Authors: KC Collier, MD (EM Resident Physician, University of Vermont) and Patricia Yang, MD (EM Attending Physician, University of Vermont) // Reviewed by: Alexander Y. Sheng, MD, MHPE (@TheShenger); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case
A 75-year-old-woman with a history of atrial fibrillation and cirrhosis presents to the emergency department (ED) for chest pain. She is at high risk for bleeding (HAS-BLED score: 3), as well as stroke (CHA2DS2-VASc score: 3) and underwent the WATCHMAN procedure approximately 5 days ago. Triage vitals show a blood pressure of 83/62 and heart rate of 107. Physical exam is significant for distended neck veins, increased work of breathing and 2+ pitting edema on the bilateral lower extremities to the mid-shin.
Introduction
Atrial fibrillation is the most common sustained cardiac arrhythmia and affects more than two million people aged 40 years and older in the United States.1 The prevalence of atrial fibrillation increases with age. Given the United States’ aging population, the number affected will continue to rise at a substantial rate. Patients with atrial fibrillation are at elevated risk of thromboembolic disease. Stroke is the leading cause of long term disability and the third leading cause of death in patients with atrial fibrillation.1 Conversely, 20% of all strokes are thought to be related to embolization from atrial fibrillation and 90% of these emboli originate from the left atrial appendage (LAA).1 The trabeculated surface and low flow state of the LAA make it prone to thrombus formation, particularly in the setting of uncoordinated cardiac contraction.1 These intracardiac emboli are larger and their effects more devastating than atheromatous emboli.1
A primary goal of treatment for atrial fibrillation focuses on reducing risk factors for thrombus formation and stroke. Traditionally, this is achieved through systemic anticoagulation agents such as warfarin and, more recently, direct acting oral anticoagulants. However, these anticoagulants introduce their own risks and logistical challenges, most importantly clinically significant hemorrhage. LAA closure devices, such as the WATCHMAN device, offer an alternative in those patients who are poor candidates for long term anticoagulation.
Left Atrial Appendage Closure Procedure
LAA closure can be performed percutaneously or by open surgical approach. Surgical closure techniques include amputation of the LAA and closure, via stapler closure, or closure with a surgical occlusion device.2 For patients already undergoing open cardiac surgery, it is recommended that they have prophylactic LAA closure if they have a CHA2DS2-VASc score of 2 or greater.2 Percutaneous LAA closure is indicated for patients with atrial fibrillation with elevated risk of stroke based on CHA2DS2-VASc score, and a contraindication for long term anticoagulation.2 It is important to note that patients who require anticoagulation for other reasons, for example atrial fibrillation due to valvular disease or those with a mechanical valve are not candidates for LAA closure.2
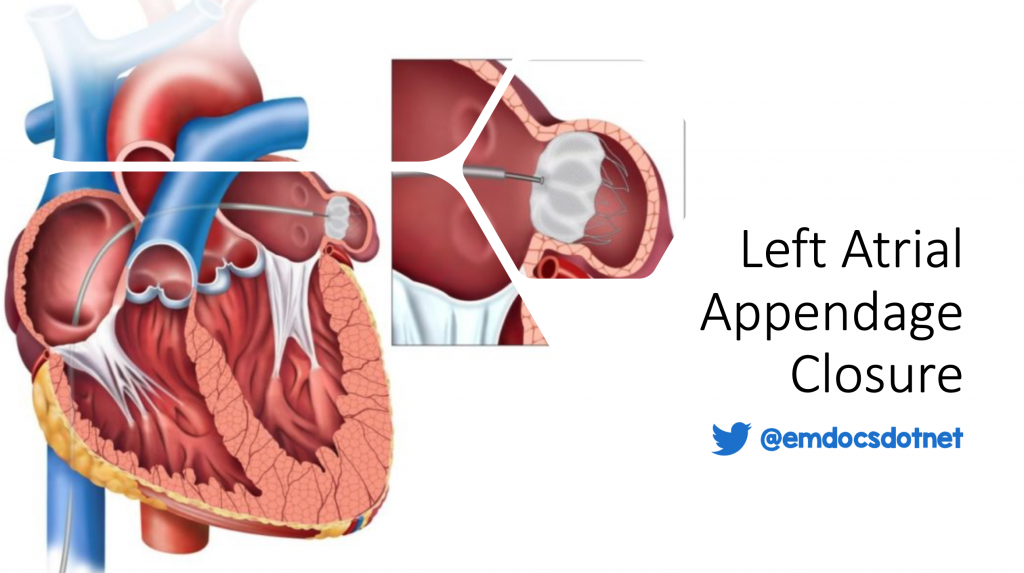
There are three different percutaneous closure devices: WATCHMAN, Amplatzer, and WaveCrest. The WATCHMAN device is the only FDA approved device in the United States.2 This device seals the ostium of the LAA via a self-expanding metal frame covered with a permeable polyethylene membrane, which then endothelializes over the next several months.1 The device is placed by a femoral approach through the inferior vena cava to the right atrium, where a transseptal puncture is performed to deliver the device to the left atrium.2 This is performed under fluoroscopic and transesophageal echocardiographic visualization.3 There is also a less commonly used transpericardial approach.2
Short term antithrombotic therapy is recommended after the WATCHMAN procedure.2 The PROTECT AF trial protocol used warfarin with a target INR of 2-3 or a DOAC plus aspirin (81-325mg) daily for 45 days, followed by clopidogrel 75mg plus aspirin (81-325mg) daily for six months, then daily aspirin (81-325mg) alone indefinitely.2
Complications
Placement of the WATCHMAN device is relatively well tolerated. However, emergency physicians should be aware of a few related complications. The thin wall of the LAA can be easily perforated during device delivery, leading to pericardial effusion, the most common complication.1,4 The severity of these effusions ranged from minor to those causing tamponade physiology. The rate of serious pericardial effusion within 7 days of device implantation ranged from 5% in the PROTECT AF cohort to 2.2% in the CAP registry.11 There are few case reports of these events presenting up to weeks following the procedure.5 It is thought that late presenting pericardial effusions are secondary to “microperforations” that occur during device implantation and postprocedural anticoagulation.5
The incidence of device related thrombus formation was 4.2% among the combined PROTECT AF trial and CAP registry.11 Risk factors associated with thrombus formation on the device included older age, personal history of stroke, larger device size, and the chronic subtype of atrial fibrillation.6,7 Thus, short-term anticoagulation is recommended in order to reduce the risk of thrombus formation, as well as resolve existing thrombi.6,7

Evaluation
History and physical examination: It is helpful to know when and where the procedure was performed. If possible, obtain a procedural report to gain a better sense of what was done and if there were any complications. It is also important to inquire about anticoagulation use. If the patient is not on any periprocedural anticoagulation, a thrombus is more likely.
Imaging: Bedside echo can help quickly assess for serious and life-threatening complications including pericardial effusion and tamponade. If the physical exam demonstrates focal neurological deficits, patients should be evaluated for hemorrhagic or thromboembolic stroke. Most institutions have protocolized stroke workups that include a head CT without contrast, CT angiography, and MRI head with and without contrast. It is important to remember that most of these patients will be on post-procedural short-term anticoagulation and that they are still at risk for intracranial hemorrhage. A CTA of the chest and abdomen can help determine if device migration has occurred. It is also useful to rule out other pathologies on the differential including hemorrhagic pericardial effusion and aortic dissection.9
Management
Although the lack of familiarity with the WATCHMAN procedure can be a source of trepidation, many of its complications can be effectively managed in the ED.
For bleeding complications, prepare to transfuse with a type and screen. A CBC may also be helpful to determine a baseline and might show a low hemoglobin depending on the time course of the bleeding. Assess for overt signs of bleeding at the access site, usually the femoral vein, and apply direct pressure.
In cases of cardiac tamponade, pericardiocentesis is required. Ideally, this is performed by interventional cardiology and/or cardiothoracic surgery in the catherization lab or operating room, where a pericardial window can be placed to prevent recurrence of the pericardial effusion. However, in cases of hemodynamic instability or patient decompensation, the pericardiocentesis may be performed in the ED using ultrasound guidance. In the absence of tamponade, pericardial effusions may be managed with nonemergent pericardiocentesis or observation.
In cases of ischemic stroke, treatment options are more limited. As most patients will be on active periprocedural anticoagulation, TPA would be contraindicated. Endovascular thrombectomy can be considered in consultation with neurology and interventional radiology for large vessel occlusions.
If the device itself has embolized, the patient will likely require surgical retrieval by cardiothoracic or vascular surgery. A case report describes a closure device migrating to the left ventricle that required emergent surgical retrieval.10 It is also possible for the device to become extracardiac and migrate to the aorta.
Case Conclusion
Bedside echo showed a moderate pericardial effusion and right atrial diastolic collapse. Interventional cardiology was consulted and determined that her WATCHMAN device remained in place with a small left atrial perforation causing hemorrhagic pericardial effusion. Due to her hemodynamic instability and clinical signs of pericardial tamponade, she underwent emergent pericardiocentesis with improvement in her clinical status.
Take Home Points
- Left atrial closure devices are an emerging technology for the treatment of patients with atrial fibrillation who have contraindications to systemic anticoagulation. With evidence of improved outcomes and reduced periprocedural complications, they will become more common in the ED patient population.
- Overall, these devices appear to be safe and effective. There exists a limited but growing data set on the associated complications and their management.
- Bedside point-of-care cardiac ultrasound is paramount for identifying the most common complication, pericardial effusion, and to identify the presence of cardiac tamponade.
- Patients should be placed on short term periprocedural anticoagulation due to risk of device related thrombus and stroke.
- In rare cases of device migration, it is essential to involve colleagues in interventional cardiology, interventional radiology, cardiothoracic surgery, and/or vascular surgery.
References/Further Reading
1Schwartz RS, Van Tassel RA, Hara H, Holmes DR. Left Atrial Appendage Closure and Stoke: Local Device Therapy for Cardioembolic Stroke Prevention. In: Textbook of Interventional Cardiology. 8th ed. Philadelphia, PA, PA: Elsevier; 2020:623-633.
2Hijazi ZM, Saw J. Atrial fibrillation: Left atrial appendage occlusion. https://www.uptodate.com/contents/atrial-fibrillation-left-atrial-appendage-occlusion/print#!Published September 2021. Accessed October 30, 2021.
3Perings SM, ed. Percutaneous left atrial appendage closure. Herz. 2011;36(6):551-554. doi:10.1007/s00059-011-3524-6
4Heaton J. Adverse events of subcutaneous loop recorders: Insights from the manufacturer and User Facility Device Experience (maude) database. Journal of invasive cardiology. July 2020. doi:10.26226/morressier.60780408dc2fa1af562468c6
5Nandkeolyar S, Parwani P, Contractor T. A case of delayed hemorrhagic effusive-constrictive pericarditis after left atrial appendage occlusion device placement. JACC: Case Reports. 2019;1(1):27-31. doi:10.1016/j.jaccas.2019.04.003
6Fauchier L, Cinaud A, Brigadeau F, et al. Device-related thrombosis after percutaneous left atrial appendage occlusion for atrial fibrillation. Journal of the American College of Cardiology. 2018;71(14):1528-1536. doi:10.1016/j.jacc.2018.01.076
7Kubo S, Mizutani Y, Meemook K, Nakajima Y, Hussaini A, Kar S. Incidence, characteristics, and clinical course of device-related thrombus after watchman left atrial appendage occlusion device implantation in atrial fibrillation patients. JACC: Clinical Electrophysiology. 2017;3(12):1380-1386. doi:10.1016/j.jacep.2017.05.006
8Boersma LVA, Schmidt B, Betts TR, et al. Implant success and safety of left atrial appendage closure with the watchman device: Peri-procedural outcomes from the EWOLUTION registry. European Heart Journal. 2016;37(31):2465-2474. doi:10.1093/eurheartj/ehv730
9Albani S, Berlier N, Pisano F, Scacciatella P. Left atrial appendage closure device complicated by late-onset pericardial effusion and tamponade: A case report. European Heart Journal – Case Reports. 2021;5(3). doi:10.1093/ehjcr/ytab058
10Gupta P, Szczeklik M, Selvaraj A, Lall KS. Emergency surgical retrieval of a migrated left atrial appendage occlusion device. Journal of Cardiac Surgery. 2013;28(1):26-28. doi:10.1111/jocs.12038
11Reddy VY, Holmes D, Doshi SK, Neuzil P, Kar S. Safety of percutaneous left atrial appendage closure. Circulation. 2011;123(4):417-424. doi:10.1161/circulationaha.110.976449
12Wintgens LIS, Maarse M, Swaans MJ, Rensing BJWM, Van Dijk VF, Boersma LVA. The Watchman left atrial appendage closure device for patients with atrial fibrillation: Current status and future perspectives. Expert Review of Medical Devices. 2020;17(7):615-626. doi:10.1080/17434440.2020.1781615