Authors: Aaron G. Matlock, MD (EM Attending Physician, Brooke Army Medical Center Department of Emergency Medicine, Assistant Professor, Uniformed Services University of Health Sciences) and Brit Long, MD (@long_brit) // Reviewed by: Alex Koyfman, MD (@EMHighAK)
Case
A 45-year-old female presents to the emergency department (ED) with 3 days of progressively worsening headache, confusion, and vision changes. En route, the patient had a 30-second generalized tonic-clonic seizure that resolved after paramedics administered 2 mg of lorazepam. There has been no recent trauma, toxic exposure, or infectious symptoms. The patient does not have a known history of mental illness. The patient has a past medical history notable for systemic lupus erythematosus (SLE), lupus nephritis, and hypertension. She underwent a kidney transplant three years ago. Her medications include tacrolimus, prednisone, mycophenolate, and lisinopril.
Vital signs include Temperature 97.4 F, HR 98, BP 190/105, RR 16, SpO2: 96%. She is awake but confused. She has no external signs of trauma. Her cardiovascular and pulmonary exams are unremarkable. Pupils are 3 mm, equal, and sluggishly reactive. Extraocular movements are intact. Visual fields are difficult to fully assess, but the patient did appear to have some degree of global visual impairment. Cranial nerves are otherwise intact, and she is able to move all extremities on command with intact strength. There is no rash.
ED Evaluation: ECG is normal with no ischemia. Point of care glucose is 127, and creatinine is 1.27. The rest of the laboratory results are normal, including VBG and electrolytes. Computed tomography (CT) of the head without contrast reveals hypoattenuation in the subcortical white matter of the parietal and occipital lobes. No acute hemorrhage or mass effect.
ED Course: The patient’s vital signs, exam, and mental status are concerning for hypertensive emergency, and a nicardipine infusion is initiated. The patient is admitted, and the following morning, the patient undergoes an MRI/MRA of her brain, which demonstrates hyperintense signal on T2 and FLAIR sequences localized to the bilateral parietal and occipital lobes, concerning for posterior reversible encephalopathy syndrome (PRES), or also known as reversible posterior leukoencephalopathy syndrome (RIPLS).
An Introduction to PRES/RIPLS
In a 1996 case series [1], the entity most commonly known as PRES was first presented as a clinical syndrome. Its recognition has become more widespread as advanced neuroimaging modalities, in particular MRI, have become increasingly available. The exact incidence of PRES is not known [2], owing in part to etiologic heterogeneity, overlap with other disease processes, and a lack of standardized diagnostic criteria and nomenclature. PRES is perhaps the most widely recognized name for this syndrome, but it has also been called reversible posterior leukoencephalopathy syndrome (RPLS) and a handful of less commonly used names. None of these names accurately reflects the complete spectrum of this disease, as its neurologic manifestations are not exclusively “posterior”, nor are the deficits always “reversible”. [3-5]
Patients with PRES will often present with encephalopathy manifesting on a spectrum from mild confusion to obtundation. Focal neurological deficits may occur. Headache is common, as are visual disturbances, including decreased visual acuity, visual field deficits and cortical blindness. Seizures occur in a majority of patients with PRES and are often the presenting symptom. [5-7] [8]
PRES is associated with a number of conditions, including hypertension, renal disease, preeclampsia, autoimmune disease, and use of immunomodulatory medications. The pathophysiology is not completely understood, but proposed mechanisms involve cerebral endothelial dysfunction and impaired cerebral autoregulation resulting from acutely elevated blood pressure. In both mechanisms, the final common pathway is reduced integrity of the cerebral endothelium and blood brain barrier with resultant vasogenic cerebral edema. [7-12]
If PRES is recognized and treated appropriately, a majority of patients recover without neurologic sequelae. [4, 5] Principles of treatment include parenteral blood pressure management, discontinuation of offending drugs and control of seizures. Emergency physicians must be aware of this disease, as prompt recognition and differentiation from other emergent neurologic processes can allow for timely treatment and improved outcomes.
What causes PRES?
There are several notable theories regarding the pathophysiology of PRES, but the final common pathway in each is endothelial dysfunction, disruption of the blood-brain barrier, and vasogenic edema. Given the heterogeneity of underlying disease conditions associated with PRES, it is likely that different mechanisms are involved at disease onset. [5]
The hyperperfusion theory posits that blood pressure elevations that exceed the autoregulatory mechanism of cerebral blood flow lead to increased vascular leakage. The posterior circulation may be particularly susceptible to these changes, owing to the relative paucity of sympathetic innervation in the posterior fossa. [8] A majority of patients with PRES present with some degree of blood pressure elevation, and treatment of acutely elevated blood pressure is associated with resolution of disease, partly supporting this theory. However, 30% of patients may present without significantly elevated (or normal) blood pressure, and accordingly, the generalizability of the hyperperfusion theory is contested. [7]
Alternately, a cytotoxic and immunologic theories have been proposed, in which circulating exogenous or endogenous chemokines and vasoactive neuropeptides lead to endothelial injury and vascular instability, blood-brain barrier disruption and cerebral edema. This theory is supported by the relatively increased incidence of PRES among patients with comorbid conditions known to cause endothelial dysfunction (e.g., autoimmune diseases, malignancy and sepsis) and among those taking medications known to be potentially toxic to the endothelium. [9]
Who’s at risk?
Multiple comorbid conditions are associated with PRES, including hypertension, renal disease, organ transplantation, autoimmune disease, preeclampsia/eclampsia, malignancy, sepsis immunosuppressive therapy and cytotoxic medications (Table 1). Renal disease and hypertension are present in over half of patients with PRES, and autoimmune disorders are present in about 40%. [3, 6, 13]. Among immunosuppressive and cytotoxic drugs, corticosteroids and calcineurin inhibitors, such as cyclosporine and tacrolimus, are particularly associated with PRES. [12, 14, 15] Among solid organ transplant recipients, the incidence of PRES has been reported as 0.5 – 5%, and bone marrow and lung transplantation may be associated with even higher risk. [14] The prevalence of PRES among patients with eclampsia is quite high, with the prevalence of PRES in this population reported to be 65-100%. [16-18] PRES is also well-described in pediatric patients, and risk factors, clinical features, and radiographic findings are similar to those seen in adults. [10, 19, 20]

Clinical Features
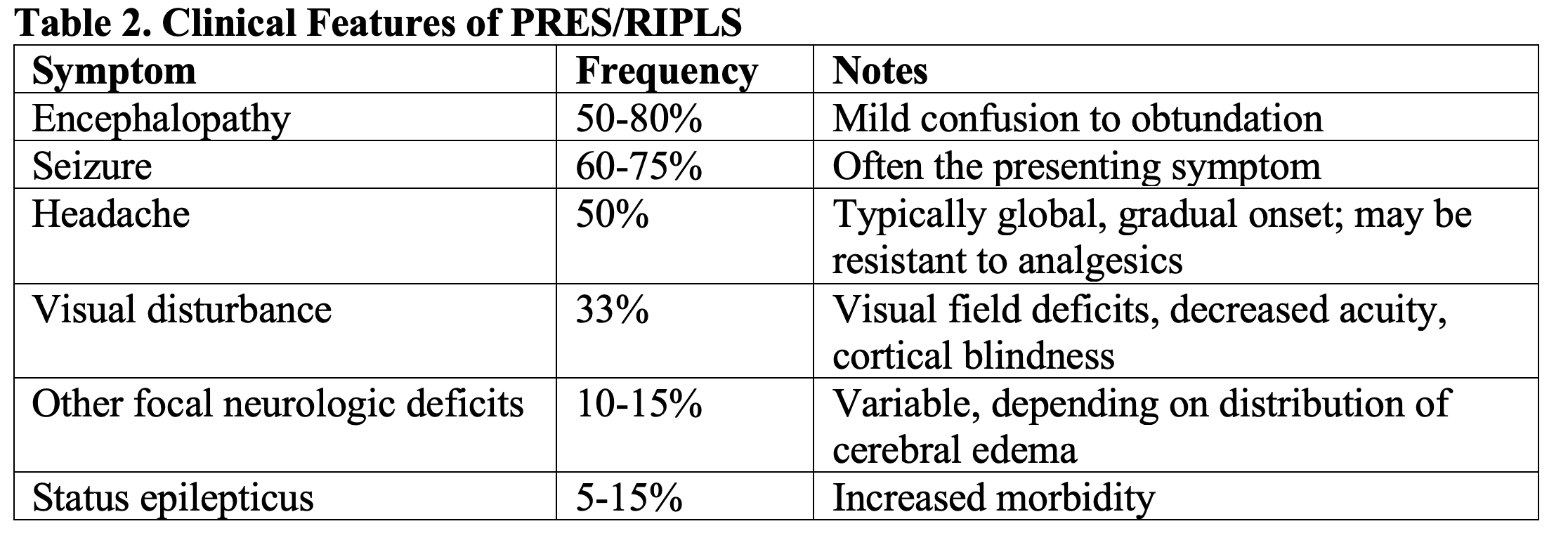
Among patients ultimately diagnosed with PRES, presentations may be variable, but some degree of encephalopathy and altered mental status is present in most. Seizures are often the presenting symptom and occur in a majority of patients. Blood pressure is often elevated on presentation but commonly does not exceed the upper autoregulatory limit. Blood pressure may also be normal on presentation. If prior records are available, a comparison of prior blood pressure readings can provide helpful information in identifying an acute change from baseline values. Clinical features of PRES are summarized in Table 2. [2, 5-8]
Differential Diagnosis
The differential diagnosis for patients with PRES is broad and many emergent etiologies must be considered. Critical considerations include ischemic stroke, intracranial hemorrhage, meningoencephalitis, malignancy, eclampsia, and toxic/metabolic encephalopathy. Hypertensive encephalopathy is unlikely to represent a distinct entity from PRES but rather a semantically different description of the same phenomenon. Diagnosis of PRES is often made after exclusion of other emergent conditions and may not be definitively recognized until an MRI is obtained and demonstrates radiographic findings characteristic of the disease.
Diagnostic Testing
There are no laboratory findings that are specific to PRES. Cerebrospinal fluid, if obtained, may demonstrate increased protein with normal cell counts. [7] Non-contrast CT is commonly the initial neuroimaging study obtained in the ED and may demonstrate findings of vasogenic edema, though it may be normal or non-specific. In one series, the initial head CT was non-specific or normal in over half of patients who were ultimately found to have MRI findings characteristic of PRES. [8, 21] Intracranial hemorrhage has been reported in up to 25% of patients with PRES, though in most instances the area of hemorrhage is small and localized to the areas adjacent to visualized edema. [8, 22] MRI is imaging modality most likely to identify abnormalities consistent with PRES, though there is no true gold standard. [8] The characteristic findings include subcortical and cortical vasogenic edema seen on T2 weighted sequences, such as fluid-attenuated inversion recovery (FLAIR). Edema is often seen in the occipital and parietal lobes, though edema is quite commonly seen in other cerebral regions as well. [8] Clinical features may be discordant with the degree of edema seen on imaging. [2]


Management
ED management of PRES involves management and prevention of seizures and management of blood pressure, though there exist no randomized trials to define the efficacy of specific treatment modalities. Potential offending drugs should be discontinued if possible.
Seizures are treated in the same manner as in other disorders, with benzodiazepines as first-line agents. There is no data to suggest one anti-epileptic drug over another in this condition, but authors have suggested fosphenytoin or levetiracetam as second-line agents for seizure control and prevention. [5, 6, 9] In pregnant patients in whom eclampsia is suspected, magnesium is the initial drug of choice for controlling seizures. [18]
Control of blood pressure is an essential component of treatment of PRES, however specific treatment parameters are not clearly defined in the literature. Authors have suggested that a target blood pressure reduction of 20-30% within the first hour of treatment is reasonable. [5-9] Nicardipine, clevidipine, and labetalol may be used for initial blood pressure management of PRES. Nicardipine and clevidipine are preferred agents, owing to their efficacy, titratability, and minimal effect on heart rate. Labetalol can cause bradycardia and has been shown to be less effective at reaching a targeted blood pressure goal compared to nicardipine. [23] Nitroglycerin and nitroprusside should be avoided in this setting due to their potential for increasing cerebral vasodilation and ICP. [6]

Outcomes
With prompt recognition and treatment of PRES, many patients will recover fully, though persistent neurologic deficits can occur in 10-44%, with delay in initiation of treatment associated with unfavorable outcomes. [4, 8, 24]. Recurrent episodes of PRES can occur, and overall mortality is reported to be in the range of 3-6%. [8]
Back to the case…
The patient was started on a nicardipine infusion with improvement in her blood pressure. Her anti-rejection and SLE medications were held, and nephrology was consulted for further management of her immunosuppressive regimen. Over the next two days, her blood pressure normalized, and her neurologic deficits resolved. Her mental status returned to baseline, and she was transferred out of the ICU two days later.
Take Home Points:
- Emergency physicians should consider PRES in patients presenting with altered mental status, seizures, and neurologic deficits who have risk factors for PRES. Heightened suspicion should be raised in those patients who are on immune-suppressive medications, those with renal disease and acutely elevated blood pressure.
- While non-contrast CT scan may show signs of vasogenic edema, expeditious MRI should be performed, as this is the most sensitive and specific imaging modality for PRES.
- Seizures are often the presenting symptom of PRES and occur in a majority of cases.
- Treatment is focused on seizure management, blood pressure control and removal of offending agents.
- PRES and eclampsia have significant overlap, and management principles are similar, though magnesium is the preferred agent for treatment of seizures in eclampsia.
References/Further Reading:
- Hinchey, J., et al., A reversible posterior leukoencephalopathy syndrome. N Engl J Med, 1996. 334(8): p. 494-500.
- Brady, E., et al., The imaging spectrum of posterior reversible encephalopathy syndrome: A pictorial review. Clin Imaging, 2018. 47: p. 80-89.
- Fugate, J.E., et al., Posterior reversible encephalopathy syndrome: associated clinical and radiologic findings. Mayo Clinic proceedings, 2010. 85(5): p. 427-432.
- Hinduja, A., et al., Predictors of poor outcome in patients with posterior reversible encephalopathy syndrome. The International journal of neuroscience, 2017. 127(2): p. 135-144.
- Liman, T.G., E. Siebert, and M. Endres, Posterior reversible encephalopathy syndrome. Curr Opin Neurol, 2019. 32(1): p. 25-35.
- Thompson, R.J., et al., Posterior reversible encephalopathy syndrome in the emergency department: case series and literature review. West J Emerg Med, 2015. 16(1): p. 5-10.
- Fischer, M. and E. Schmutzhard, Posterior reversible encephalopathy syndrome. J Neurol, 2017. 264(8): p. 1608-1616.
- Fugate, J.E. and A.A. Rabinstein, Posterior reversible encephalopathy syndrome: clinical and radiological manifestations, pathophysiology, and outstanding questions. Lancet Neurol, 2015. 14(9): p. 914-925.
- Granata, G., et al., Posterior reversible encephalopathy syndrome–Insight into pathogenesis, clinical variants and treatment approaches. Autoimmun Rev, 2015. 14(9): p. 830-6.
- Solh, Z., et al., Neurological PRESentations in Sickle Cell Patients Are Not Always Stroke: A Review of Posterior Reversible Encephalopathy Syndrome in Sickle Cell Disease. Pediatr Blood Cancer, 2016. 63(6): p. 983-9.
- Vargas, A. and F.D. Testai, Posterior Reversible Encephalopathy Syndrome in adult sickle-cell patients: Case series and literature review. J Clin Neurosci, 2019. 70: p. 249-250.
- Dhar, R., Neurologic Complications of Transplantation. Neurocrit Care, 2018. 28(1): p. 4-11.
- Ferreira, T.S., F. Reis, and S. Appenzeller, Posterior reversible encephalopathy syndrome and association with systemic lupus erythematosus.Lupus, 2016. 25(12): p. 1369-76.
- Chen, S., et al., Posterior Reversible Encephalopathy Syndrome After Transplantation: a Review. Mol Neurobiol, 2016. 53(10): p. 6897-6909.
- Parikh, N.S., et al., Corticosteroid therapy and severity of vasogenic edema in posterior reversible encephalopathy syndrome. J Neurol Sci, 2017. 380: p. 11-15.
- Brewer, J., et al., Posterior reversible encephalopathy syndrome in 46 of 47 patients with eclampsia. American journal of obstetrics and gynecology, 2013. 208(6): p. 468.e1-468.e4686.
- Wagner, S.J., et al., Posterior reversible encephalopathy syndrome and eclampsia: pressing the case for more aggressive blood pressure control. Mayo Clinic proceedings, 2011. 86(9): p. 851-856.
- Garg, R.K., N. Kumar, and H.S. Malhotra, Posterior reversible encephalopathy syndrome in eclampsia. Neurol India, 2018. 66(5): p. 1316-1323.
- Zama, D., et al., A survey on hematology-oncology pediatric AIEOP centres: The challenge of posterior reversible encephalopathy syndrome.Eur J Haematol, 2018. 100(1): p. 75-82.
- Gavrilovici, C., et al., Posterior reversible encephalopathy syndrome in children with kidney disease. Int Urol Nephrol, 2017. 49(10): p. 1793-1800.
- Bartynski, W.S. and J.F. Boardman, Distinct imaging patterns and lesion distribution in posterior reversible encephalopathy syndrome. AJNR. American journal of neuroradiology, 2007. 28(7): p. 1320-1327.
- Sharma, A., R.T. Whitesell, and K.J. Moran, Imaging pattern of intracranial hemorrhage in the setting of posterior reversible encephalopathy syndrome. Neuroradiology, 2010. 52(10): p. 855-863.
- Peacock, W.F., et al., CLUE: a randomized comparative effectiveness trial of IV nicardipine versus labetalol use in the emergency department. Critical care (London, England), 2011. 15(3): p. R157-R157.
- Legriel, S., et al., Determinants of recovery from severe posterior reversible encephalopathy syndrome. PloS one, 2012. 7(9): p. e44534-e44534.
Disclaimer:
The views expressed herein are those of the author(s) and do not reflect the official policy or position of Brooke Army Medical Center, the U.S. Army Medical Department, the U.S. Army Office of the Surgeon General, the Department of the Army, the Department of the Air Force, or the Department of Defense, or the U.S. Government.
The appearance of name-brand products in this article does not constitute endorsement by Brooke Army Medical Center, the U.S. Army Medical Department, the U.S. Army Office of the Surgeon General, the Department of the Army, the Department of the Air Force, or the Department of Defense, or the U.S. Government of the information, products, or services contained therein.





