Authors: Brandon Carius, PA-C (Emergency Medicine, San Antonio, TX), Rachel Bridwell, MD (EM Resident Physician, San Antonio, TX), and Gillian Schmitz, MD (Attending Emergency Physician, ACEP board member, San Antonio, TX) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case
A 27-year-old F G1P0 at 17 weeks gestation presents with fever, chills, vomiting, and abdominal pain for 1 day. She denies any vaginal bleeding. She has had an uneventful pregnancy and a confirmed IUP on pelvic ultrasound earlier in her first trimester. She is currently on prenatal vitamins and acetaminophen, which is not adequately treating her pain. She has no other medical or surgical history.
On exam, her vitals are: T 38.7 C, HR 125, BP 80/60, RR 16, 98% RA. She appears uncomfortable and dehydrated. She has dry mucous membranes, her abdomen is diffusely tender to palpation, and you appreciate a gravid uterus a few centimeters below the umbilicus. She is tachycardic and crying, asking if her baby is going to be ok.
Background
Sepsis is the fourth leading cause of maternal death in the United States [1-3]. Despite continued refinement of sepsis guidelines, mortality in pregnant patients has nevertheless risen consistently at an average of 9% per year from 2001 to 2010 [1,4]. Despite these trends, there is little consensus regarding diagnostic and therapeutic interventions in pregnant patients with sepsis. A lack of focus on maternal sepsis is in part due to the significantly low proportion of pregnant patients comprising sepsis, and septic shock (as previously defined) occurs as a pregnancy complication in only 0.001% of pregnancies as of 2008 and in 0.002-0.01% of women in the postpartum period [4]. In addition to its relative rarity, all major sepsis trials have used pregnancy as an exclusion criterion, thereby leaving clinical diagnosis and treatment practice to be largely based on expert opinion and individual provider preference or experience.
The presentation of sepsis, a condition based on an altered immune response, can be further complicated by the already altered and dysregulated physiology of pregnancy. Beyond basic assessment, clinicians must be cognizant of the competing priorities of mother and fetus. While treatment of the mother is the priority, the altered physiology of pregnancy can alter medication regimens, and medication effects on the fetus can affect the mother as well.
The Pregnant Body: Shifting Homeostasis
The changes of normal pregnancy must be understood in order to approach the dysregulated immune response that can occur with sepsis. Physiologic changes occur secondary to anatomic changes with fetal growth and uterine enlargement, as well as altered hormonal levels that begin with conception and continue after delivery. Normal changes in pregnancy include significant physiologic shifts. Expanding plasma and red blood cells increase intravascular volume, creating a physiologic anemia. Additionally, rise in respiratory tidal volume increases minute ventilation and thus a baseline respiratory lean towards alkalemia.
The cardiovascular system changes throughout pregnancy. Early in the first trimester, systemic vasodilation occurs. A drop of up to 35-40% of systemic vascular resistance (SVR) begins in the first trimester and plateaus in the middle of the second trimester, while a compensatory increase in heart rate occurs to maintain cardiac output [5]. Heart rate peaks in the late third trimester and has been shown, on average, to be approximately 17-24% higher than the patient’s prepartum baseline [5,6]. However, throughout this time, studies have shown significant variations both in the timeline and relative shift in heart rate, with increases of 10 to 30 beats per minute above prenatal resting heart rates [7-9]. It is important to note additional cardiac variability in the pregnant patient carrying multiple fetuses, with research finding a further increase in heart rate compared to a single fetus pregnancy [10]. In the postpartum period, these changes resolve generally within two weeks, although a small proportion of patients maintain their pregnant cardiovascular measures at 12 weeks postpartum [11,12]. Increases in plasma volume and red blood cell mass occur soon after pregnancy begins, generally within the first 6-12 weeks [13]. These increases continue gradually until around 30 to 34 weeks of gestation, maintaining a steady level for the duration of the pregnancy. Increases in plasma volume and red blood cell mass further offset decreased SVR during pregnancy to maintain normotension. Blood pressure may fall, however, by 10-15 mmHg in a normal pregnancy and nadir around 28 weeks gestation.
Pregnancy creates significant shifts in immune regulation as well. Leukocytosis occurs during pregnancy, and some studies demonstrate that white blood cell (WBC) levels double in the same patient during the pregnancy period compared to postpartum levels [14]. The proposed mechanism for this increase is both a generalized inflammatory state and decreased WBC apoptosis [15-18]. WBC counts can reach 25,000/mm3during the course of normal healthy pregnancy [19]. Likely due to the physiologic stress of delivery, WBC counts have also been found to increase from levels as low as 8-10,000/mm3to as high as 25,000/mm3in the hours immediately following delivery [15]. Further complicating the interpretation of leukocytosis in pregnancy is research demonstrating that WBC counts can acutely spike in the setting of preeclampsia [20,21]. Coupled with cardiovascular changes and a generalized inflammatory response, concerns for preeclampsia in the emergency setting may obscure subtle hints of isolated or concomitant infection when assessing leukocytosis. Further normal physiologic changes in pregnancy are highlighted in table 1.

Physiologic changes in pregnancy may become additionally complicated with increasing success in medical management of chronic conditions. Increasing rates of obesity, type 2 diabetes mellitus, and hypertension in pregnancy have resulted in the need for increasing rates of stabilizing medications [22, 23]. Retrospective studies have found rate increases as large as 60% for prescription medication use in pregnancy over the last 30-40 years [24]. The use of antihypertensives and glucose-controlling medications alter the physiologic baseline of pregnant patients and may conceal changes that can occur in the setting of infection. These medications can mask systemic response markers to the clinician beyond the distortion that can be seen in a pregnant patient without comorbidities. A dearth of data describing rates of medication noncompliance in pregnant patients with chronic medical conditions further clouds the clinical picture. Elevated glucose and blood pressure could obfuscate appropriate references ranges in a patient presenting with acute illness.
The Evolution of Sepsis: Issues with Diagnosis and Guidance
Sepsis has evolved significantly over time from its initial characterization nearly 30 years ago. The previous use of systemic inflammatory response syndrome (SIRS, table 2) criteria, combined with a suspected source of infection, was critical to the identification of sepsis. Although the studies generating these criteria did not include pregnant patients, and therefore did not consider pregnancy physiology, their metrics provided the only standardized assessment tool for sepsis recognition. Prior to the second sepsis updates in 2012 however, retrospective reviews demonstrated that SIRS and the sepsis guidelines did not accurately identify maternal sepsis, appropriately identifying less than two-thirds of obstetric patients [25].
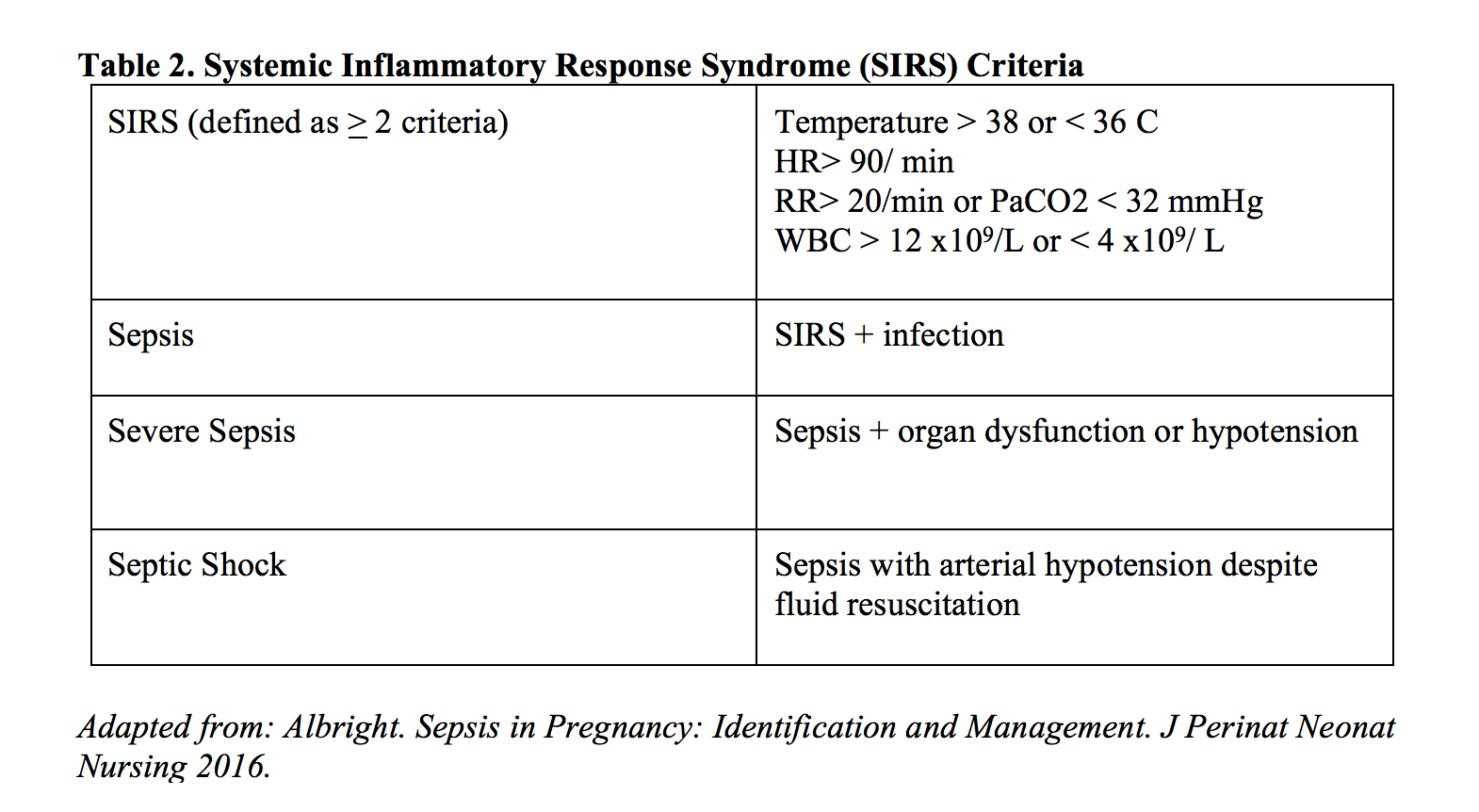
Working in parallel to the surviving sepsis campaigns, criteria aimed at identifying maternal sepsis have been largely clouded with definitions by proxy. Until recently, the broader category of maternal sepsis had been largely pigeonholed as puerperal sepsis under the World Health Organization (WHO). This wording confined pregnant or postpartum sepsis to only infections of the genitourinary tract in the period between the rupture of membranes and the end of the 6-week postpartum period [26,27]. The WHO additionally defined septic abortion, but this definition likewise remained isolated to genitourinary tract infections [27,28]. Therefore, most studies of maternal sepsis focused solely on the diagnosis and treatment of genitourinary-based sepsis rather than a more generalized sepsis encompassing infectious sources from all systems that was studied in non-pregnant populations.
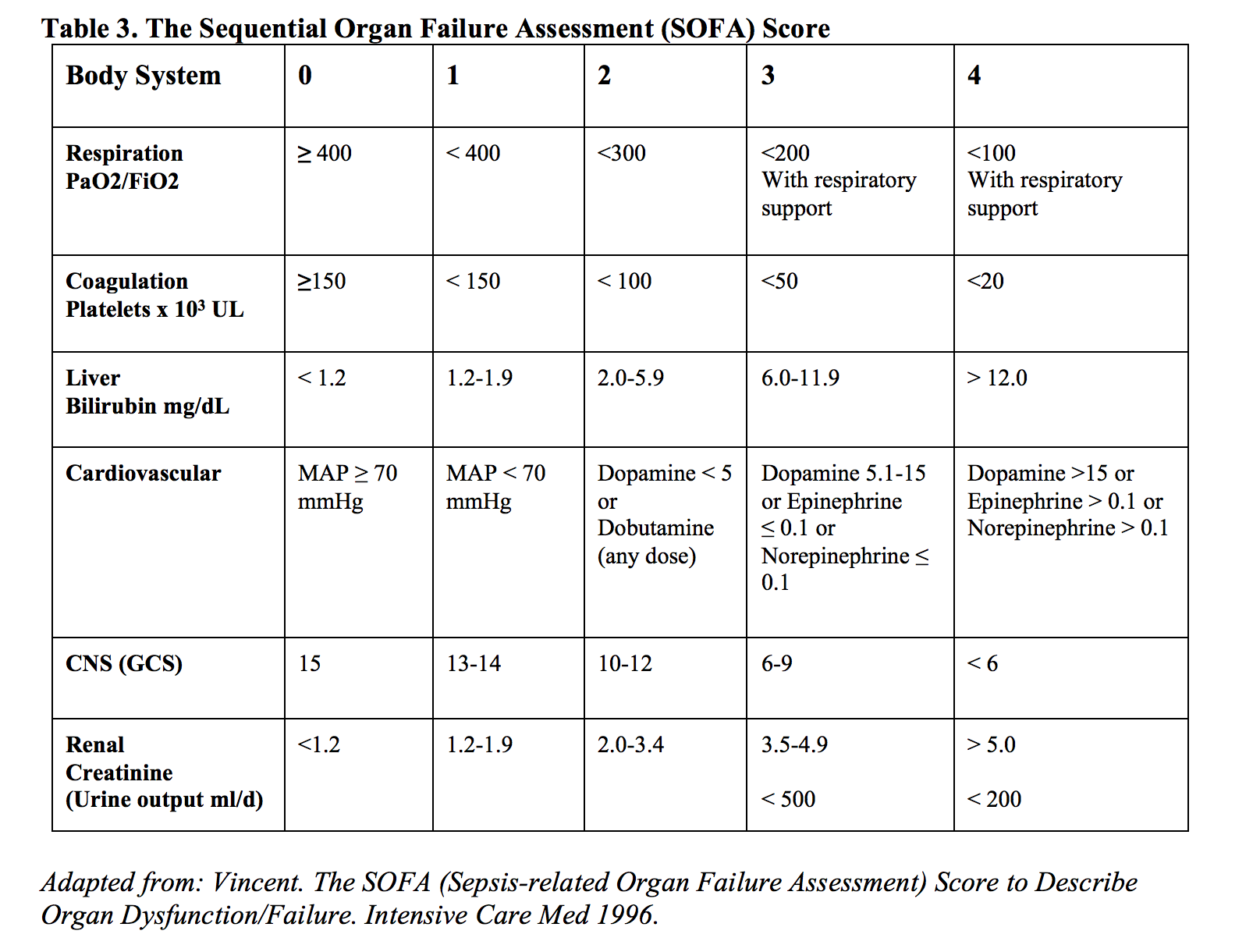
Using SIRS criteria as the clinical foundation for sepsis, diagnoses were most recently supplemented in the Third International Consensus Definitions for sepsis and septic shock in 2016 by the Sepsis-related Organ Failure Assessment (SOFA) and the quick Sepsis-related Organ Failure Assessment (qSOFA), defined in tables 3 and 4. Like the majority of sepsis before it, the trials that established and validated the SOFA and qSOFA scores excluded pregnant patients from the sample population, thereby undercutting its application in pregnant patients. As of this review, there has been no external validation of applying SOFA or qSOFA scores in pregnant patients [26], even though the variables that constitute these scores have been validated in various combinations in pregnant populations.

The establishment of the modified early warning scoring systems (MOEWS) and the sepsis in obstetrics (SOS) scores were created as an attempt to better describe and stratify maternal patients concerning for sepsis. However, validation attempts have yielded mixed results. The MOEWS has numerous international variants (table 5), and thus simply applying the term MOEWS does not appropriately identify regional variations. Overall, MOEWS is limited because its intent is “to help detect the early signs of illness and trigger timely medical review with appropriate intervention,” rather than specifically target sepsis [29]. The major validation study of MOEWS in 2015 analyzed a total 913 cases of chorioamnionitis, but only 5 cases met the definition of severe sepsis [30]. Originally designed to predict severe sepsis by 2.0 guidelines, MOEWS restricts its utility not only by using a now outdated term, but also with its singular focus on chorioamnionitis rather than all causes of pregnancy in sepsis.

Similar to the MOEWS, the SOS was introduced in 2014 as a means to establish an obstetric-focused scoring system that incorporated many of the altered baselines for cardiovascular, respiratory, and immune system functions that were previously discussed (table 6) [31]. The authors acknowledged that there were no standardized criteria for ICU admission for sepsis at the hospital where the original study was performed. Most recently, a single prospective validation trial of the SOS conducted in the United States examined its performance as a predictor of maternal ICU admission specifically for sepsis [32]. This prospective trial found that the SOS score cut-off of less than 6 points had a 64% sensitivity and 98.6% negative predictive value, while a score greater than 6 demonstrated a specificity of 88%. This poor sensitivity is a significant limitation. Furthermore, of the 1,250 pregnant or postpartum patients who presented to the ED in the approximately three-year study period, only 14 were admitted to the ICU [32]. Therefore, while this sole prospective validation study supports the negative predictive value of the SOS, greater validation and a larger sample population are needed.

Although increasing focus has established the foundations of obstetric-focused scores for sepsis in recent years, emergency clinicians are nevertheless still without substantially validated systems to back ED decision-making or hospital admission when confronted with a sick pregnant or postpartum patient.
Key Points:
- All major sepsis trials have excluded pregnant patients.
- Major physiologic changes in pregnancy: increased minute ventilation, decreased SVR, increased HR, and general increase in CO.
- Hematologic changes: dilutional anemia and physiologic leukocytosis.
- Pregnancy specific sepsis scoring are MOEWS and SOS, though they require further external validation.
Treatment Considerations Specific to Pregnancy
Pyelonephritis
The presence of pregnancy alone automatically designates pyelonephritis as complicated, requiring intravenous antibiotics and admission for continued monitoring of both mother and fetus [33,34]. Pyelonephritis accounts for the largest proportion of maternal inpatient admissions, occurring in approximately 2% of pregnancies in the United States, with up to 20% occurring in the second and third trimester [34-36]. Dilation of renal calyces secondary to progesterone, stagnation of ureteral peristalsis, mechanical compression of the bladder, and increased glomerular filtration rate (GFR) resulting in glucosuria and alkaluria facilitating bacterial growth are all factors that predispose pregnant women to pyelonephritis [37].
A recent 18-year retrospective analysis found that acute pyelonephritis in pregnancy significantly increased the risk of maternal anemia, acute renal failure, respiratory distress, and preterm birth (4). Additionally, patients with maternal pyelonephritis were discovered to have a 33% increased risk of chorioamnionitis, further predisposing them to sepsis [38]. Greater than 80% of acute pyelonephritis cases in pregnancy are from E. coli, but other uropathogens include Klebsiella, Streptococcal spp, Proteus mirabilis, and Enterococcus [38,39]. Pregnant patients were specifically excluded from IDSA guidelines, but based on multiple reviews, ceftriaxone, cefepime, or ampicillin plus gentamicin are all feasible treatment options in treating these common pathogens and considerations for both maternal and fetal safety and side effects [38,39]. In patients less than 24 weeks estimated gestational age, ceftriaxone has been shown to have equal efficacy in length of hospitalization and days until resolution of infection [40]. Ceftriaxone should be avoided in the periparturition period, however, due to the risk of neonatal kernicterus. Additionally, urine culture and local resistance patterns should guide empiric therapy.
In immunocompromised patients or those with severe pyelonephritis impairing urinary drainage, carbapenems or piperacillin-tazobactam should be considered for broader coverage, though imipenem should be avoided if possible due to adverse fetal effects demonstrated in vivo. E. coli and other gram-negative rods cause the vast majority of pyelonephritis in pregnancy, and their endotoxins have the potential to cause large-scale endothelial cell damage in capillary beds. This destruction has been shown to specifically target renal and pulmonary tissue, generating acute respiratory distress syndrome (ARDS) in 1-8% of cases, further complicating the maternal patient [37]. Unlike the non-pregnant urosepsis population, test of cure is required in maternal patients with negative urine cultures following clinical resolution.
Key Points:
- Pregnant patients with pyelonephritis should be admitted for IV antibiotics.
- In pyelonephritis in pregnancy, up to 8% will develop ARDS.
- Do not use ceftriaxone in treating peripartum pyelonephritis to avoid kernicterus in the neonate.
Appendicitis
The incidence of appendicitis in pregnancy is 1 in 1500, which peaks in the second trimester, as compared to the non-pregnant population with an incidence of 3.5 in 1500 [41-43]. However, 1 in 1000 pregnancies undergo surgical evaluation for possible appendicitis with little option for non-operative management due to increased perforation risk as well as mortality [41,42]. Maternal mortality secondary to appendicitis is 4%, and complications of perforated appendicitis result in an estimated 43% fetal mortality rate [44,45].
The constellation of physiologic changes of pregnancy and typical presenting signs and symptoms of appendicitis make maternal diagnosis particularly challenging. Anatomically, the fundus rises and displaces the appendix from the right lower quadrant (RLQ). This abdominal shift and fundal displacement of the omentum can prevent sealing of an inflamed appendix [45a]. Given this shift, only 75% of maternal appendicitis cases present with RLQ pain and tenderness, while another 20% of cases present with RUQ pain [46]. However, up to 45% of these cases present with rectal tenderness which is not commonly thought to be associated with or examined in a patient with suspected appendicitis [46]. Rectal tenderness could therefore make the clinician more suspicious appendicitis, as well as rectal-centric conditions, such as hemorrhoids, rectosigmoiditis, colitis, diverticulitis, or other perianal or perirectal disease. The usual nausea and vomiting commonly seen with appendicitis can likewise become confused with hormonally-induced symptoms. It is therefore important to note any new or significant shifts in the “normal” course of “morning sickness” symptoms during the history in pregnant patients. Additionally, as discussed earlier, physiologic leukocytosis of pregnancy further obfuscates the presence of an underlying pregnancy-based shift in homeostasis or a true immune response to infection. Interestingly, the presence of mild bilirubinemia greater than 1.0 mg/dL has a sensitivity of 70% and specificity of 86% in evaluating for perforation in appendicitis [47].
Imaging presents an additional challenge in maternal appendicitis diagnosis. While ultrasound (US) is safe in pregnancy, wide variation in appendiceal location makes true location and evaluation difficult and likewise widely variable in confidence. Sensitivity and specificity of US for the diagnosis of maternal appendicitis ranges from 67% to 100% and 83 to 95%, respectively. This is significantly less favorable for diagnosis, or at least rule-out, compared to non-pregnant populations where sensitivity and specificity are around 86% and 96%, respectively [48]. In cases where ultrasound is equivocal, magnetic resonance imaging (MRI) is employed, sparing ionizing radiation to both mother and fetus. A meta-analysis of MRI in the diagnosis of maternal appendicitis revealed a sensitivity of 96.8% and a specificity of 99.2% based on 12 studies to include 933 patients, and demonstrated that MRI can also provide information regarding peri-appendiceal findings, which can identify the etiology of abdominal pain [49]. Additionally, MRIs are routinely run without gadolinium contrast based on the hypothetical risk to the fetus but can be employed if critical [50].
Key Points:
- While appendicitis most commonly presents with RLQ, 20% of cases present with RUQ pain.
- Pregnant patients with appendicitis may present with rectal pain.
- If other options exist, CT should be avoided for diagnostic imaging. US has sensitivity of 67-100% and MRI can be used in cases where US is equivocal.
Pelvic Inflammatory Disease
While one of the major complications of pelvic inflammatory disease (PID) is infertility, PID-induced maternal sepsis, although rare, is associated with high-mortality for both mother and fetus, as well as increased risk of preterm delivery [54]. While uncommon, PID in pregnancy presents more commonly in the first trimester with fever and abdominal pain, adnexal tenderness, and cervical motion tenderness, while TOA can present similarly to an ectopic pregnancy with adnexal tenderness, the presence of adnexal mass, and fever. Bacteria can ascend prior to the mucus plug sealing off the decidua around 12 weeks [51]. PID can rapidly progress to tubo-ovarian abscess (TOA), with a mortality up to 9% [52]. The presentation of fever, leukocytosis, and diarrhea should prompt consideration of TOA, independently predicted by elevated CRP and CA-125 levels, though the latter has little utility in the ED [53]. According to the IDSA guidelines, pregnancy with PID requires hospitalization for treatment, which confers its own risks to which their non-pregnant counterparts may not be subjected [54]. Doxycycline, the mainstay treatment for PID per IDSA guidelines, has been repeatedly proven to have severe teratogenicity and therefore cannot be used in the pregnant patient [54]. Azithromycin (1 gram orally each day) should be substituted, in conjunction with an intravenous second-generation cephalosporin, such as cefotetan or cefoxitin [54]. Fortunately, penicillin cross-reactivity with second generation cephalosporins is negligible, providing a wide in-patient treatment strategy [55,56]. This regimen also covers M. genitalium, accounting for up to 8.7% of the non-chlamydial and non-gonococcal PID cases [57].
Key Points:
- Pregnant patients with PID require admission.
- Doxycycline should be avoided. Treatment is intravenous second-generation cephalosporin, such as cefotetan or cefoxitin, plus azithromycin.
Endometritis
Endometritis occurs with the ascension of bacteria during labor that colonize amniotic fluid and decidua, presenting with a postpartum fever, tachycardia, and foul lochia or malodorous vaginal discharge [58]. Cases are generally polymicrobial, with two-thirds containing both anaerobic bacteria (Bacteroides,Clostridium, and Peptostreptococcus spp.) and aerobic bacteria (Group B Streptococcus, E.coli, and enterococcus) [59]. However, the presence of hematoma brings S. pyogenesand S. aureusinto the differential and the concern for toxic shock syndrome [41]. Intravenous gentamicin and clindamycin demonstrate a 95% response rate, though this regimen does not cover enterococcus [60]; Doxycycline plus cefoxitin or ampicillin/sulbactam is an additional regimen. In those who do not respond within the first 48-72 hours, ampicillin is added to empirically cover for these pathogens [62]. In those delivering via cesarean section who then develop endometritis, parametrial cellulitis with phlegmon formation in the broad ligament, or less-commonly parametrial phlegmon, can cause persistent fevers and require immediate consult to interventional radiology for guided drainage [62]. Additionally, venous drainage post-cesarean section can also spread infection, generating septic pelvic thrombophlebitis [63].
Presenting from 5 days to 3 weeks postpartum, pelvic thrombophlebitis is usually refractory to broad-spectrum antibiotics alone and requires heparinization along with broad polymicrobial coverage prior to clinical improvement [63-65]. While this particular complication requires a high index of suspicion, liberal use of postpartum computed tomography (CT) has a significant impact on management; in a retrospective cohort study of 238 postpartum patients, the use of CT changed management, with alteration of antibiotic therapy in 10%, addition of low-molecular weight heparin in 12%, and surgical intervention in 17%. In total, this study demonstrated that approximately 40% of patients had significant alteration to their clinical course by the addition of CT [66].
Key Points:
- In febrile postpartum patients refractory to antibiotics and resuscitation, consider septic thrombophlebitis, requiring anticoagulation.
Resuscitation in Pregnancy
Although treatment of the pregnant patient involves the treatment of two patients, the mother’s condition is the priority; stabilizing the hemodynamics in the mother generally improves that of the fetus [68]. Initial resuscitation should include intravenous fluid administration, and positioning should be optimized. The left lateral decubitus position optimizes the hemodynamics of the pregnant patient, improving preload since the fetus is displaced from lying atop the inferior vena cava. Fluid resuscitation should begin within the first three hours of presentation if either hypotension or lactic acid >4 mmol/L are present [69]. However, due to increased blood volume in pregnancy, a lactic acid threshold of 4 mmol/L may lack sensitivity in septic pregnant patients if the level is above a normal limit but still beneath this standard. In a retrospective cohort of 159 septic pregnant patients, the mean lactic acid level of those admitted for ICU level care was on average only 2.6 mmol/L, and those with positive blood cultures had a level of 2.2 mmol/L [70]. This study found increased morbidity with elevated lactic acid, with an adjusted odds ratio of 2.34 for per 1 mmol/L increase in lactic acid [70].
Hypotension despite these interventions requires vasopressor administration; however, no specific guidelines exist for pregnant patients. There is no explicit recommendation for a mean arterial pressure goal for sepsis in pregnancy, thus 65 mm Hg remains the resuscitation goal, though fetal monitoring can provide further feedback for titration [71]. The 2016 Society of Critical Care Medicine guidelines do not offer recommendations tailored for the pregnant patient, though their current data support the use of norepinephrine as the first line vasopressor in pregnant septic patients [67-69]. Animal models have demonstrated that maternal endogenous norepinephrine causes dose-dependent vasoconstriction of uterine vessels, though some in vitro models using human uterine arteries elicit the specific ability of this artery to accumulate, metabolize, and remove norepinephrine [72-73]. Due to the paucity of data, there is scant evidence to suggest that administration of norepinephrine causes a negative outcome for the fetus [74].
The choice for second-line vasopressor also remains controversial, extrapolated from controlled studies with spinal anesthetics in place. Phenylephrine and ephedrine are often used as second-line agents since they both have been shown to increase maternal blood flow [69, 75-76]. Unlike ephedrine, phenylephrine does not alter the fetal acid-base status, though its sole alpha stimulation generates maternal reflex bradycardia and thus diminished cardiac output [76-77]. In comparison, ephedrine does not generate bradycardia, though its indirect action to release pre-existing maternal catecholamines may prove less efficacious in a septic patient who has already exhausted her endogenous stores [76-78].
These data are from Cesarean deliveries, many of which are elective, and thus the data could be skewed by significant maternal endogenous catecholamine stores in a non-exhausted patient who also has optimized hemodynamics in a planned operative environment. Conversely, a pregnant patient in septic shock likely has expended much of her cardiovascular reserve as well as the vast majority of her endogenous catecholamines. Additionally, both phenylephrine and ephedrine have known tachyphylaxis associated with their administration, further complicating their use [76]. In their Task Force on Obstetric Anesthesia, the American Society of Anesthesiologists recommended phenylephrine over ephedrine because of the preferred fetal acid-base status, though this decision was made for uncomplicated pregnancies [79]. While this choice was echoed by UK and Canadian counterpart agencies, the extrapolation of hemodynamics of spinal anesthesia for uncomplicated pregnancies to septic shock in pregnancies remains to be seen with the dearth of data currently present.
While an uncommon case, myocardial dysfunction may occur secondary, most likely due to septic myocarditis. In this setting, dobutamine is the preferred inotrope (78). Despite its very limited use in the non-pregnant septic population, administered in approximately 1% of the patients in the ProCESS trial, dobutamine presents a viable option for inotropy in pregnant patients already on vasopressors and fluid resuscitation [79-80]. Based on previous ovine models, dobutamine provides inotropy in pregnant sheep, although it decreases uterine blood flow [81]. Further study is required in human patients.
Other treatment considerations in pregnant patients include glucose control and venous thromboembolism (VTE) prophylaxis. Maternal hyperglycemia can directly cause fetal hyperglycemia and ultimately acidosis, decreasing uterine blood flow and lowering fetal oxygenation [71]. With this in mind, the recommendation is to maintain maternal blood glucose less than 180 g/dl, though this may require additional adjustment for gestational diabetic patients in labor, for whom a blood glucose of 80-120 g/dL is recommended [71]. Blood glucose ranges likely required individualization in a laboring, septic patient with additional comorbidities.
Pregnancy alone confers a 5-fold increased risk of deep vein thrombosis (DVT) as compared to the non-pregnant population [82]. In individuals in septic shock on guideline recommended VTE prophylaxis, there was a 37% incidence in VTEs despite these prophylactic interventions; taken together, septic pregnant patients are at an extremely high risk of VTE [83]. The current recommendation is both intermittent compression devices and either daily low molecular weight heparin or 2-3 times daily administration of unfractionated heparin in these patients who do not have obvious contraindications [71]. NOACs are not currently recommended.
Key Points:
- For most patients, resuscitate with 30 ml/kg of crystalloid and even mild elevated lactate can portend a poor prognosis.
- While there are no specific vasopressor guidelines for sepsis in pregnancy, norepinephrine is first choice.
- VTE prophylaxis in sepsis consists of low molecular weight heparin or unfractionated heparin with sequential compression devices.
Continuing to Search for Answers
The anatomic and physiologic changes that occur during pregnancy pose a challenge in early recognition and management of sepsis. Current guidelines that identify the presence and severity of sepsis were extrapolated from randomized control trials that specifically excluded pregnant patients. New guidelines with risk stratification for pregnancy patients have been developed but are still in early stages with little validation for widespread use. Further research and validation are needed to help properly recognize and treat this small but significant population effectively for best outcomes for both mother and fetus.
Take Aways:
- Keep a high index of suspicion for sepsis in pregnancy, as current sepsis guidelines do not necessarily predict disease nor its severity well.
- Physiological and anatomic changes in pregnancy can mask the presentation of sepsis in pregnancy.
- Pyelonephritis accounts for the greatest number of in-patient admissions in pregnancy.
- Resuscitate with early and targeted antibiotic as well as 30 cc/kg of crystalloids, with norepinephrine as the first line pressor.
- VTE prophylaxis is required with the confluence of 2 highly hypercoagulable states.
References/Further Reading:
- Albright CM, Mehta ND, Rouse DJ, Hughes BL. Sepsis in Pregnancy: Identification and Management. J Perinat Neonat Nursing. 2016;30:95-105.
- Chang J, Elam-Evans LD, Berg CJ, Herndon J, Flowers L, Seed KA, et al. Pregnancy-related mortality surveillance—United States, 1991–1999. MMWR Surveill Summ 2003;52:1–8.
- van Dillen J, Zwart J, Schutte J, van Roosmalen J. Maternal sepsis: epidemiology, etiology and outcome. Curr Opin Infect Dis 2010;23:249-54.
- Cordioli RL, Cordioli E, Negrini R, Silva E. Sepsis and pregnancy: do we know how to treat this situation? Rev Bras Ter Intensiva. 2013 Oct-Dec;25(4):334-44.
- Meah VL, Cockcroft JR, Backx K, Shave R, Stohr EJ. Cardiac output and related haemodynamics during pregnancy: a series of meta-analyses.Heart. 2016;102(7):518.
- Gabbe SG, Niebyl JR, Galan H, et al. Obstetrics: normal and problem pregnancies, 6th ed. Philadelphia: Elsevier/Saunders; 2012.
- Robson SC, Hunter S, Boys RJ, Dunlop W. Serial study of factors influencing changes in cardiac output during human pregnancy. Am J Physiol. 1989;256(4):H1060.
- Clapp JF 3rd, Capeless E. Cardiovascular function before, during, and after the first and subsequent pregnancies. Am J Cardiol. 1997 Dec;80(11):1469-73.
- Mahendru AA, Everett TR, Wilkinson IB, Lees CC, McEnjery CM. A longitudinal study of maternal cardiovascular function from preconception to the postpartum period. J Hypertens. 2014;32(4):849.
- Kametas NA, McAuliffe F, Krampl E, Chambers J, Nicolaides KH. Maternal cardiac function twin pregnancy. Obstet Gynecol. 2003;102(4):806.
- Duvekot JJ, Peeters LL. Maternal cardiovascular hemodynamic adaptation to pregnancy. Obstet Gynecol Surv. 1994 Dec;49(12 Suppl):S1-14.
- Capeless EL, Clapp JF. When do cardiovascular parameters return to their preconception values? Am J Obstet Gynecol. 1991 Oct;165(4):883-6.
- Berstein IM, Ziegler W, Badger GJ. Plasma volume expansion in early pregnancy. Obstet Gynecol. 2001;97(5):669.
- Pitkin RM, Witte DL. Platelet and leukocyte counts in pregnancy. JAMA. 1979;242(24):2696-98.
- Chandra S, Tripathi AK, Mishra S, Amzarul M, Vaish AK. Physiological changes in hematological parameters during pregnancy. Indian Journal of Hematology & Blood Transfusion. 2012;28(3):144-146.
- Gatti L, Tinconi PM, Guarneri D, Bertuijessi C, Ossola MW, Bosco P, Gianotti G. Hemostatic parameters and platelet activation by flow-cytometry in normal pregnancy: a longitudinal study. Internat J Clin Lab Res. 1994;24(4):217–219.
- Fleming AF. Hematological changes in pregnancy. Clin Obstet Gynecol. 1975;2:269.
- Rukavina D, Gill TJ: Immunobiology and immunopathology of early pregnancy. Early Pregnancy 1997; 3:141–145.
- Çintesun E, Incesu Çintesun FN, Ezveci H, Akyürek F, Çelik Ç. Systemic inflammatory response markers in preeclampsia. J Lab Physicians. 2018;10(3):316-319.
- Canzoneri BJ, Lewis DF, Groome L, Wang Y. Increased Neutrophil Numbers Account for Leukocytosis in Women with Preeclampsia. Am J Perinatol. 2009;26(10):729-732.
- Yucel B, Ustun B. Neutrophil to lymphocyte ratio, platelet to lymphocyte ratio, mean platelet volume, red cell distribution width and plateletcrit in preeclampsia.Pregnancy Hypertens. 2017 Jan;7:29-32.
- Montan S. Increased risk in the elderly parturient. Curr Opin Obstet Gynecol. 2007;19(2):110-12.
- Lynch CP, Baker N, Korte JE, Mauldin JG, Mayorga ME, Hunt KJ. Increasing prevalence of diabetes during pregnancy in south carolina. Journ of Women’s Health. 2015;24(4):316-323.
- Mitchell AA, Gilboa SM, Werler MM, et al. Medication use during pregnancy, with particular focus on prescription drugs: 1976-2008. American journal of obstetrics and gynecology. 2011;205(1):51.e1-8.
- Lappen JR, Keene M, Lore M, Grobman WA, Gossett DR. Existing models fail to predict sepsis in an obstetric population with intrauterine infection. Am J Obstet Gynecol. 2010 Dec;203(6):573 e1-5.
- Bonet M, Nogueira Pileggi V, Rijken MJ, et al. Towards a consensus definition of maternal sepsis: results of a systematic review and expert consultation. Reprod Health. 2017;14:67.
- WHO. Managing puerperal infections.Education material for teachers of midwifery Midwifery education modules – second edition. Geneva: 2008. http://www.who.int/maternal_child_adolescent/documents/4_9241546662/en/. Accessed 9 Aug 2018.
- WHO. Managing Complications in Pregnancy and Childbirth: A guide for midwives and doctors. Integrated Management of Pregnancy and Childbirth. Geneva: 2007. http://www.who.int/maternal_child_adolescent/documents/9241545879/en/.Accessed 9 Aug 2018.
- Maguire PJ, Turner MJ. Modified obstetric early warning systems. Am J Obstet Gynecol. 2015 Nov;213(5):748.
- Edwards SE, Grobman WA, Lappen JR, et al. Modified obstetric early warning scoring systems (MOEWS): validating the diagnostic performance for severe sepsis in women with chorioamnionitis. Am J Obstet Gynecol 2015;212:536.e1-8.
- Albright CM, Ali TN, Lopes V, Rouse DJ, Anderson BL. The sepsis in obstetrics score: a model to identify risk of morbidity from sepsis in pregnancy. Am J Obstet Gynecol. 2014 Jul;211(1):39.e1-8.
- Albright CM, Has P, Rouse DJ, Hughes BL. Internal validation of the sepsis in obstetrics score to identify risk of morbidity from sepsis in pregnancy. Obstet Gynecol. 2017 Oct;130(4):747-755.
- Bergeron MG. Treatment of pyelonephritis in adults. Med Clin North Am. 1995;79:619-649.
- Gilstrap LC III, Ramin SM. Urinary tract infections during pregnancy. Obstet Gynecol Clin North Am 2001;28:581-91.
- Nicolle LE, Bradley S, Colgan R, Rice JC, Schaeffer A, Hooton TM. Infectious Diseases Society of America guidelines for the diagnosis and treatment of asymptomatic bacteriuria in adults. Clin Infect Dis 2005;40:643-54.
- Duff P. Pyelonephritis in pregnancy. Clin Obstet Gynecol 1984;27:17-31.
- Jolley JA, Wing DA. Pyelonephritis in Pregnancy, An Update on Treatment Options for Optimal Outcomes. Drugs 2010; 70(13):1643-1655
- Wing DA, Fasset MJ, Getahun D. Acute Pyelonephritis in pregnancy: an 18-year retrospective analysis. Am J Ob Gyn. 2014;210(3):219e1-6.
- Thapa L. Acute pyelonephritis in pregnancy: A retrospective study. ANZJOG. 2007;47(4):313-315.
- Wing DA, Hendershott CM, Debuque L, Millar LK. A randomized trial of three antibiotic regimens for the treatment of pyelonephritis in pregnancy. Obstet Gynecol. 1998;92(2):249.
- Morgan J, Roberts S. Maternal sepsis. Obstet Gynecol Clin North Am. 2013 Mar;40(1)69-87.
- Gilo NB, Amini D, Landy HJ. Appendicitis and cholecystitis in pregnancy. Clin Obstet Gynecol. 2009;52(4):586–596.
- Addiss DG, Shaffer N, Fowler BS, Tauxe RV. The epidemiology of appendicitis and appendectomy in the United States. Am J Epidemiol. 1990;132(5):910.
- Mazze RI, Kallen B. Appendectomy during pregnancy: a Swedish registry study of 778 cases. Obstet Gynecol 1991;77:835.
- Hodjati H, Kazerooni T. Location of the appendix in the gravid patient: a reevaluation of the established concept. Int J Gynaecol Obstet 2003;81:245–7.
- Mahmoodian S. Appendicitis complicating pregnancy. South Med J. 1992 Jan;85(1):19-24.
- Sand M, Bechara FG, Holland-Letz T, Sand D, Mehnert G, Mann B. Diagnostic value of hyperbilirubinemia as a predictive factor for appendiceal perforation in acute appendicitis. Am J Surg. 2009;198(2):193.
- Williams R, Shaw J. Ultrasound scanning in the diagnosis of acute appendicitis in pregnancy. Emerg Med J. 2007 May;24(5):359-60.
- Duke E, Kalb B, Arif-Tiwari H, Daye ZJ, Gilbertson-Dahdal D, Keim SM, Martin DR. A Systematic Review and Meta-Analysis of Diagnostic Performance of MRI for Evaluation of Acute Appendicitis. AJR Am J Roentgenol. 2016 Mar;206(3):508-17.
- Kanal E, Barkovich AJ, Bell C, Borgstede JP, Bradley WG Jr, Froelich JW, Gilk T, Gimbel JR, Gosbee J, Kuhni-Kaminski E, Lester JW Jr, Nyenhuis J, Parag Y, Schaefer DJ, Sebek-Scoumis EA, Weinreb J, Zaremba LA, Wilcox P, Lucey L, Sass N, ACR Blue Ribbon Panel on MR Safety. ACR guidance document for safe MR practices: 2007. AJR Am J Roentgenol. 2007;188(6):1447.
- Zeger W, Holt K. Gynecologic infections. Emerg Med Clin North Am. 2003;21(3):631.
- Piegzova A, Unzetitig V. Tubo-ovarian abscess in the 39thweek of pregnancy.Ceska Gynekol. 2017 Fall;82(4):322-326.
- Abdou R, Miller T. Postpartum tubo-ovarian abscess, likely arising from pelvic inflammatory disease during pregnancy.
- AASLD-IDSA. Sexually Transmitted Disease Treatment Guidelines, 2015. Pelvic Inflammatory Disease (PID).https://www.cdc.gov/std/tg2015/pid.htm. Sep 3 2018.
- Park MA, Koch CA, Klemawesch P, et al. Increased adverse drug reactions to cephalosporins in penicillin allergy patients with positive penicillin skin test. International Arch Allergy Immunol 2010;153:268–73.
- Novalbos A, Sastre J, Cuesta J, et al. Lack of allergic cross-reactivity to cephalosporins among patients allergic to penicillins. Clin Exp Allergy 2001;31:438–43.
- Bjartling C, Osser S, Persson K. The association between Mycoplasma genitaliumand pelvic inflammatory disease after termination of pregnancy. BJOG 2010;117:361–364.
- American College of Emergency Physicians., Tintinalli, J. E., & Cline, D. (2015). Tintinalli’s emergency medicine manual(8th ed.). New York: McGraw-Hill Medical.
- Gilstrap LC, Cunningham FG. The bacterial pathogenesis of infection following cesarean section. Obstet Gynecol 1979;53:545–9.
- French LM, Smaill FM. Antibiotic regimens for endometritis after delivery. Cochrane Database Syst Rev 2002;(1):CD001067.
- Brumfield CG, Hauth JC, Andrews WW. Puerperal infection after cesarean delivery: evaluation of a standardized protocol. Am J Obstet Gynecol 2000; 182:1147.
- Brown CEL, Stettler RW, Twickler D, et al. Puerperal septic pelvic thrombophlebitis: incidence and response to heparin therapy. Am J Obstet Gynecol 1999;181:183.
- Nezhat C, Farhady P, Lemyre M. Septic Pelvic Thrombophlebitis Following Laparoscopic Hysterectomy. JSLS. 2009. Jan-Mar; 13(1):84-86.
- da Silva Cunha M, Godinho AB, Botelho R, de Almeida JP. Postpartum septic pelvic thrombophlebitis after caesarean delivery: a case report. Case Rep Womens Health. 2018 Jan; 17:5-7.
- Fishel Bartal M, Sibai BM, Ben-Mayor Bashi T, Dangot A, Schushan Eisen I, Dulitzki M, Inbar Y, Mazaki-Tovi S, Hendler I. Abdominal computer tomography (CT) scan in the evaluation of refractory puerperal fever: impact on management.J Matern Fetal Neonatal Med. 2018 Jul 4:1-151.
- Barton JR, Sibai BM. Severe Sepsis and Septic Shock in Pregnancy. Obstet Gynecol 2012;120:689-706.
- Neligan PJ, Laffey JG. Clinical review: Special populations–critical illness and pregnancy. Crit Care. 2011;15(4):227.
- Morgan J, Roberts S. Maternal sepsis. Obstet Gynecol Clin North Am. 2013 Mar;40(1):69-87.
- Rhodes A et al. Surviving Sepsis Campaign: International Guidelines for Management of Sepsis and Septic Shock: 2016. Crit Care Med. 2017;45(3):486.
- Albright CM, Ali TN, Lopes V, Rouse DJ, Anderson BL. Lactic acid measurement to identify risk of morbidity of sepsis in pregnancy. Am J Perinatal.2015 Apr;32(5):481-6.
- Plante LA. Management of Sepsis and Septic Shock for the Obstetrician–Gynecologist. Ob Gyn Clin. 2016; 43(4):659-678.
- Segal S, Wang SY. The effect of maternal catecholamines on the caliber of gravid uterine microvessels. Anesth Analg. 2008 Mar;106(3):888-92.
- Branco D, Caramona M, Martel F, de Almeida JA, Osswald W. Predominance of oxidative deamination in the metabolism of exogenous noradrenaline by the normal and chemically denervated human uterine artery. Naunyn Schmeidebergs Arch Pharmacol. 1992 Sep;346(3):286-93.
- Van Nimwegen D, Dyer DC. The action of vasopressors on isolated uterine arteries. Am J Obstet Gynecol. 1974;118(8):1099.
- Lee A, Ngan Kee WD, Gin T. A quantitative, systematic review of randomized controlled trials of ephedrine versus phenylephrine for the management of hypotension during spinal anesthesia for cesarean delivery. Anesth Analg. 2002;94(4):920.
- Nag DS, Samaddar DP, Chatterjee A, Kumar H, Dembla A. Vasopressors in obstetric anesthesia: A current perspective. World J Clin Cases. 2015 Jan 16; 3(1): 58–64.
- Stewart A, Fernando R, McDonald S, Hignett R, Jones T, Columb M. The dose-dependent effects of phenylephrine for elective cesarean delivery under spinal anesthesia. Anesth Analg. 2010;111:1230–1237.
- Ngan Kee WD, Khaw KS, Tan PE, Ng FF, Karmakar MK. Placental transfer and fetal metabolic effects of phenylephrine and ephedrine during spinal anesthesia for cesarean delivery. Anesthesiology. 2009;111:506–512.
- American Society of Anesthesiologists Task Force on Obstetric Anesthesia. Practice guidelines for obstetric anesthesia: an updated report by the American Society of Anesthesiologists Task Force on Obstetric Anesthesia. Anesthesiology. 2007;106:843–863.
- Yealy D.M., Kellum J.A., Huang D.T., et al: A randomized trial of protocol-based care for early septic shock. N Engl J Med 2014; 370: pp. 1683-1693
- Fishburne J.I., Meis P.J., Urban R.B., et al: Vascular and uterine responses to dobutamine and dopamine in the gravid ewe. Am J Obstet Gynecol 1980; 137: pp. 944-952.
- Pomp ER, Lenselink AM, Rosendaal FR, Doggen CJ. Pregnancy, the postpartum period and prothrombotic defects: risk of venous thrombosis in the MEGA study. J Thromb Haemost. 2008 Apr. 6(4):632-7.
- Kaplan D et al. VTE Incidence and Risk Factors in Patients With Severe Sepsis and Septic Shock. Chest. 2015 Nov;148(5):1224-1230.
45 a. Sivanesaratnam V. The acute abdomen and the obstetrician. Baillieres Best Pract Res Clin Obstet Gynaecol. 2000;14(1):89.






