Today on the emDOCs cast, Brit Long, MD (@long_brit) covers Part 2 in a series on complications of CAR T-cell therapy, specifically immune effector cell-associated neurotoxicity syndrome (ICANS), infusion reactions, cytopenias, graft versus host disease, and cardiac toxicity.
Episode 78: CAR T-cell Therapy, ICANS, and other complications
Immune effector cell-associated neurotoxicity syndrome
- Neurotoxicity (ICANS and cytokine release encephalopathy syndrome (CRES)) can occur days to weeks following CAR T-cell therapy, as well as other types of immunotherapy such as blinatumomab.
- Those with immature B cell diseases are more commonly affected (87% of cases).
- ICANS occurs in 39-64% of patients in the first 8 weeks of therapy. More common with certain regimens (axicabtagene and brexucabtegene), which cause faster T-cell expansion and metabolism.
- Younger age, preexisting comorbidities, higher disease burden, increased intensity of cancer therapies, and concomitant CRS increase the risk of ICANS.
- Up to 90% of those with ICANS also have CRS.
Pathophysiology:
- ICANS is thought to be due to cytokines (L-1, IL-6, IL-8, IL-10, interferon, and TNF-alpha) causing activation of endothelial cells and disruption of the blood-brain barrier, but it’s not completely understood.
- Neurotoxic substances including glutamate and quinolinic acid are also significantly elevated in patients with neurotoxicity. Once the blood-brain barrier is disrupted, the substances and cytokines cause neuronal inflammation and damage.
Features:
- Variable presentation: headache, memory loss, dizziness, meningismus, altered mental status, movement disorders (myoclonus, tremor), speech impairment (aphasia, dysarthria), seizures, encephalopathy, and coma.
- Think about this in the patient with evidence of CRS plus neuro findings.
- Symptoms may last for 6-17 days.
Evaluation:
- Laboratory testing can demonstrate elevated lactate dehydrogenase, thrombocytopenia, and elevated inflammatory markers (ferritin, CRP, ESR).
- If LP performed, findings are typically normal or may demonstrate a mild elevation in protein and pleocytosis.
- CT of the head recommended to look for other complications (bleed, abscess, mets). In severe ICANS, cerebral edema will be present on CT with sulcal effacement and white matter changes. SAH, SDH, and stroke can occur.
- Magnetic resonance imaging (MRI) is the imaging modality of choice, but this is normal in over half of patients. 30% will demonstrate T2/FLAIR hyperintensities.
- EEG can demonstrate findings of encephalopathy in 76%, with theta-delta slowing.
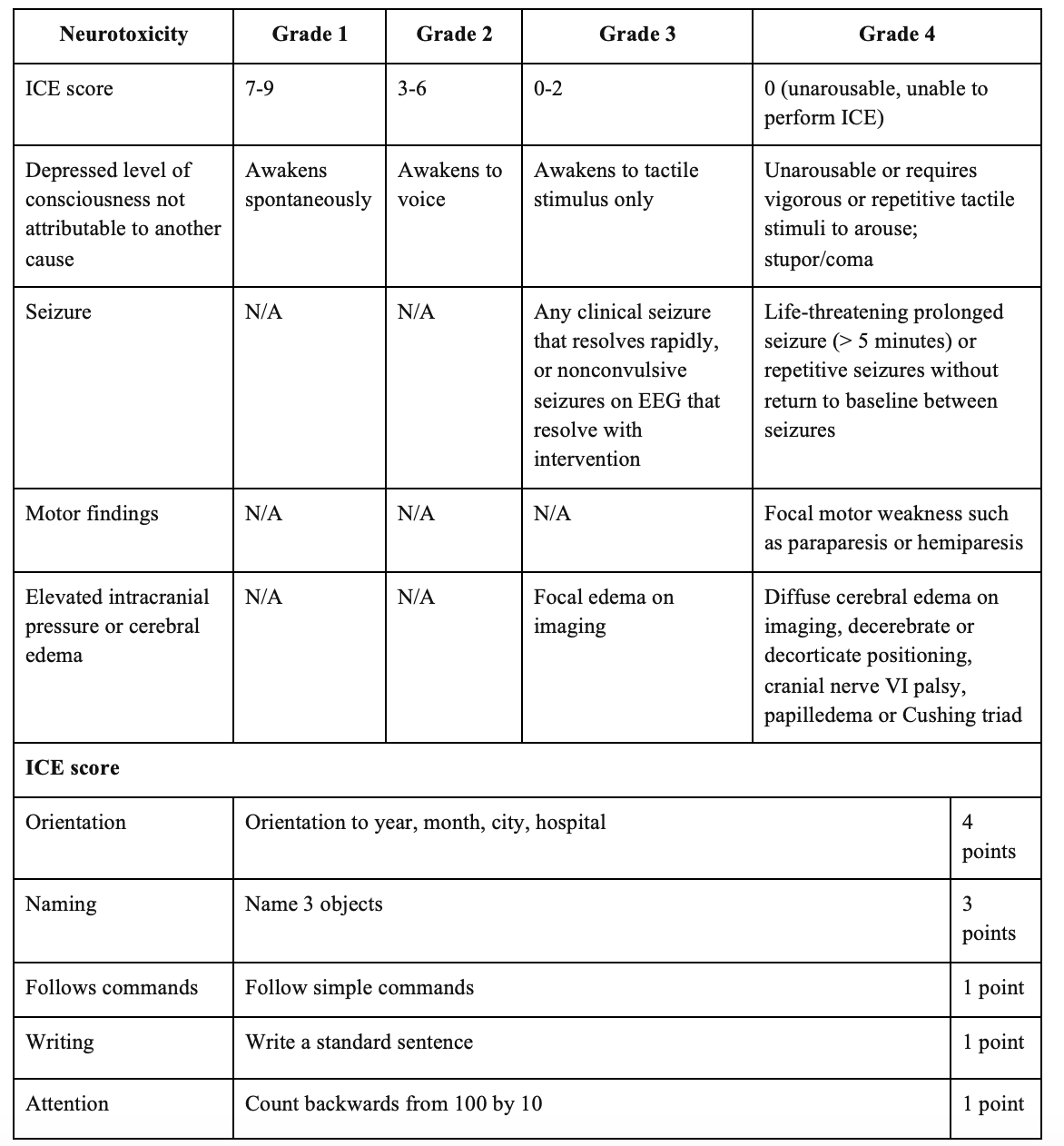
Grading:
Based on the most severe symptoms, considering five components: encephalopathy using the Immune Effector Cell-Associated Encephalopathy (ICE) score
- ICE score, level of consciousness, presence of seizure, motor examination, and elevated intracranial pressure.
- Grade 1 is mild, with some inattentiveness and mild disorientation, grade 2 is moderate, and grades 3-4 are severe.
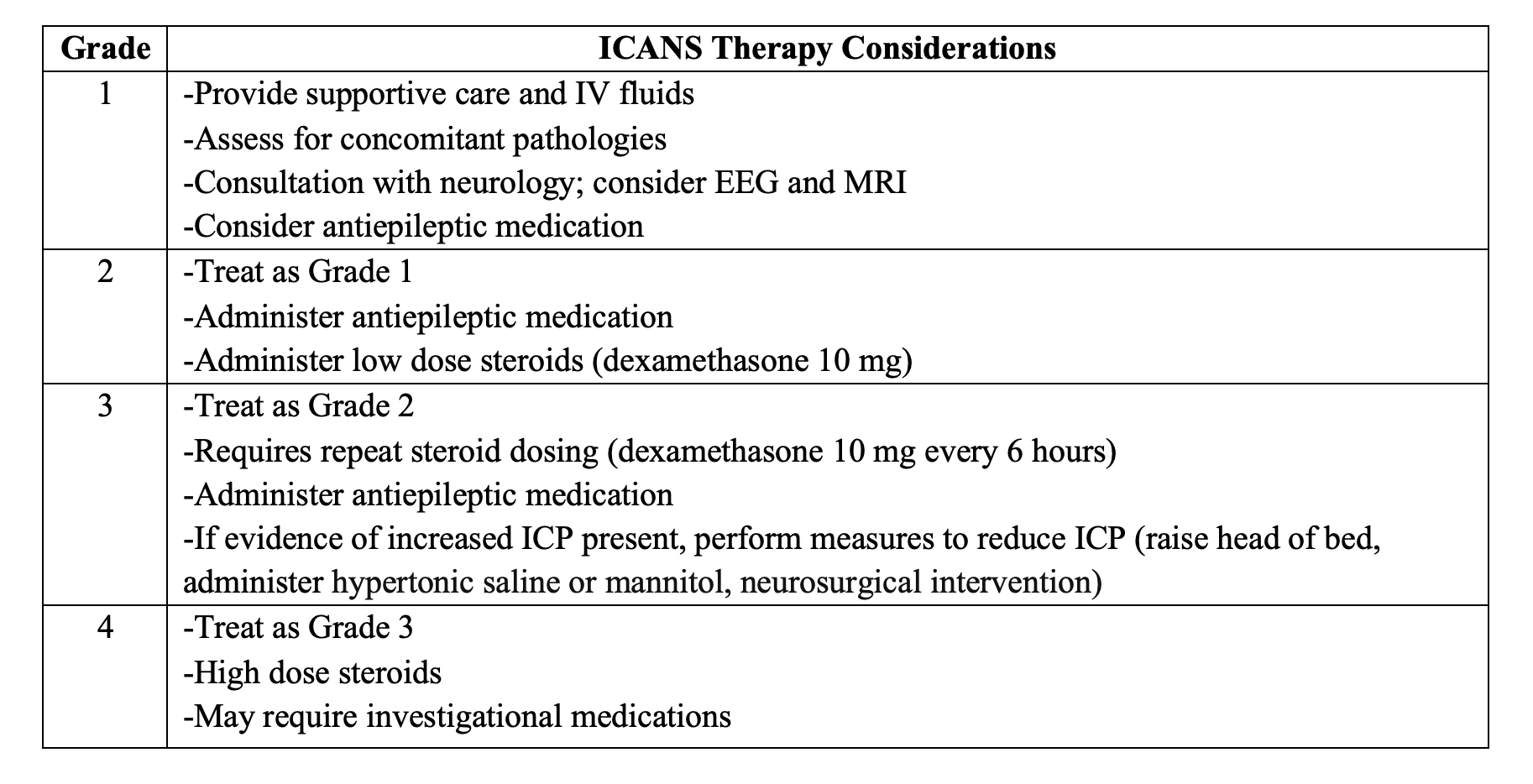
- Consult hematology/oncology.
- Supportive care with steroids and seizure treatment.
- Dexamethasone 10 mg 2-4 times per day will improve symptoms in most cases. These are tapered once patient begins to return to baseline neurological status.
- If ICP elevated and patient decompensating, elevate head of bed, provide analgesia/antiemetics, and give HTS or mannitol. However, steroids reduce ICP in most cases.
- Prophylactic seizure therapy is recommended with levetiracetam, which has less cross reactivity with other medications and has lower risk of cardiac or liver toxicity.
Other Complications
- Infusion reactions are usually mild and infrequent. Symptoms include nausea and vomiting, but hypotension and anaphylaxis have been reported.
- Hypogammaglobulinemia: B-cell aplasia and persistence of CAR T-cell therapy effects. Associated with complete response to therapy. IgG levels decrease 1-3 months after CAR T-cell therapy and may remain low up to 4 years. Patients are typically treated with IVIG.
- Cytopenias can occur for several weeks following therapy; the most common is neutropenia.
- Absolute neutrophil counts are below 1.0 x 109/L in 53-78% of cases, with neutropenic fever occurring in 17-37%.
- Thrombocytopenia occurs in 11-38%.
- GVHD: May occur in patients receiving donor-derived T cells, but it’s unlikely due to the in vitro modification of T cells to express the CAR through introduction of the encoding by a viral or non-viral vector.
- Prior episodes of GVHD increase the risk of future GVHD, current literature suggests the risk of GVHD is low in patients receiving CAR T-cell therapy; a meta-analysis found 6.9% of patients receiving CAR T-cell therapy with donor-derived cells developed GVHD. In this meta-analysis, GVHD presented most commonly with skin symptoms or liver involvement.
- Cardiac Toxicity can occur in up to 39% of patients and includes arrhythmias, cardiomyopathy, heart failure, and cardiac arrest
- Most likely due to IL-6, which has direct cardiac effects. CAR T-cell therapy can also cause direct cardiotoxicity due to the potential of cross-reactivity with proteins expressed by cardiac tissue
- New onset cardiomyopathy associated with CAR T-cell therapy appears to be more common in patients of older age and those with preexisting cardiovascular risk factors
- CRS is also associated with cardiac complications, including arrhythmias, cardiomyopathy, and ischemia.
- Patients with STEMI should be treated similar to those not on CAR T-cell therapy with PCI. Tocilizumab may treat cardiac toxicity related to CAR T-cells.
References:
- Long B, Yoo MJ, Brady WJ, Holian A, Sudhir A, Gottlieb M. Chimeric antigen receptor T-cell therapy: An emergency medicine focused review. Am J Emerg Med. 2021 Dec;50:369-375.
- Neelapu SS, Tummala S, Kebriaei P, et al. Chimeric antigen receptor T cell therapy-assessment and management of toxicities. Nat Rev Clin Oncol. 2018;15:47–62.
- Lee DW, Santomasso BD, Locke FL, et al. ASTCT Consensus Grading for Cytokine Release Syndrome and Neurologic Toxicity Associated with Immune Effector Cells. Biol Blood Marrow Transplant. 2019 Apr;25(4):625-638.
- Holtzman NG, Xie H, Bentzen S, et al. Immune effector cell-associated neurotoxicity syndrome after chimeric antigen receptor T-cell therapy for lymphoma: predictive biomarkers and clinical outcomes. Neuro Oncol. 2021 Jan 30;23(1):112-121.
- Karschnia P, Jordan JT, Forst DA, et al Clinical presentation, management, and biomarkers of neurotoxicity after adoptive immunotherapy with CAR T cells. Blood. 2019 May 16;133(20):2212-2221.
- Rubin DB, Danish HH, Ali AB, et al. Neurological toxicities associated with chimeric antigen receptor T-cell therapy. Brain. 2019 May 1;142(5):1334-1348.
- Rubin DB, Al Jarrah A, Li K, et al. Clinical Predictors of Neurotoxicity After Chimeric Antigen Receptor T-Cell Therapy. JAMA Neurol. 2020 Dec 1;77(12):1536-1542.
- Rice J, Nagle S, Randall J, Hinson HE. Chimeric Antigen Receptor T Cell-Related Neurotoxicity: Mechanisms, Clinical Presentation, and Approach to Treatment. Curr Treat Options Neurol. 2019 Jul 20;21(8):40.
- Cruz CR, Hanley PJ, Liu H, et al. Adverse events following infusion of T cells for adoptive immunotherapy: a 10-year experience. Cytotherapy. 2010;12:743–749
- Kochenderfer JN, Wilson WH, Janik JE, et al. Eradication of B-lineage cells and regression of lymphoma in a patient treated with autologous T cells genetically engineered to recognize CD19. Blood. 2010;116:4099–4102.
- Park JH, Rivière I, Gonen M, et al. Long-term follow-up of CD19 CAR therapy in acute lymphoblastic leukemia. N Engl J Med. 2018;378:449–459.
- Doan A, Pulsipher MA. Hypogammaglobulinemia due to CAR T-cell therapy. Pediatr Blood Cancer. 2018;65:2017–2018.
- Alvi RM, Frigault MJ, Fradley MG, et al. Cardiovascular Events Among Adults Treated With Chimeric Antigen Receptor T-Cells (CAR-T).J Am Coll Cardiol. 2019;74:3099-108.
- Lefebvre B, Kang Y, Smith AM, et al. Cardiovascular Effects of CAR T Cell Therapy: A Retrospective Study.JACC CardioOncol. 2020;2:193-203.






