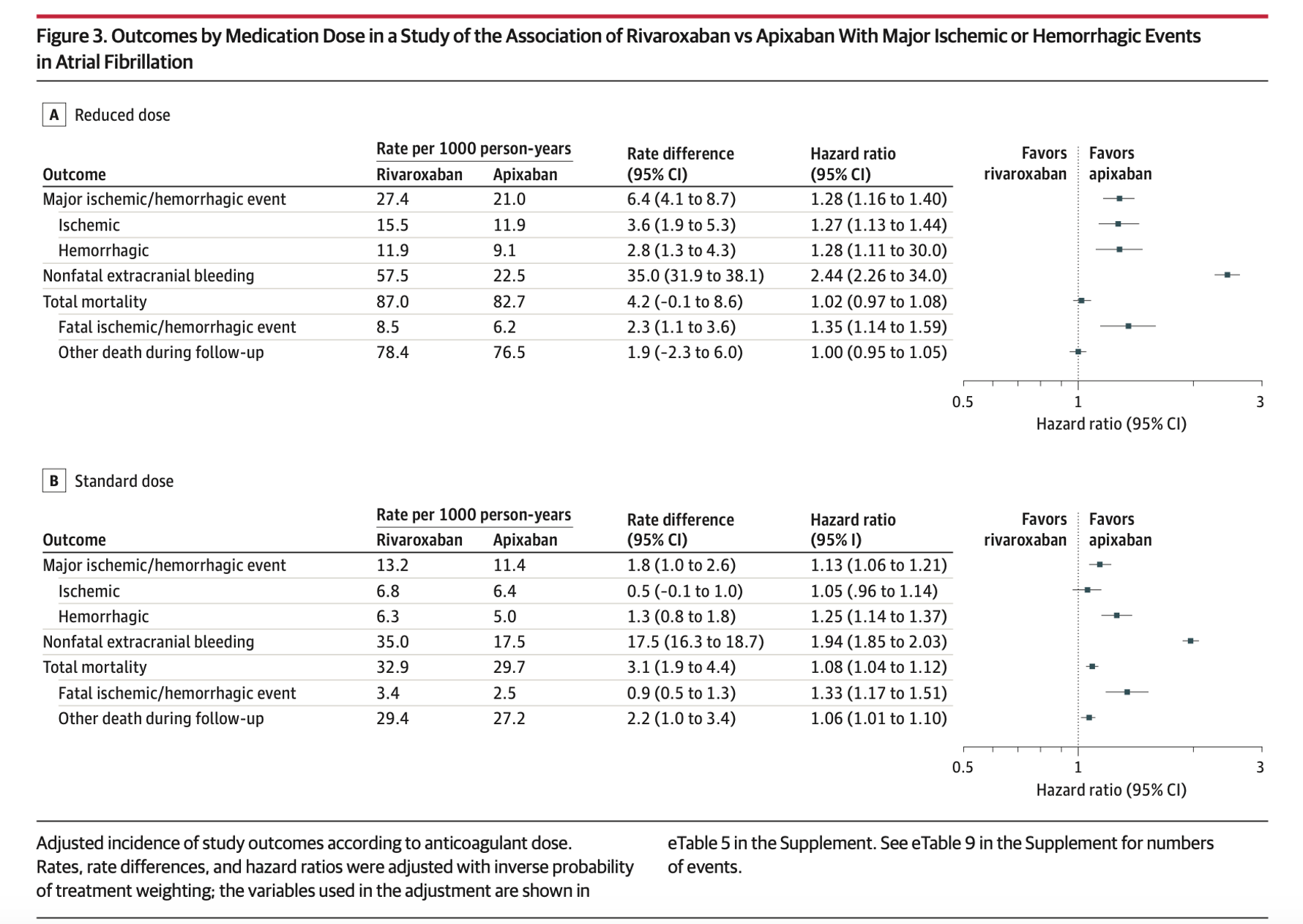
- The standard dose (5 mg twice daily for apixaban and 20 mg once daily for rivaroxaban)
- The reduced dose (2.5 mg twice daily for apixaban and 15 mg once daily for rivaroxaban) for patients with factors likely to in- crease plasma concentrations.
- The medication was prescribed for atrial fibrillation
- The medication was prescribed between January 1, 2013 and November 30, 2018
- During the preceding year, patients had to have continuous enrollment in Medicare and,
- Patients had to have at least 1 outpatient visit and 1 prescription filled (other than the drug being studied)
- Patients had to be new users of an outpatient oral anticoagulant
- Exclusion criteria
- Medicare Advantage Plan (Part C) insurance coverage (cited as having less reliable extractable encounter data)
- Terminal illness
- Long-term care facility residency ≥ 30 days
- Mitral valve stenosis
- Chronic kidney disease stage 4 or 5 or end-stage (ESRD)
- Mechanical Heart valve (bovine or other bioprosthetic heart valves were enrolled)
- Any oral anticoagulant use as an outpatient in the preceding year.
- A stroke or hospitalization for bleeding the preceding 30 days

- Patients were followed for 4 years (starting the day after the prescription for the anticoagulant was filled)
- Follow-up of the patient ended under the following circumstances:
- A gap of > 30 days was documented (based on prescription filling dates), as this was considered medication discontinuation
- The patient had a change in anticoagulant dose
- Patient developed stage 4, 5, or end-stage chronic kidney disease
- Loss of full Medicare enrollment
- Occurrence of study outcome or death
- The study period ended (Nov 30, 2018) or the patient completed 4 years of anticoagulation
- Follow-up of the patient ended under the following circumstances:
Intervention and Comparator:
- Rivaroxaban vs. apixaban
Outcome:
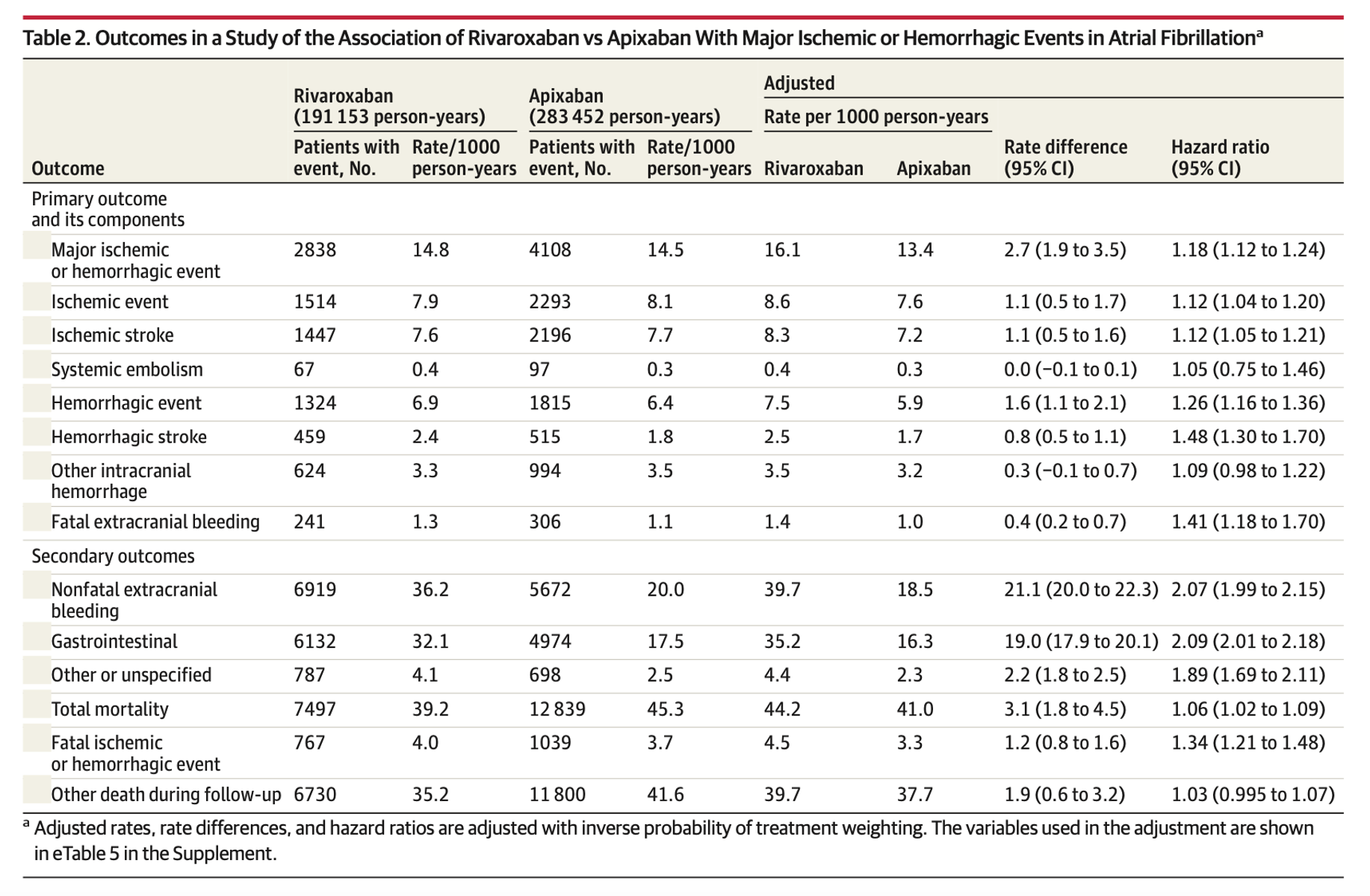
- Rate of major ischemic and hemorrhagic events in the rivaroxaban/apixaban groups
- 1 vs 13.4 per 1000 person-years
- Rate difference, 2.7 [95% CI, 1.9- 3.5] – significant
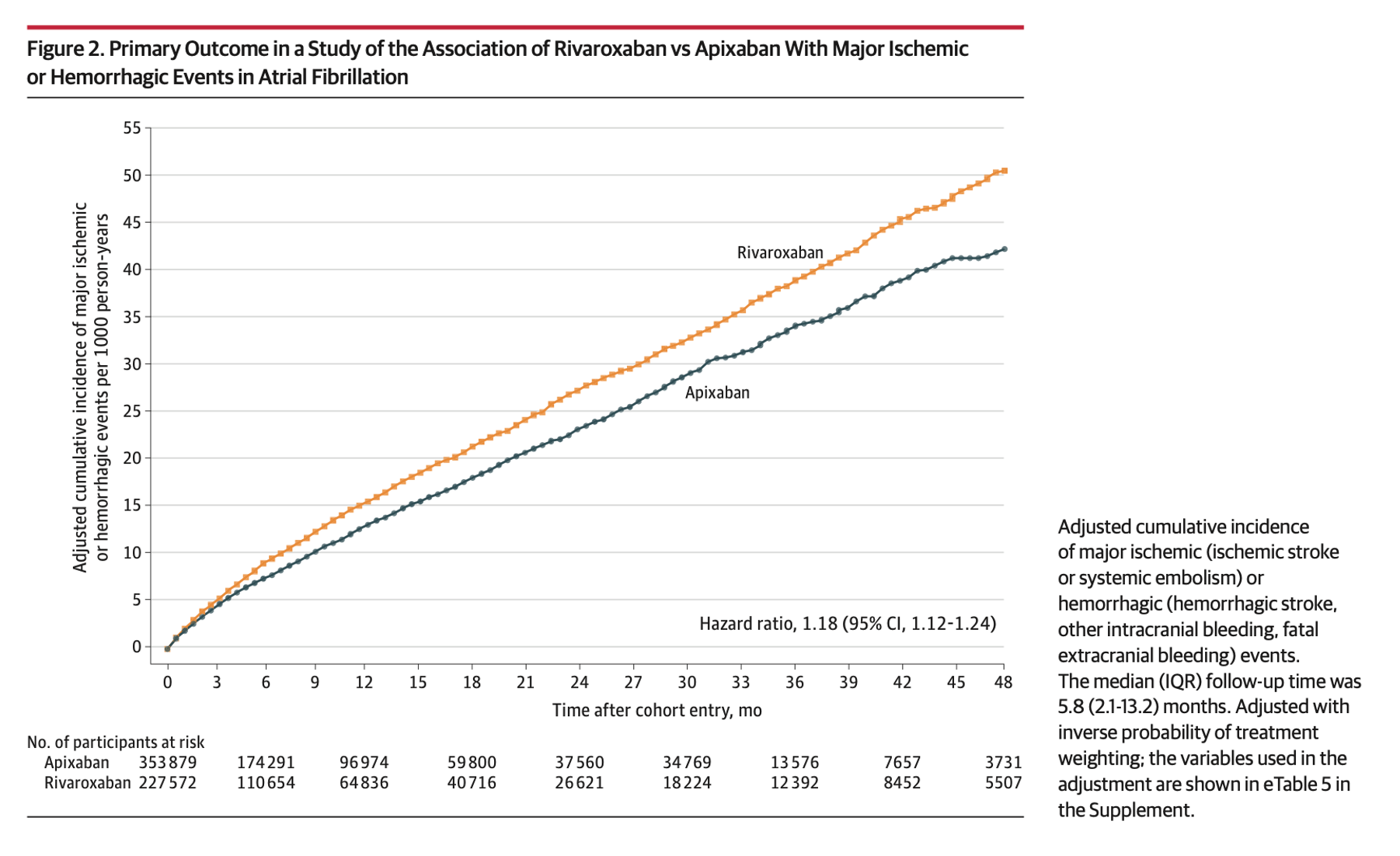
- Hazard ratio, 1.18 [95% CI, 1.12-1.24] – significant
- Major ischemic events:Aadjusted rate, 6 vs 7.6 per 1000 person-years
- Rate difference, 1.1 [95% CI, 0.5-1.7] – significant
- Hazard ratio, 1.12 [95% CI, 1.04-1.20] – significant
- Major Hemorrhagic events
- Adjusted rate, 5 vs 5.9 per 1000 person-years
- Rate difference, 1.6 [95% CI, 1.1-2.1] – significant
- Hazard ratio, 1.26 [95% CI, 1.16-1.36] – significant
- Ischemic stroke
- Adjusted rate, 3 vs 7.2 per 1000 person-years
- Rate difference, 1.1 [95% CI, 0.5 – 1.6] – significant
- Hazard ratio, [95% CI, 1.16-1.36] – significant
- Hemorrhagic stroke
- Adjusted rate, 5 vs 1.7 per 1000 person-years
- Rate difference, 0.8 [95% CI, 0.5-1.1] – significant
- Hazard ratio, 1.48 [95% CI, 1.30-1.70] – significant
- Fatal non-intracranial bleeding
- Adjusted rate, 4 vs 1.0 per 1000 person-years
- Rate difference, 0.4 [95% CI, 0.2-0.7] – significant
- Hazard ratio, 1.41 [95% CI, 1.18-1.70] – significant
Take away:
- Statistically higher odds of ischemic and hemorrhagic events in the rivaroxaban group compared to apixaban.
- Overall, this was an interesting study comparing 2 of the most popular direct oral anticoagulants currently used in the United States.
- It’s not very often you get to see studies comparing one brand name drug to another. Even more interesting is this study was funded by a grant from the national heart lung and blood institute (NHLBI) rather than one of the pharmaceutical companies of interest.
- While the authors utilized a relatively convenient sample of registry patients, this was a large study with hundreds of thousands of patients in each group.
- While the methods were highly selective, they were well outlined in the study.
- There were more patients in the apixaban group than the rivaroxaban group. This resulted in the data having to be adjusted to patient years in order to make meaningful interpretations from the data. With > 100,000 patients in each group, it’s less likely observations are due to chance.
- These are still observational data. The reasons for clinician choice regarding one medication over the other was not captured in this study.
- I would love to see a randomized controlled trial. However, with another registry trial on the horizon looking at the recurrence of DVTs, this will likely be the highest quality evidence we receive on this subject.
My Take:
- Based on these data, when I need to send a patient with atrial fibrillation home on direct oral anticoagulation, it will probably be apixaban (assuming it is financially viable, and the BID dosing is realistic for the patient in question).
References:
- Ray WA, Chung CP, Stein CM, et al. Association of Rivaroxaban vs Apixaban With Major Ischemic or Hemorrhagic Events in Patients With Atrial Fibrillation [published correction appears in JAMA. 2022 Apr 5;327(13):1294]. JAMA. 2021;326(23):2395-2404. doi:10.1001/jama.2021.21222






