Authors: Andrew Pirotte, MD (Department of Emergency Medicine, Lawrence Memorial Hospital, Lawrence, Kansas), Jacquelyn Wagner, MS2 (University of Kansas School of Medicine, Kansas City, Kansas), Matthew Pirotte, MD (Northwestern Memorial Hospital, Department of Emergency Medicine, Chicago, Illinois) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit, EM Attending Physician at SAUSHEC)
A 68-year-old male with history of diabetes mellitus, hypertension, and hyperlipidemia presents to the emergency department with a chief complaint of a severe right upper extremity (RUE) pain, hives, and diffuse erythema. He reports a possible insect bite after donning a sweater. He had initially visited an urgent care facility where he was diagnosed with cellulitis and prescribed doxycycline. His pain increased, and he developed a diffuse erythematous and urticarial rash, which led to his visit to the emergency department.
Initial vital signs include temperature 100.4 F (oral), heart rate 109, blood pressure 145/86, respirations 18, and pulse oximetry 95% on room air. On physical examination, the patient has diffuse hives and erythema particularly over trunk and extremities without mucosal involvement. The right upper extremity has an 8 cm area of induration, mottling, and tenderness to palpation just distal to the axilla. The neurovascular exam of the affected extremity is intact.
The initial differential diagnosis included spider bite with a consideration of brown recluse envenomation (Loxosceles reclusa), worsening cellulitis, necrotizing skin and soft tissue infection, and allergic reaction. The decision was made to monitor the patient in ED and draw labs for further evaluation. Diphenhydramine 50 mg PO x 1 was provided to treat possible allergic reaction from the doxycycline.
Labs return with the following: Hg 15.2, WBC 9.8, Plt 144, Chem 135/95/17/3.5/29/1.3, Gluc 146, and Lactic Acid 2.4
Upon reassessment, the hives and erythema had significantly improved, but he continued to complain of significant pain in his right upper extremity. Admission was considered, though ultimately the patient was discharged home given improving clinical status, near-total resolution of hives, and non-toxic appearance. As brown recluse bite was the leading diagnosis, complicated by likely allergic reaction to doxycycline, this antibiotic was discontinued, and the patient was started on antihistamines and analgesia (hydrocodone-acetaminophen 5-325 mg po q4-6 prn pain). Steroids were withheld given the patient’s history of diabetes mellitus.
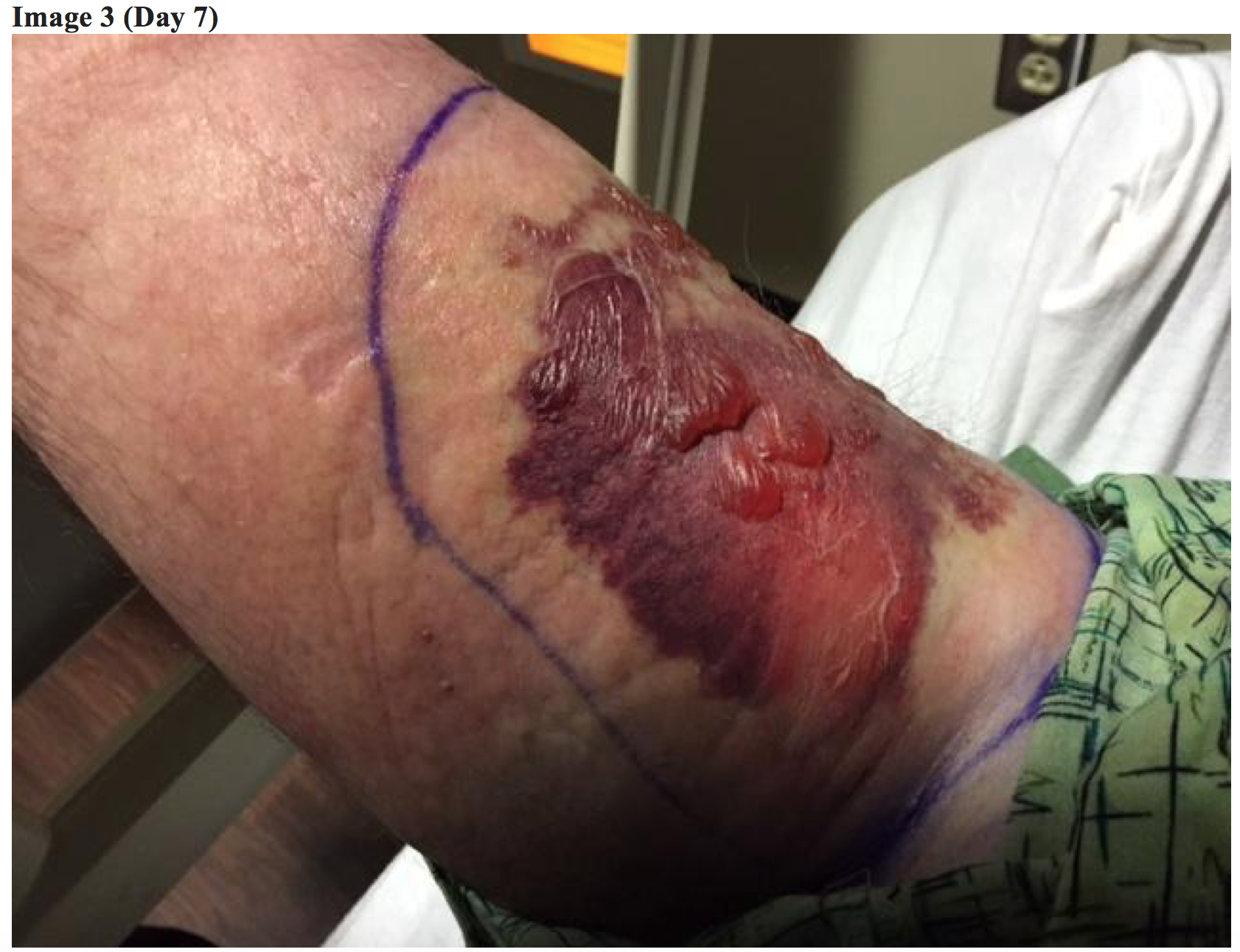
The following day, the patient returned to the ED with significant progression of swelling, pain, and redness. The patient reported that he now felt systemically ill. His repeat vitals were temperature 101.2 F (oral), heart rate 90, blood pressure 120/53, respirations 16, and pulse oximetry 98% on room air. His hives and erythema had resolved. His right upper extremity exam now demonstrated deepening of the previously noted mottling with two punctate puncture wounds centrally located suggestive of a bite. The surrounding tissue was edematous and ecchymotic with approximately 15 centimeter diameter of involved tissue. The skin was warm to touch and markedly tender to palpation. No crepitus was noted. An intravenous line was placed, IV fluids initiated, and labs were redrawn.
Repeat lab results included Hg 14.2 WBC 9.8, Plt 141, Chem 136/3.2/95/29/17/1.3/142, Lactic acid 2.1.
After IV fluids, patient continued to feel ill and his pain persisted despite IV analgesia. Clinical history, laboratory results, and physical exam favored brown recluse bite (necrotic cutaneous loxoscelism) over cellulitis or necrotizing fasciitis, though his disposition was guarded. The patient was admitted for further monitoring, pain control, serial laboratory draws, and general surgery evaluation. Overnight the patient’s fever persisted despite antipyresis, and vancomycin was initiated.
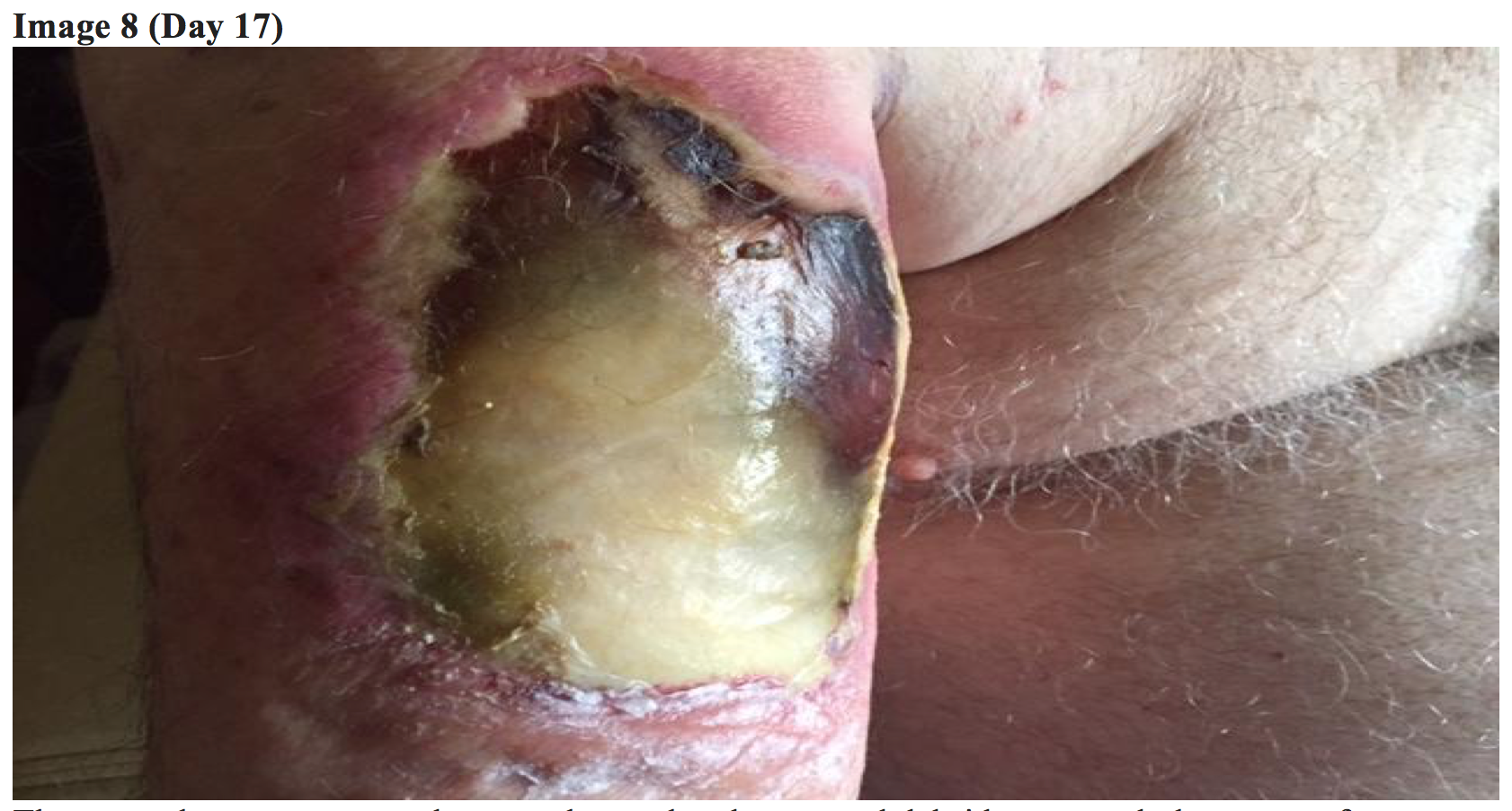
 Through his subsequent 12-day hospitalization, the patient’s RUE lesion evolved significantly and caused persistent and severe pain requiring morphine patient-controlled analgesia (PCA). Infectious disease and general surgery teams were consulted and recommended continued antibiotics and elevation.
Through his subsequent 12-day hospitalization, the patient’s RUE lesion evolved significantly and caused persistent and severe pain requiring morphine patient-controlled analgesia (PCA). Infectious disease and general surgery teams were consulted and recommended continued antibiotics and elevation.
Ultimately the patient was discharged on hospital day 13 with oral narcotics, amoxicillin, and silvadene, with general surgery follow up for wound care.
After discharge from the hospital the patient continued to experience persistent, severe pain. His wound morphology continued to evolve:
The general surgery team subsequently escalated to wound debridement and placement of a commercial wound suction 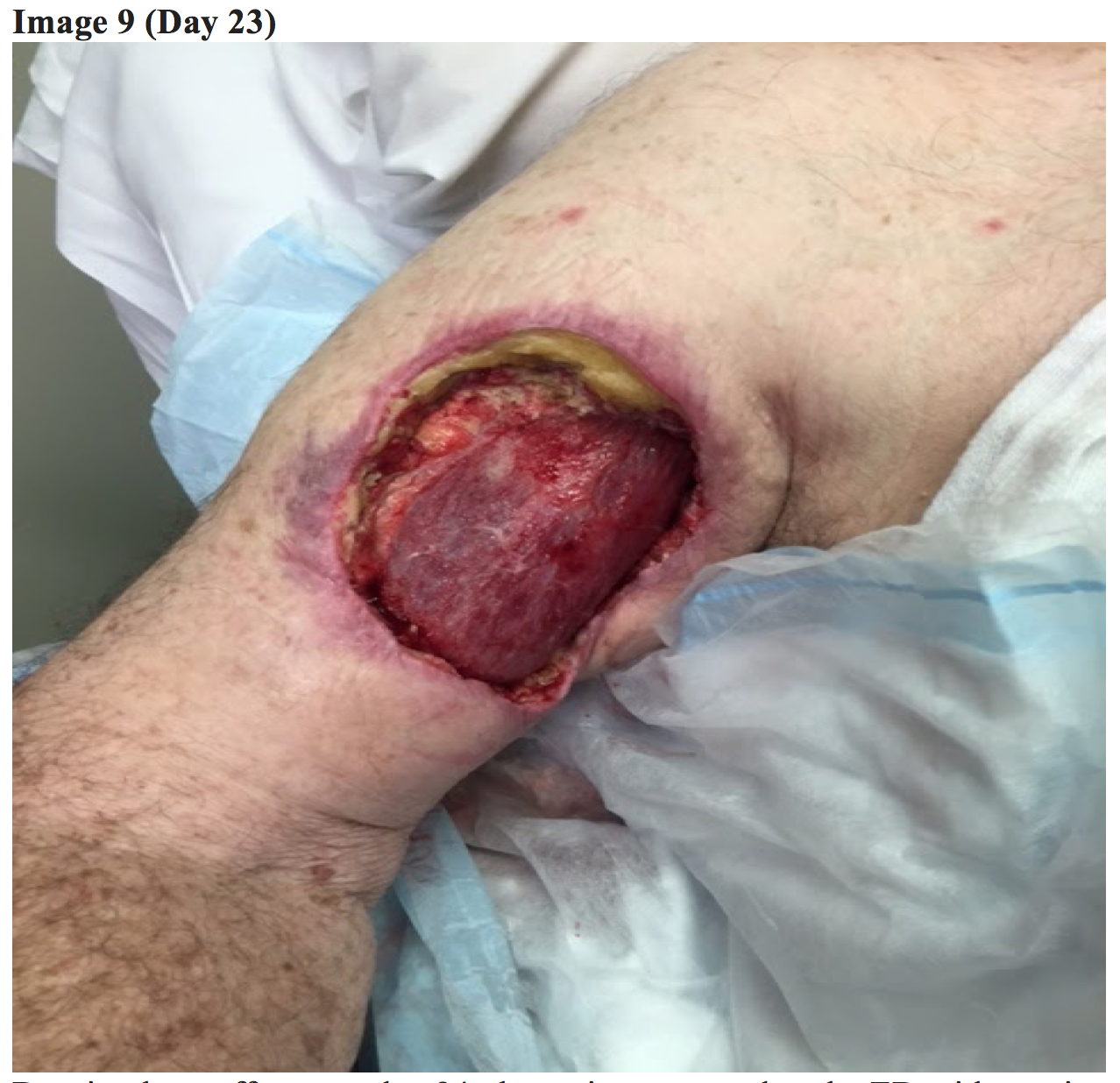 dressing (Day 23).
dressing (Day 23).
Despite these efforts, on day 24, the patient returned to the ED with continued severe RUE pain, in addition to vomiting and right upper quadrant abdominal pain.
Vitals were as follows: Temperature 100.4 F, heart rate 97, blood pressure 148/71, respirations 20, and pulse oximetry 91% on room air.
Lab results returned with Hg 12.9, WBC 10.3, Plt 222; Chem: 138/3.2/97/25/18/0.9, Gluc 135; Lipase 1252
AST 96, ALT 84, Tbili 1.1, Alk Phos 201; Lactic acid 2.1
Given these findings, particularly the transaminitis with elevated lipase, the initial working diagnosis was gallstone pancreatitis. However, there was only moderate LFT/bilirubin elevation and no biliary dilation or stones noted on CT or ultrasound. Another diagnostic consideration – though rare and ultimately not proven to be the case with this patient – was so-called viscerocutaneous loxoscelism (further explored in the discussion below).
The patient was readmitted to the hospital for IV fluids and analgesia. Due to the elevated LFTs and lipase, a decision was made to proceed with laparoscopic cholecystectomy. Following this procedure the patient’s LFTs and lipase began to downtrend, and he was discharged home.
Although the patient’s systemic symptoms had resolved, the RUE lesion continued to evolve showing severe tissue destruction, soft tissue defect, and minimal improvement with the wound vacuum.
On post-operative day 3, the initial outcome of the skin graft was acceptable, though some tissue loss was noted peripherally by the surgical team.
On post-operative day 10, there was a small area of tissue located proximally and medially that required intermittent packing, but a good general progression was noted by the surgical team.
At around day 100, the patient reported he was beginning to feel normal again, with near total resolution of symptoms.
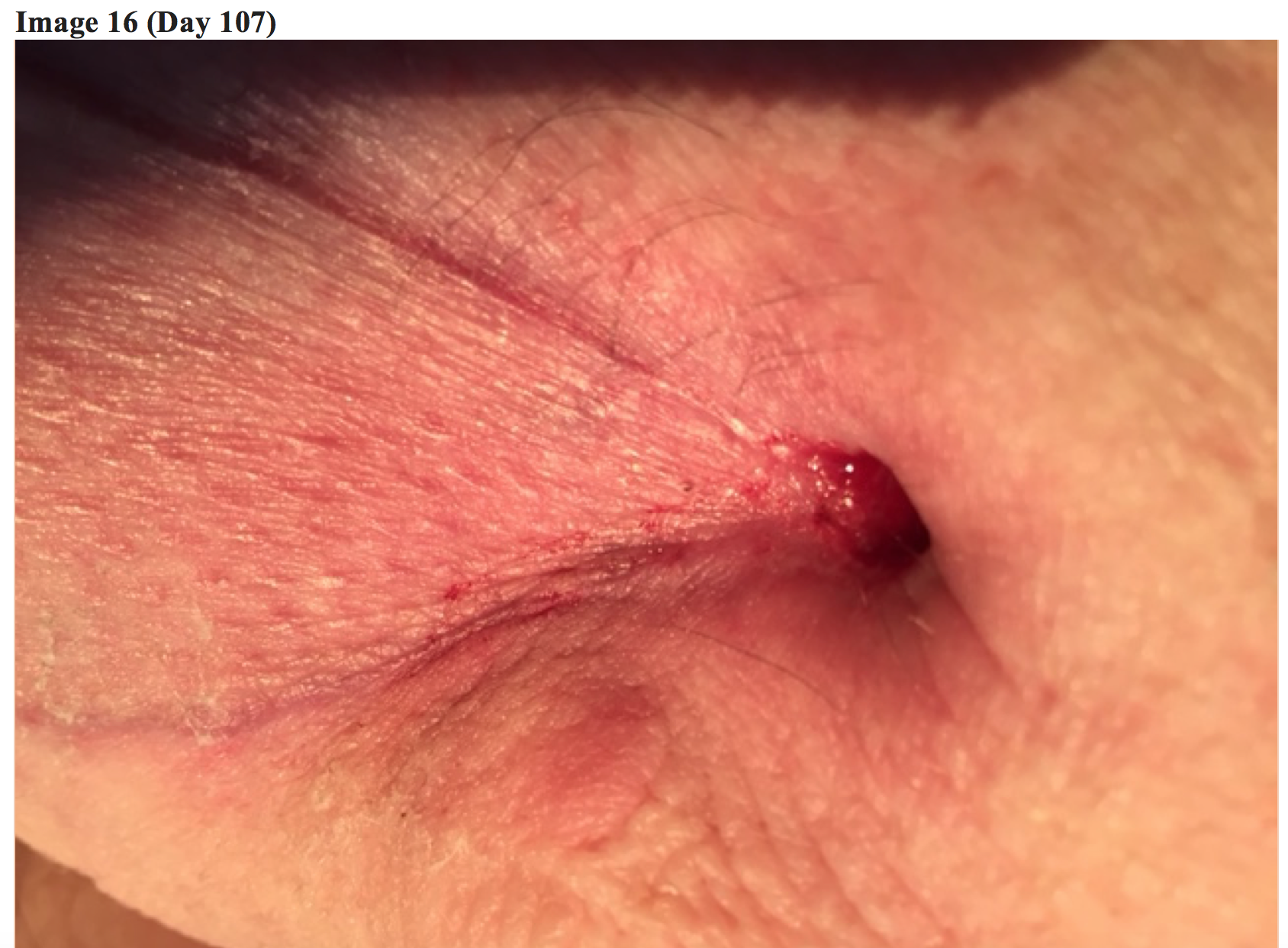
At approximately four months (Day 127) from initial bite, the patient’s arm began showing definitive healing.
Discussion
Morphology
The brown recluse spider, Loxosceles reclusa, is known commonly as Fiddleback, violin spider, and brown spider. The scientific name translates as follows: Loxos – slanting or oblique; skeles – leg; reclusa – hermit or enclosed. Combined these terms essentially become: Slanted-leg recluse. 
These solitary spiders have six eyes arranged in dyads, are about 7mm in body length and are tan or brown, with their legs often darker than the cephalothorax. With legs extended the total width of the spiders is about 2.5 cm. Upon close examination, a violin-shaped pattern is evident on the thorax – this pattern is common to all members of the loxosceles genus, although it is not present in juveniles (9)(6).
Geography
 Loxosceles spiders are common in the North American mid-west, as they inhabit much of this geographic area. The genus as a whole includes thirteen species and inhabits a large portion of the United States (2).
Loxosceles spiders are common in the North American mid-west, as they inhabit much of this geographic area. The genus as a whole includes thirteen species and inhabits a large portion of the United States (2).
Characteristics
Brown recluses are commonly found in many locations, though protected cover is most common. Examples of these may include leaf beds, rotting tree bark, wood piles. Additionally, the spiders thrive in man-made environments. Examples of these may include less disturbed areas such as basements, garages, and closets. Recluses can also be found in clothing piles, boxes, shoes, etc., where they rest, breed, and form irregular webs (9). They are most active from spring to fall at night when they leave their webs to stalk food, which generally consists of silverfish and other insects. They are incredibly resilient — capable of surviving 6 months without food or water (6). In addition, efforts to eliminate them with chemicals has questionable efficacy (6)(19). As the name implies, brown recluse spiders are not social creatures. Despite suggestions on the aggressiveness of their predatory nature, they do not seek out humans as prey. Rather, Loxosceles reclusa bite in defense or in response to being crushed between an object and skin (15)(9).
Venom/Toxin
The venom of Loxosceles reclusa contains a myriad of toxins including metalloproteinases, hyaluronidases, insecticidal peptides, deoxyribonucleases, and alkaline phosphatase (2). Sphingomyelinase D is the toxic protein of most interest (present only in the genera Loxosceles and Sicarius). Once delivered, it acts quickly to attack phospholipid structure and integrity. The toxin acts by altering the typical head/tail orientation of the phospholipid molecule into a ring structure, with complete destruction of the hydrophilic head. This creates a manipulated molecular structure, greatly dissimilar from the linear phospholipid, resulting in destabilization of the cell membranes (12).
Regarding host response, Loxosceles reclusa venom has been defined as an endothelial cell agonist that results in differential stimulation of the inflammatory response. This causes a loss of endothelial cell integrity and junctions, which results in extracellular space edema, hemorrhage, vessel wall degeneration, polymorphonuclear lymphocyte accumulation, and edema of the immediate muscle and soft tissue (17)(18)(13).
In addition to these toxic effects, sphingomyelinase D induces red blood cell (RBC) hemolysis and platelet aggregation by degrading the sphingomyelin component of the erythrocyte membrane (17)(10).
Finally, Loxosceles reclusa venom can induce the cleavage of endogenous metalloproteinase, resulting in degradation of glycophorins on the RBC surface (17)(13). Complement-mediated lysis of the erythrocytes is then activated via the alternative pathway (17).
Envenomation/Bites
Generally speaking, bites from recluses are minimally symptomatic. When there is a significant response to envenomation, the syndrome is so-called loxoscelism. This syndrome is most commonly a local response characterized by pain, erythema, soft tissue swelling, and central pallor around the bite site for the first several hours. Often the evolution of the bite stops at this stage. In cases with progressive cutaneous change, the morphology of the bite evolves to a blotchy blue or violet color and a hard depressed center (17)(10). Assuming the bite is on the torso or lower extremity, the expanding lesion is often oval shaped and wider inferiorly, possibly due to gravity ‘pulling’ the venom inferiorly. Envenomation can also result in vesicles, blisters, or necrotic ulcers of the cutaneous tissue and tissue loss, known as necrotic arachnidism or necrotic cutaneous loxoscelism (1)(2). As the cutaneous lesion develops, the venom is capable of causing intravascular clotting resulting in the occlusion of venules and arterioles (10). In more severe cases wounds can cause significant geographic progression and skin sloughing. The site can extend up to 40 cm wide and can involve deep structures. These wounds can take many weeks to heal by secondary intent (17).
Rarely, envenomation from Loxosceles reclusa can result in a serious systemic response syndrome known as viscerocutaneous loxoscelism (as considered in the above case study). This response can include pancreatitis, hemolytic anemia, jaundice, fever, disseminated intravascular coagulation (DIC), rhabdomyolysis, myonecrosis, and renal failure (3).
While the mechanism of viscerocutaneous loxoscelism is not fully understood, recluse venom is likely capable of inflicting direct visceral damage, particularly to the renal, hematologic, and gastrointestinal systems. Renal injury, for example, includes multiple pathways: glomerular edema, tubular necrosis, and hyalinization of tubules (7). Immunofluorescence studies have shown dermonecrotic toxin deposits on renal intrinsic structures – suggesting renal insult is a direct result of the envenomation of Loxosceles reclusa, rather than simply a manifestation of severe systemic illness (7). These findings would suggest visceral injury is a direct result of the envenomation, rather than simply a manifestation of critical illness.
A single study showed a potential relationship between glucose-6-phosphate deficiency (G6PD) and viscerocutaneous loxoscelism (3). In this case series, two out of seven patients screened with VL were positive for G6PD, possibly manifesting VL through oxidative strain resulting in hemolytic anemia.
Finally, fatal loxoscelism is more commonly associated with a fellow member of the genus (Loxosceles laeta), which is found in South America (2).
Differential Diagnosis
When assessing a possible brown spider bite, two of the most important considerations are geographic location, and Loxosceles reclusa sighting or presence. Because there is a lack of a commercially available assay for L. reclusa venom, definitively diagnosing an envenomation is difficult. The lesions can mimic various infections, ulcerations from trauma, vascular diseases, pyoderma gangrenosum, Stevens-Johnson syndrome, erythema nodosum, or erythema multiforme (15)(17). Cases within affected geographic areas, with a suggestive clinical story (eg pain following donning of clothing) should result in a high suspicion for recluse envenomation.
Treatment
Routine first aid treatment of L. reclusa bites is generally indicated to control inflammation and pain. This includes immobilization, ice, elevation, local wound care, NSAID administration as needed, and tetanus prophylaxis (1)(8). Because the majority of bites are minimally symptomatic, most can be treated with little medical intervention. Treatment of more severe manifestations of envenomation is more challenging. For secondary infections at the wound site, characterized by suppuration, increased erythema, and fluctuance, antibiotics targeted at cellulitis are indicated (1)(11). For necrotic wounds, multiple therapies have been considered, though are not well supported with outcome data. As illustrated in the case above, prognostication for these bites can be challenging. In addition to treatment, it is critical to counsel patients regarding the variable outcomes from suspected L. reclusa bites.
Summary:
– Brown recluse spiders are found in southern North America, with six eyes and a violin pattern on the thorax.
– The spiders are commonly found in isolated, man-made locations with cover. These spiders are very resistant to adverse enviroments.
– A bite is normally in defense, with venom containing metalloproteinases, hyaluronidases, insecticidal peptides, deoxyribonucleases, and alkaline phosphatase. These proteins destroy cells, resulting in tissue death.
– Bites normally have few symptoms. Loxoscelism occurs with significant local response that may transition through stages, ultimately resulting in skin breakdown. Systemic reactions are rare.
– The differential includes infections, ulcerations from trauma, vascular diseases, pyoderma gangrenosum, Stevens-Johnson syndrome, erythema nodosum, or erythema multiforme.
– Treatment includes immobilization, ice, elevation, local wound care, NSAID administration as needed, and tetanus prophylaxis. For necrotic wounds, multiple therapies have been considered, though are not well supported with outcome data.
References / Further Reading:
- Andersen, Rebecca J., Jennifer Campoli, Sandeep K. Johar, Katherine A. Schumacher, and E. Jackson Allison. “Suspected Brown Recluse Envenomation: A Case Report and Review of Different Treatment Modalities.” The Journal of Emergency Medicine 41.2 (2011): E31-37. Web.
- Arnold, Thomas. “Brown Recluse Spider Envenomation.” Emedicine. Web. 18 March 2016.
- Barretto O, Cardoso L, De Cillo D. Viscerocutaneous Loxoscelism and Erythrocyte Glucose-6-PhosphateDeficiency. Rev Inst Med. Trop S. Paulo. Internet. 1985 October; 27(5): 264-267
- “Brown Recluse Spiders: Facts, Identification & Control” orkin.com. Web.
- “Brown Recluses – Brown Recluse Spider Map.” University of California Riverside Entomology. Web.
- “Brown Recluse: Loxosceles Reclusa.” Arachnipedia.wiki.com. Web. 23 March 2016.
- Chaim, Olga Meiri, Youssef Bacila Sade, Rafael Bertoni Da Silveira, Leny Toma, Evanguedes Kalapothakis, Carlos Chávez-Olórtegui, Oldemir Carlos Mangili, Waldemiro Gremski, Carl Peter Von Dietrich, Helena B. Nader, and Silvio Sanches Veiga. “Brown Spider Dermonecrotic Toxin Directly Induces Nephrotoxicity.” Toxicology and Applied Pharmacology 211.1 (2006): 64-77. Web. July 2016.
- Elston, Dirk M., Scott D. Miller, Russell J. Young, Jeff Eggers, David Mcglasson, William H. Schmidt, and Anneke Bush. “Comparison of Colchicine, Dapsone, Triamcinolone, and Diphenhydramine Therapy for the Treatment of Brown Recluse Spider Envenomation.” Archives of Dermatology 141.5 (2005): 595-97.
- Forks, T. P. “Brown Recluse Spider Bites.” The Journal of the American Board of Family Medicine 13.6 (2000): 415-23. Web.
- Futrell, Josephine M. “Loxoscelism.” The American Journal of the Medical Sciences 304.4 (1992): 261-67.
- Hogan, Christopher J., Katia Cristina Barbaro, and Ken Winkel. “Loxoscelism: Old Obstacles, New Directions.” Annals of Emergency Medicine 44.6 (2004): 608-24. Web.
- Lajoie, Daniel M., Pamela A. Zobel-Thropp, Vlad K. Kumirov, Vahe Bandarian, Greta J. Binford, and Matthew H. J. Cordes. “Phospholipase D Toxins of Brown Spider Venom Convert Lysophosphatidylcholine and Sphingomyelin to Cyclic Phosphates.” PLoS ONE 8.8 (2013): n. pag. Web.
- Patel, K. D., V. Modur, G. A. Zimmerman, S. M. Prescott, and T. M. Mcintyre. “The Necrotic Venom of the Brown Recluse Spider Induces Dysregulated Endothelial Cell-dependent Neutrophil Activation. Differential Induction of GM-CSF, IL-8, and E-selectin Expression.” Journal of Clinical Investigation J. Clin. Invest. 94.2 (1994): 631-42. Web. July 2016.
- Málaque, Ceila Maria Sant’ana, Jaime Enrique Castro-Valencia, João Luiz Costa Cardoso, Francisco Oscar De Siqueira França, Kátia Cristina Barbaro, and Wen Fan Hui. “Clinical and Epidemiological Features of Definitive and Presumed Loxoscelism in São Paulo, Brazil.” Revista Do Instituto De Medicina Tropical De São Paulo Rev. Inst. Med. Trop. S. Paulo 44.3 (2002): 139-43. Web.
- Potter, Mike. “Brown Recluse Spider.” UKY Entomology. University of Kentucky College of Agriculture, June 2005. Web. July 2016.
- Szalay, Jessie. “Brown Recluse Spiders: Facts, Bites, Symptoms.” LiveScience.com. Web.
- Swanson, David L., and Richard S. Vetter. “Loxoscelism.” Clinics in Dermatology 24.3 (2006): 213-21. Web July 2016.
- Vetter, Richard S., MS, and David L. Swanson, MD. “Bites of Recluse Spiders.” UpToDate. Ed. Daniel F. Danzl, Stephen J. Traub, and James F. Wiley. Web. July 2016.
- “Where Do Brown Recluse Spiders Live? (Dwellings & Range).” Orkin. Web. July 2016.