
Author: Christiaan van Nispen, MD (Resident, Emergency Medicine Physician, San Antonio, TX); Brannon Inman, MD (Chief Resident, Emergency Medicine Physician, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Thrombolysis Guided by Perfusion Imaging up to 9 Hours after Onset of Stroke
AKA: The “EXTEND” Trial
Clinical question:
Amongst patients presenting with acute ischemic stroke between 4.5 and 9 hours after onset of symptoms, with non-infarcted brain tissue, does the administration of alteplase improve functional outcome at 90 days?
Design: Multicentered, double-blinded, randomized controlled trial
PICO:
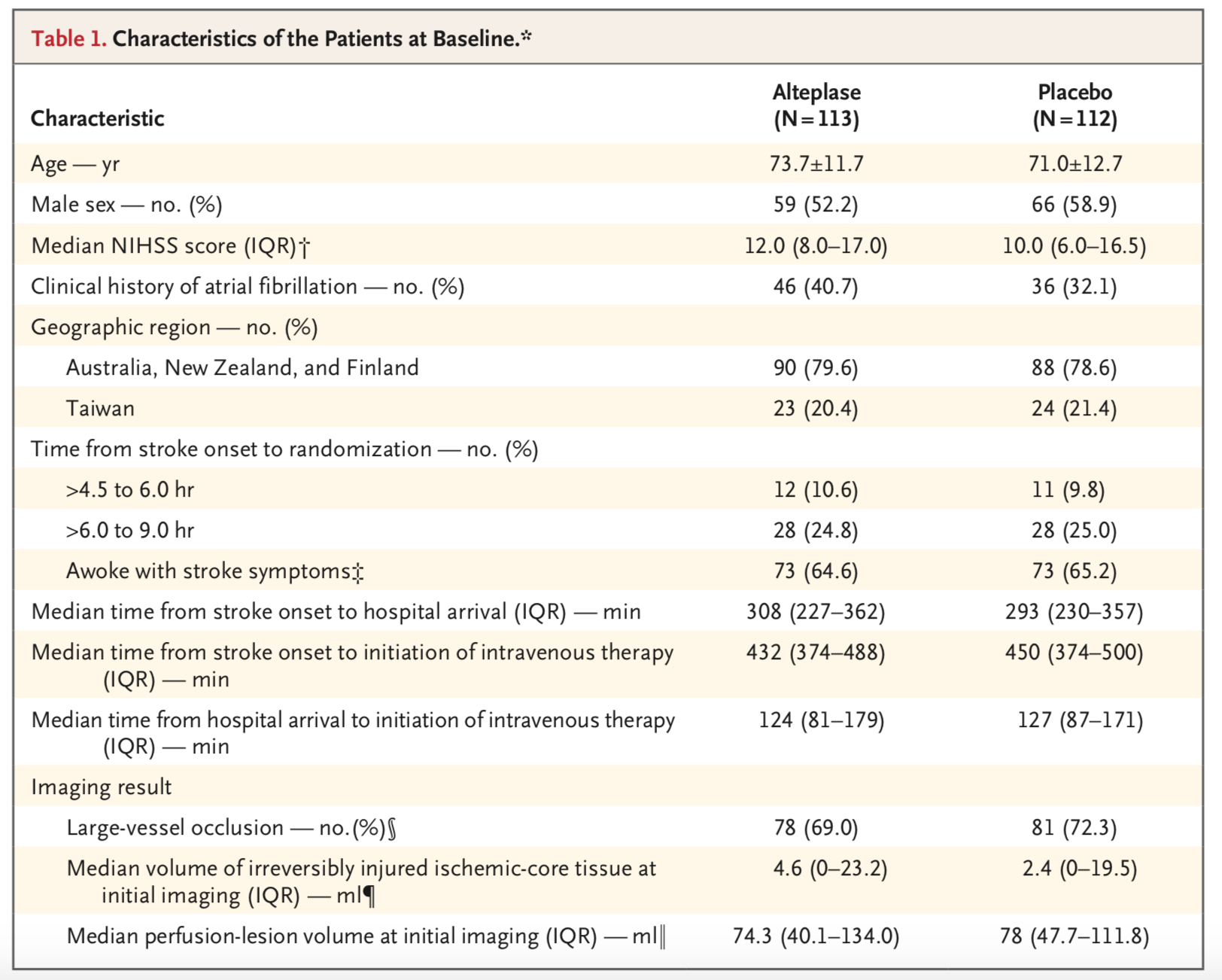
Population:
- Inclusion Criteria:
- 18 years of age or older
- Baseline modified Rankin Scale (mRS) of < 2
- Presented between 4.5 and 9 hours of symptom onset; stroke symptoms that started during sleep were assumed to have started halfway between the actual last known well and the time of wake-up.
- NIH Stroke Scale (NIHSS) of 4 to 26
- Hypo-perfused but non-infarcted region of brain detected on advanced perfusion imaging (CT perfusion imaging or perfusion-diffusion MRI, depending on the hospital center)
- Exclusion Criteria:
- Rapidly improving symptoms
- Hemorrhagic stroke
- Stroke within last 3 months
- Infarct core greater than one third of the middle cerebral artery territory
- Current use of anticoagulants, glycoprotein IIb/IIIa inhibitors
- Blood pressure > 185/110 refractory to antihypertensive therapy
- Patient under consideration for endovascular thrombectomy
- Enrollment
- 16 centers in Australia, 10 centers in Taiwan, 1 center in New Zealand, and 1 center in Finland.
- Pre-enrollment calculations estimated that 400 enrollments would be necessary to provide the trial with 80% power to detect a 15% difference in the primary outcome, allowing for up to 90 patients to be lost to follow-up or to have insufficient data documented.
- Subsequently reduced enrollment to 310 patients because no patients had been lost to follow-up or had insufficient data documented.
- Trial discontinued prematurely by data and safety monitoring board due to “loss of equipoise,” citing positive findings in the trial “Efficacy and Safety of MRI-Based Thrombolysis in Wake-Up Stroke (the WAKE-UP trial)”
- Ultimately, 225 patients were enrolled from August 2010 through June 2018.
Intervention:
- Alteplase 0.9 mg/kg body weight (maximum 90 mg) administered intravenously as a 10% bolus followed by 90% infusion over 1 hour
Comparator:
- Infusion of a matching placebo
Outcome:
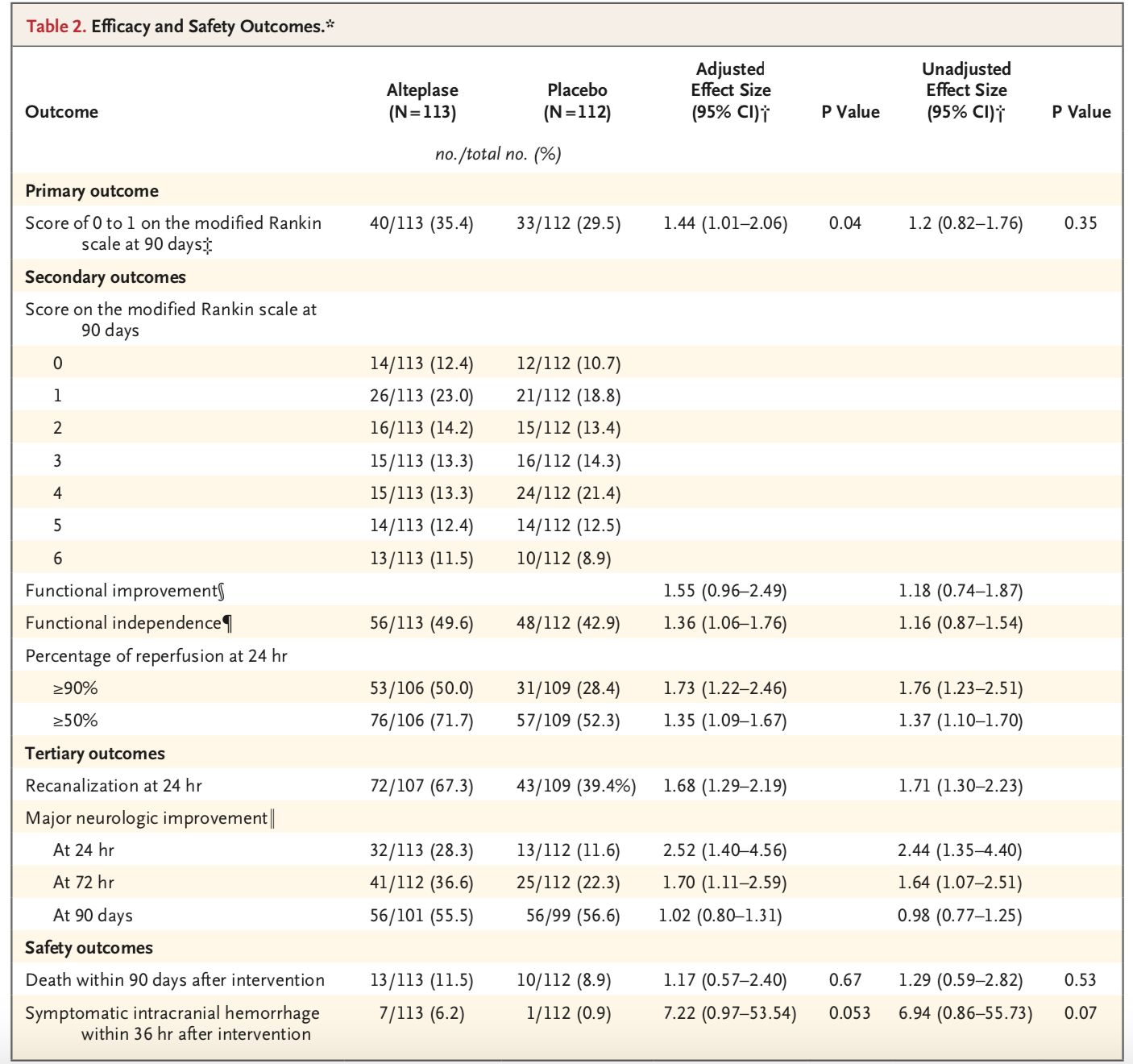
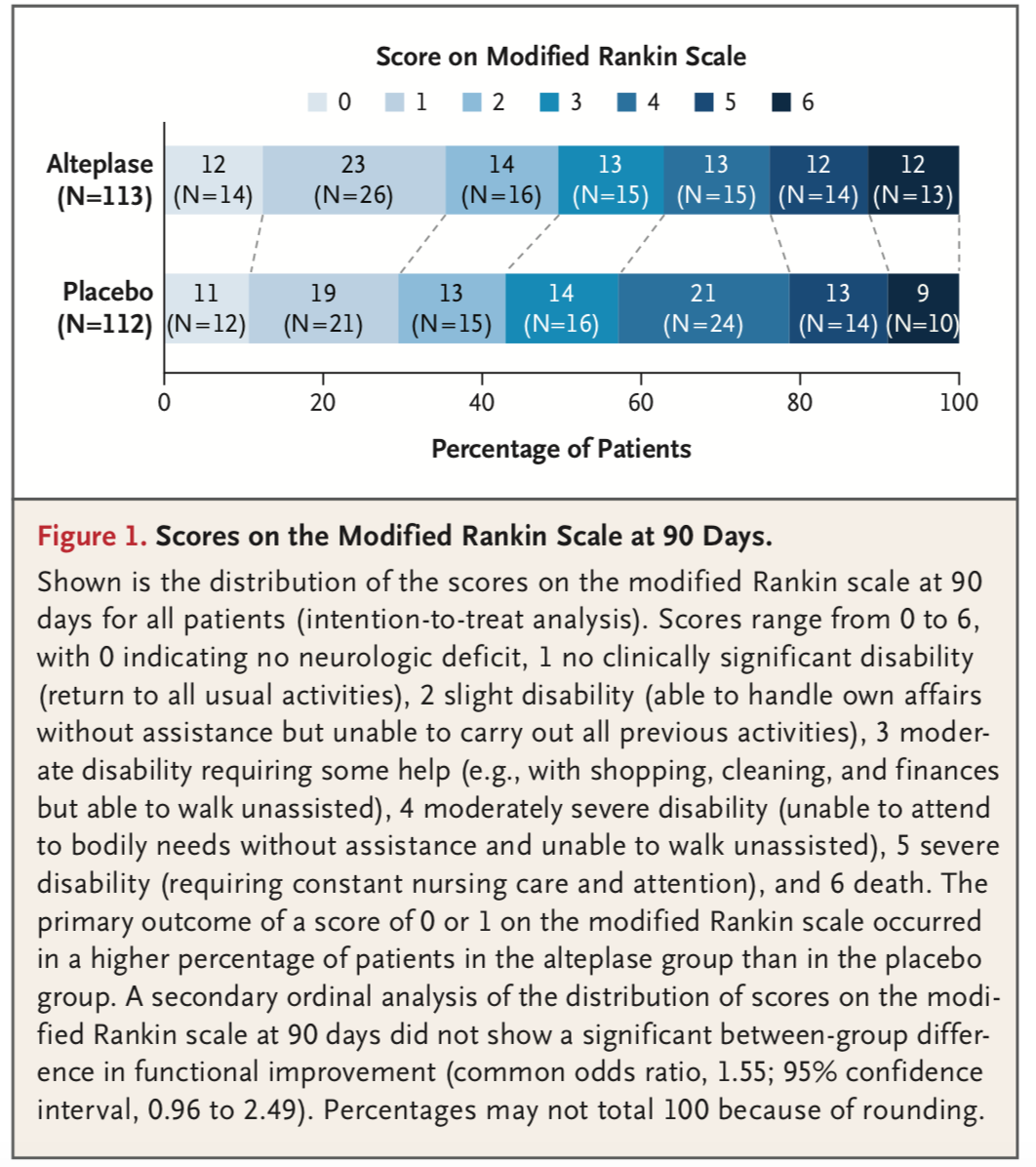
- Primary Outcome: mRS of 0 or 1 at 90 days
- Alteplase: 40 of 113 (35.4%)
- Placebo: 33 of 112 (29.5%)
- These unadjusted results did not achieve statistical significance (Effect Size 1.2, 95% CI 0.82-1.76, p=0.35)
- Authors then adjusted for baseline mRS to calculate an effect size of 1.44, achieving statistical significance (95% CI 1.01 2.06, p=0.04)
- Secondary and tertiary outcomes with significant difference in favor of alteplase:
- Functional independence at 90 days (defined by mRS of 0 through 2) when adjusting for baseline mRS, but not in unadjusted results
- Percentage of tissue reperfusion at 24 hours, in both adjusted and unadjusted results
- Recanalization at 24 hours, in both adjusted and unadjusted results
- Major neurological improvement (defined as improvement in NIHSS of 8 or greater) at 24 and 72 hours, in both adjusted and unadjusted results
- Secondary and tertiary outcomes with no significant differences:
- Ordinal score on the mRS
- Major neurological improvement at 90 days
- Death within 90 days
- Symptomatic intracranial hemorrhage within 36 hours of intervention (although this was quite close to achieving statistical significance; 6.2% in alteplase group versus 0.9% in placebo group, with adjusted effect size 7.22 (95% CI 0.97 to 53.54, p=0.053).
Take Aways:
- The signal toward alteplase administration producing a mRS of 0 or 1 at 90 days is small, and only appreciated when adjusting for baseline mRS, but nonetheless achieved statistical significance.
- Strengths of this article include it being a multicenter, double blinded, randomized controlled trial with intention to treat analysis and no loss to follow-up.
- There are several key weaknesses:
- This trial was discontinued prior to full enrollment due to positive results in favor of alteplase in the WAKE-UP Trial; however, the inclusion criteria for that study had important differences and it had a fragility index of 7. More research is needed on this topic, yet the authors of the EXTEND trial discontinued enrollment.
- That the authors frequently adjusted for baseline mRS in order to declare their trial positive is an intriguing decision given that random assignment into groups should have produced similar baseline group characteristics. Of note, the authors declared that their groups were well balanced, but they do not report on the baseline mRS of each group.
- Without explanation, the authors changed the statistical analytic plan from a binary logistic regression to Poisson regression; had they used binary logistic regression, the trial would be negative.
- Analyses were not adjusted for multiple comparisons.
- Declaring the midpoint between last known well and time of awakening with stroke-like symptoms as the time of onset for the stroke is arbitrary; data from patients who woke up with stroke-like symptoms were combined with data from patients with a specifically known time of onset.
My take:
- More date are needed prior to the routine administration of thrombolytic agents in patients presenting between 4.5 and 9 hours after stroke onset, as well as their use in patients who awaken with stroke-like symptoms.
Reference:
Ma H, Campbell BC, Parsons MW, et al. Thrombolysis Guided by Perfusion Imaging up to 9 Hours after Onset of Stroke. NEJM.2019; 380:1795-1803. DOI: 10.1056/NEJMoa1813046
Appendix:
Modified Rankin Scale
- No symptoms at all
- No significant disability despite symptoms; able to carry out all usual duties and activities.
- Slight disability; unable to carry out all previous activities, but able to look after own affairs without assistance.
- Moderate disability; requiring some help, but able to walk without assistance.
- Moderately severe disability; unable to walk and attend to bodily needs without assistance.
- Severe disability; bedridden, incontinent, and requiring constant nursing care and attention.





