Author: Marina Boushra, MD (EM Attending Physician, Vidant Medical Center) // Reviewed by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case:
A 27-year-old G4P2 female at 25-weeks gestation presents to the emergency department with right upper quadrant abdominal pain. She also notes repetitive vomiting, which she was attributing to worsening of her baseline “morning sickness” as her pregnancy progressed. Her vital signs on arrival to the emergency department are T 37.2 C, HR 125 bpm, RR 25, BP 150/90. Abdominal examination reveals localized right upper quadrant tenderness, hyperreflexia, and 3+ pitting edema of bilateral lower extremities.
Introduction:
Abdominal pain is an extremely common complaint during pregnancy, affecting approximately 1 in 500-635 pregnant women.1 This pain is often caused by physiologic changes associated with the growing uterus, such as strain on the broad ligament. obstetric and non-obstetric pathologies may rarely present together with acute abdominal pain in pregnant women. This article focuses on the evaluation of obstetric causes of pathologic abdominal pain. The assessment of these patients is particularly challenging due to physiological and anatomic changes that result in atypical presentations of relatively common diseases. The choice of therapeutic and diagnostic procedures in pregnant patients is likewise challenging and must include an assessment of safety both to the patient and the fetus. Thus, the assessment of pregnant patients with acute abdominal pain requires a structured approach that considers both obstetric and non-obstetric causes of abdominal pain. Since medical and surgical emergencies in pregnant patients are relatively rare, many clinicians may be uncomfortable with their diagnosis and management, leading to diagnostic delay and complications for this particularly high-risk patient population. Early multi-disciplinary involvement in the care of these patients is important.
Obstetric Causes of Acute Abdominal Pain in Pregnancy:
Ectopic pregnancy
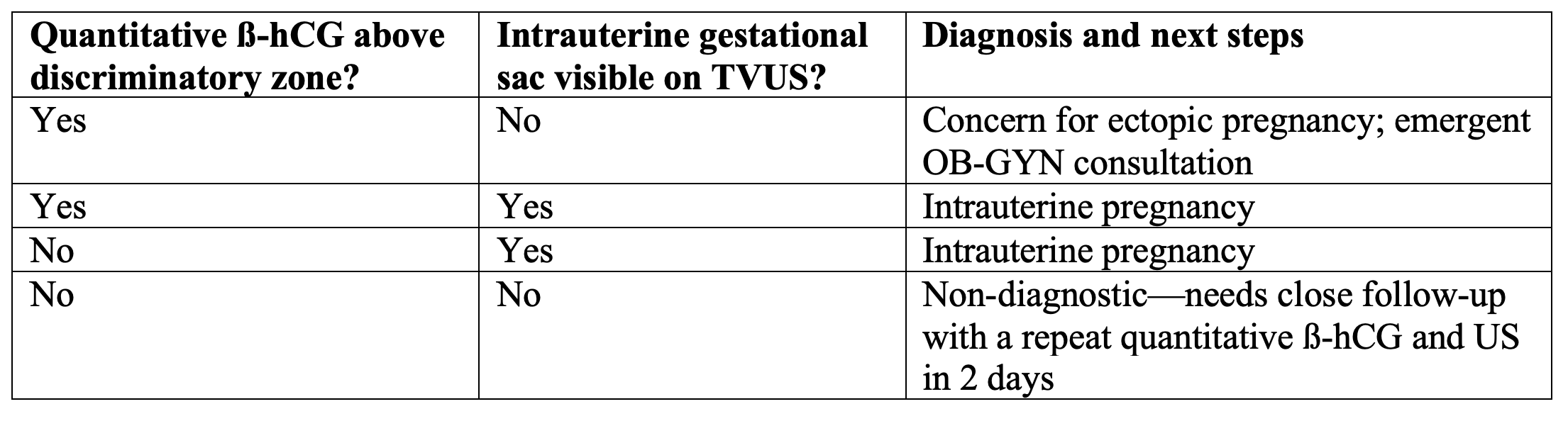
Ectopic pregnancy is defined as the implantation of a pregnancy in any site other than the endometrial lining of the uterus, most commonly in the fallopian tubes. Risk factors for the development of ectopic pregnancy include prior tubal surgery, a history of ectopic pregnancy or pelvic inflammatory disease, or fertility treatments.2,3 Ectopic pregnancies are relatively common, accounting for an estimated 1-2% of pregnancies, and incidence may be rising due to more widespread use of assisted reproductive technology.2 Rupture of ectopic pregnancies accounts for approximately 6% of maternal mortality in industrialized nations.4 Transvaginal ultrasound is the initial diagnostic test of choice for diagnosis of ectopic pregnancy in the emergency department.5 A quantitative ß-hCG may be drawn in conjunction with the transvaginal ultrasound to aid in diagnosis based on the “discriminatory zone” (Table 1).5 The discriminatory zone describes a concept that there is a ß-hCG level above which the gestational sac associated with a normal intrauterine pregnancy should reliably be visible on ultrasound. In the case of transvaginal ultrasound, the discriminatory zone ß-hCG level has traditionally been set at >1,500 mIU/ml.6,7 However, the concept of the discriminatory zone has been recently been challenged.8,9 In fact, the American College of Obstetrics and Gynecology now recommends that, if the discriminatory zone is used as an aid in the diagnosis of ectopic pregnancy, the value be set conservatively high, such as at 3,500 mIU/mL.5 A transvaginal ultrasound that fails to demonstrate a gestational sac with a quantitative ß-hCG greater than the discriminatory zone is concerning for an ectopic pregnancy. A transvaginal ultrasound that fails to demonstrate an intrauterine pregnancy with a ß-hCG below the discriminatory zone is an indication for repeat ultrasound and measurement of the quantitative ß-hCG in two days.5 These patients must be given strict return precautions on discharge and must be made aware that an ectopic pregnancy has not definitively been ruled out. Follow-up should ideally be with the patient’s obstetrician, but patients who do not have primary care should be instructed to return to the emergency department for repeat ß-hCG and transvaginal ultrasound in two days. 5,10
A diagnosed ectopic pregnancy is an obstetric emergency, and immediate obstetric consultation is indicated. In patients without hemodynamic compromise, therapeutic options include outpatient management with methotrexate versus surgical removal of the ectopic pregnancy.3 In patients with evidence of hemodynamic instability or rupture, emergent surgical management is indicated.11
Table 1: Use of a discriminatory zone quantitative ß-hCG and ultrasound in the diagnosis of an ectopic pregnancy.
Abruption
Placental abruption, also known as abruptio placentae, is bleeding at interface of the placenta and the uterine decidua, causing complete or partial detachment of the placenta from the uterus before delivery of the fetus. Abruption is estimated to affect 0.6-1% of pregnancies, and it is the second most common cause of vaginal bleeding after 20 weeks of gestation.12 Risk factors for placental abruption include maternal trauma, cocaine use, multiple gestation, anti-phospholipid antibodies, hypertension, preeclampsia/eclampsia, tobacco use, and previous placental abruption.3 Placental abruption typically presents with late pregnancy bleeding accompanied by uterine pain or contractions.13 Almost 20% of placental abruptions, however, do not present with vaginal bleeding, with the blood accumulating between the placenta and the uterine wall. Retroplacental hematoma is the most classic finding of placental abruption on ultrasound.14 Other findings include echogenic debris in the amniotic fluid or placental thickening. However, while specific, ultrasound has a sensitivity of 57% for placental abruption, and a normal ultrasound does not exclude the diagnosis.14,15 The diagnosis of placental abruption is clinical, and high suspicion must be maintained for any woman with late pregnancy vaginal bleeding or abdominal pain. Physical examination may show a uterus that is tense, tender, and larger than expected for gestational age.3 Fibrinogen levels greater than 200 mg/dL have the best correlation with severe maternal hemorrhage.16 The complications of placental abruption include placental or uterine insufficiency leading to fetal demise, maternal coagulopathy with disseminated intravascular coagulation (DIC), and hemorrhagic shock.13,17 Management of placental abruption depends on maternal and fetal status. Stable mothers and fetuses with reassuring heart rates may attempt a vaginal delivery.17 Mothers with unstable vitals or those with fetuses with non-reassuring heart rates should proceed to emergent cesarean section.17
Hemolysis, Elevated Liver enzymes, Low Platelet (HELLP) Syndrome
HELLP syndrome is a severe form of preeclampsia associated with hemolysis, transaminitis, and thrombocytopenia. While most cases of HELLP are associated with the hallmark hypertension and proteinuria of preeclampsia, hypertension and proteinuria are not necessary for the diagnosis of HELLP syndrome (Table 2). In fact, hypertension and proteinuria are absent in 13-18% of cases of HELLP syndrome.18 The development of abdominal pain in HELLP syndrome is concerning for the development of a hepatic subcapsular hematoma or hepatic rupture, rare but life-threatening complications of HELLP syndrome.19 Hepatic hematomas occur in 0.9-1.6% of patients with HELLP syndrome and affect about 1 in 40,000 pregnancies.20 These patients typically complain of pain localized to the right upper quadrant and will show evidence of hemodynamic compromise due to hemorrhage.21 These patients should be aggressively resuscitated with blood products.21 Early obstetric and surgical specialist involvement in these patients is advisable. Like in preeclampsia and eclampsia, delivery is the only definitive management of HELLP.22
Table 2: Diagnostic criteria for HELLP syndrome.23

Uterine Rupture
Uterine rupture most commonly occurs at the site of prior uterine surgical manipulation, such as scar sites of prior caesarian sections or myomectomies.24 Less commonly, uterine rupture can occur in a previously intact uterus, an event termed primary uterine rupture and most common in cases of grand-multiparity.25 Uterine rupture most commonly occurs during labor but may be seen in the emergency department in patients who have experienced significant blunt abdominal trauma, such as a motor vehicle collision.25 The pain experienced in uterine rupture occurs secondary to peritoneal irritation from hemoperitoneum, and diaphragmatic irritation from the hemoperitoneum can result in pain referring to the shoulder or scapular regions.25 The first sign of uterine rupture is non-reassuring fetal heart tracings, including significant fetal bradycardia. Maternal examination may reveal a tense and tender uterus and palpation of fetal parts.25 Uterine rupture is an obstetric emergency, and management is surgical intervention for delivery and uterine repair or hysterectomy. Definitive diagnosis can be made at the time of surgical intervention.
Ovarian torsion
Ovarian torsion is a relatively common gynecologic emergency that occurs when the ovary twists around on the fallopian tube, impairing venous drainage and arterial supply and ultimately leading to necrosis with resultant fertility implications. The rate of ovarian torsion in pregnancy is estimated to be 1 in 1000-2500.26–28 Pregnancy after assisted reproductive treatments is a risk factor for torsion, with up to 16% of these pregnancies complicated by torsion, and recurrence of torsion is most common in pregnant patients.26 The presentation of torsion is similar in pregnant and non-pregnant patients and typically includes lower abdominal pain, nausea, and vomiting.26 Despite this, pregnant patients are much more likely than non-pregnant patients to be misdiagnosed and discharged on the first encounter.27 Torsion is most common in the first trimester of pregnancy, which has been attributed to the higher incidence of functional cysts at that time.26 Notably, the majority of torsions in the second and third trimester have been reported in otherwise normal ovaries, and the cause is hypothesized to be movement of the ovaries by the rapidly growing uterus or hypermobility of the ovarian ligaments during this stage of pregnancy.27 Transvaginal ultrasound is the initial test of choice, with laparoscopic evaluation being the gold standard for diagnosis. Ultrasonographic findings consistent with torsion include enlarged ovaries, stromal edema, free pelvic fluid, and para-ovarian cysts or masses.27 Pelvic ultrasound is 80% sensitive and 95% specific for the diagnosis of ovarian torsion.29 It is particularly important to remember that torsion is a dynamic process, and that a negative ultrasonographic examination does not definitively rule out torsion, especially in pregnant patients in whom ultrasonographic evaluation is more difficult.26,27 If discharge is being considered, these patients need strict return precautions if symptoms return. Definitive management of ovarian torsion is surgical de-torsion.
Pelvic Inflammatory Disease (PID)
PID is infection of the upper genital tract (uterus, endometrium, fallopian tubes, ovaries) in women. PID may extend to involve adjacent structures, causing peri-appendicitis, pelvic peritonitis, and peri-hepatitis (Fitz-Hugh-Curtis syndrome). The majority of PID is caused by ascending sexually-transmitted infections (STI), with Neisseria gonorrhea and Chlamydia trachomatis being the most commonly implicated pathogens in PID.30 Any sexually active woman is at risk for PID, but women with multiple sexual partners are at the highest risk.31 Pregnancy decreases the risk of PID due to the mucus plug preventing ascending infection from the lower to the upper genital tract, though PID can still develop in the first 12 weeks of pregnancy.6
In symptomatic patients, lower abdominal pain, typically bilateral, is often the presenting symptom.1 Dyspareunia and onset of pain with or shortly after menstruation should also raise suspicion for PID.7 A history of abnormal uterine bleeding, such as menorrhagia, metrorrhagia, or post-coital bleeding, is seen in one third of patients with PID.8 Physical examination typically reveals lower abdominal pain, which may be unilateral or bilateral, although diffuse abdominal pain may also be seen.32 Cervical motion tenderness and the classic “chandelier sign”—severe pain with cervical motion such that the patient “jumps for the chandelier” on examination—may be present.32 Fever, rebound, hypoactive bowel sounds are indicative of more severe disease and should increase suspicion for a pelvic abscess.33 Associated right upper quadrant pain is concerning for Fitz-Hugh-Curtis syndrome inflammation of the hepatic capsule. The most common cause of death in PID is rupture of a tubo-ovarian abscess (TOA), which has a mortality rate of 5-10%.33 Pregnancy is an indication for admission in patients with suspected PID and pregnant patients should receive parenteral therapy for PID. A second-generation cephalosporin (cefoxitin or cefotetan) and azithromycin are the antibiotics of choice, and regimens using doxycycline or gentamicin should be avoided during pregnancy due to adverse fetal effects.11,34
Table 3: Summary table for obstetric and gynecologic causes of abdominal pain in pregnancy.

Key Takeaways:
- Abdominal pain in pregnant patients can be caused by the physiologic and anatomic changes associated with pregnancy as well as acute pathology, which may be obstetric or non-obstetric in nature. An appropriate work-up of abdominal pain in pregnant patients should aim to systematically exclude both obstetric and non-obstetric pathologic causes of abdominal pain before attributing the pain to normal physiologic changes in pregnancy.
- Pregnant patients are susceptible to the same diseases, including appendicitis and biliary disease, that are common in the general population. These conditions need to be considered in addition to obstetric causes in the work-up of abdominal pain in pregnant patients.
- “Mild” vital sign abnormalities such as hypertension or an increase in the pulse pressure need to be taken seriously in pregnant patients, as they can be the earliest indictors of serious pathology.
- Consider both uterine (rupture, abruption) and extrauterine pathology (ectopic, HELLP, ovarian torsion, PID) as potential causes of abdominal pain in pregnancy.
- Early involvement of OB-GYN specialists can aid in diagnosis as well as ensure disposition and follow-up in these high-risk patients.
From Dr. Katy Hanson at Hanson’s Anatomy:
References/Further Reading:
- Augustin G, Majerovic M. Non-obstetrical acute abdomen during pregnancy. Eur J Obstet Gynecol Reprod Biol. 2007;131(1):4-12. doi:10.1016/j.ejogrb.2006.07.052
- Clayton HB, Schieve LA, Peterson HB, Jamieson DJ, Reynolds MA, Wright VC. Ectopic Pregnancy Risk With Assisted Reproductive Technology Procedures. Obstet Gynecol. 2006;107(3):595-604. doi:10.1097/01.AOG.0000196503.78126.62
- Zachariah SK, Fenn M, Jacob K, Arthungal SA, Zachariah SA. Management of acute abdomen in pregnancy: current perspectives. Int J Womens Health. 2019;Volume 11:119-134. doi:10.2147/IJWH.S151501
- Berg CJ, Callaghan WM, Syverson C, Henderson Z. Pregnancy-related mortality in the United States, 1998 to 2005. Obstet Gynecol. 2010;116(6):1302-1309. doi:10.1097/AOG.0b013e3181fdfb11
- ACOG Practice Bulletin No. 191. Obstet Gynecol. 2018;131(2):e65-e77. doi:10.1097/AOG.0000000000002464
- Surampudi K, Gundabattula SR. The Role of Serum Beta hCG in Early Diagnosis and Management Strategy of Ectopic Pregnancy. J Clin Diagn Res. 2016;10(7):QC08-10. doi:10.7860/JCDR/2016/19342.8110
- Lozeau A-M, Potter B. Diagnosis and management of ectopic pregnancy. Am Fam Physician. 2005;72(9):1707-1714. http://www.ncbi.nlm.nih.gov/pubmed/16300032. Accessed July 21, 2019.
- Connolly A, Ryan DH, Stuebe AM, Wolfe HM. Reevaluation of Discriminatory and Threshold Levels for Serum β-hCG in Early Pregnancy. Obstet Gynecol. 2013;121(1):65-70. doi:10.1097/AOG.0b013e318278f421
- Doubilet PM, Benson CB. Further Evidence Against the Reliability of the Human Chorionic Gonadotropin Discriminatory Level. J Ultrasound Med. 2011;30(12):1637-1642. doi:10.7863/jum.2011.30.12.1637
- Seeber BE, Sammel MD, Guo W, Zhou L, Hummel A, Barnhart KT. Application of redefined human chorionic gonadotropin curves for the diagnosis of women at risk for ectopic pregnancy. Fertil Steril. 2006;86(2). doi:10.1016/j.fertnstert.2005.12.056
- Ness RB, Soper DE, Holley RL, et al. Effectiveness of inpatient and outpatient treatment strategies for women with pelvic inflammatory disease: results from the Pelvic Inflammatory Disease Evaluation and Clinical Health (PEACH) Randomized Trial. Am J Obstet Gynecol. 2002;186(5):929-937. http://www.ncbi.nlm.nih.gov/pubmed/12015517. Accessed December 17, 2016.
- TIKKANEN M. Placental abruption: epidemiology, risk factors and consequences. Acta Obstet Gynecol Scand. 2011;90(2):140-149. doi:10.1111/j.1600-0412.2010.01030.x
- Li Y, Tian Y, Liu N, Chen Y, Wu F. Analysis of 62 placental abruption cases: Risk factors and clinical outcomes. Taiwan J Obstet Gynecol. 2019;58(2):223-226. doi:10.1016/J.TJOG.2019.01.010
- Jha P, Melendres G, Bijan B, et al. Trauma in pregnant women: assessing detection of post-traumatic placental abruption on contrast-enhanced CT versus ultrasound. Abdom Radiol. 2017;42(4):1062-1067. doi:10.1007/s00261-016-0970-x
- Shinde GR, Vaswani BP, Patange RP, Laddad MM, Bhosale RB. Diagnostic Performance of Ultrasonography for Detection of Abruption and Its Clinical Correlation and Maternal and Foetal Outcome. J Clin DIAGNOSTIC Res. 2016;10(8):QC04-7. doi:10.7860/JCDR/2016/19247.8288
- de Lloyd L, Bovington R, Kaye A, et al. Standard haemostatic tests following major obstetric haemorrhage. Int J Obstet Anesth. 2011;20(2):135-141. doi:10.1016/j.ijoa.2010.12.002
- Oyelese Y, Ananth C V. Placental abruption. Obstet Gynecol. 2006;108(4):1005-1016. doi:10.1097/01.AOG.0000239439.04364.9a
- Sibai BM. Diagnosis, Controversies, and Management of the Syndrome of Hemolysis, Elevated Liver Enzymes, and Low Platelet Count. Obstet Gynecol. 2004;103(5, Part 1):981-991. doi:10.1097/01.AOG.0000126245.35811.2a
- Haddad B, Barton JR, Livingston JC, Chahine R, Sibai BM. Risk factors for adverse maternal outcomes among women with HELLP (hemolysis, elevated liver enzymes, and low platelet count) syndrome. Am J Obstet Gynecol. 2000;183(2):444-448. doi:10.1067/mob.2000.105915
- Sibai BM, Ramadan MK, Usta I, Salama M, Mercer BM, Friedman SA. Maternal morbidity and mortality in 442 pregnancies with hemolysis, elevated liver enzymes, and low platelets (HELLP syndrome). Am J Obstet Gynecol. 1993;169(4):1000-1006. doi:10.1016/0002-9378(93)90043-i
- DITISHEIM A, SIBAI BM. Diagnosis and Management of HELLP Syndrome Complicated by Liver Hematoma. Clin Obstet Gynecol. 2017;60(1):190-197. doi:10.1097/GRF.0000000000000253
- ACOG Committee Opinion No. 767 Summary. Obstet Gynecol. 2019;133(2):409-412. doi:10.1097/AOG.0000000000003082
- ACOG Practice Bulletin No. 202: Gestational Hypertension and Preeclampsia. Obstet Gynecol. 2019;133(1):e1-e25. doi:10.1097/AOG.0000000000003018
- Sturzenegger K, Schäffer L, Zimmermann R, Haslinger C. Risk factors of uterine rupture with a special interest to uterine fundal pressure. J Perinat Med. 2017;45(3):309-313. doi:10.1515/jpm-2016-0023
- Mukherjee R, Samanta S. Surgical emergencies in pregnancy in the era of modern diagnostics and treatment. Taiwan J Obstet Gynecol. 2019;58(2):177-182. doi:10.1016/J.TJOG.2019.01.001
- Ginath S, Shalev A, Keidar R, et al. Differences Between Adnexal Torsion in Pregnant and Nonpregnant Women. J Minim Invasive Gynecol. 2012;19(6):708-714. doi:10.1016/J.JMIG.2012.07.007
- Smorgick N, Pansky M, Feingold M, Herman A, Halperin R, Maymon R. The clinical characteristics and sonographic findings of maternal ovarian torsion in pregnancy. doi:10.1016/j.fertnstert.2008.09.028
- Condous G, Khalid A, Okaro E, Bourne T. Should we be examining the ovaries in pregnancy? Prevalence and natural history of adnexal pathology detected at first-trimester sonography. Ultrasound Obstet Gynecol. 2004;24(1):62-66. doi:10.1002/uog.1083
- Swenson DW, Lourenco AP, Beaudoin FL, Grand DJ, Killelea AG, Mcgregor AJ. Ovarian torsion: Case-control study comparing the sensitivity and specificity of ultrasonography and computed tomography for diagnosis in the emergency department. Eur J Radiol. 2014;83:733-738. doi:10.1016/j.ejrad.2014.01.001
- Workowski KA, Bolan GA, Centers for Disease Control and Prevention. Sexually transmitted diseases treatment guidelines, 2015. MMWR Recomm reports Morb Mortal Wkly report Recomm reports. 2015;64(RR-03):1-137. http://www.ncbi.nlm.nih.gov/pubmed/26042815. Accessed December 17, 2016.
- Lee NC, Rubin GL, Grimes DA. Measures of sexual behavior and the risk of pelvic inflammatory disease. Obstet Gynecol. 1991;77(3):425-430. http://www.ncbi.nlm.nih.gov/pubmed/1992411. Accessed December 17, 2016.
- Brunham RC, Gottlieb SL, Paavonen J. Pelvic Inflammatory Disease. Campion EW, ed. N Engl J Med. 2015;372(21):2039-2048. doi:10.1056/NEJMra1411426
- Chappell CA, Wiesenfeld HC. Pathogenesis, Diagnosis, and Management of Severe Pelvic Inflammatory Disease and Tuboovarian Abscess. Clin Obstet Gynecol. 2012;55(4):893-903. doi:10.1097/GRF.0b013e3182714681
- Walker CK, Wiesenfeld HC. Antibiotic Therapy for Acute Pelvic Inflammatory Disease: The 2006 Centers for Disease Control and Prevention Sexually Transmitted Diseases Treatment Guidelines. Clin Infect Dis. 2007;44(Supplement 3):S111-S122. doi:10.1086/511424









