Authors: Kasha Bornstein, MSc (@Borstein-Kasha); Jean Paul Bryant, MSc; Tim Montrief MD, MPH (@EMinMiami) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case:
A 75-year-old male with a past surgical history of a right sided craniotomy for subdural hematoma evacuation 10 days prior presents to the emergency department with acute-onset decreased responsiveness. Per family members, since he got home from the hospital, he has become increasingly somnolent, complaining of a headache and was found earlier today unresponsive. Vital signs reveal bradycardia, hypertension, and irregular, decreased respirations. Exam demonstrates a right sided craniotomy site without bleeding or signs of infection and a Glasgow Coma Scale of 6. You intubate the patient and begin to think of your next actions.
Introduction
Craniotomies comprise nearly 25% of some 2.3 million inpatient neurosurgical procedures in the United States each year. Following traumatic cranial injuries, the use of craniotomies to evacuate hematomas is a mainstay of treatment in many hospital systems.1 This procedure carries post-surgical complications ranging from debilitating to fatal. Approximately 13-27% of patients experience major complications following craniotomies.2 Additionally, a 1.0-2.5% 30-day mortality rate has been reported in the perioperative period.3 Post-craniotomy sequelae can be divided into normal and abnormal findings. Normal findings include surgical wounds such as burr holes, bleeding in the form of small hematomas, pneumocephalus without clinical deterioration, and post-operative radiologic enhancement.4 Abnormal findings include postoperative intracranial hemorrhage (incidence of 10.8%), soft tissue and bone flap infection (6.8%), tension pneumocephalus (2.5%), extradural abscesses and rarely, subdural empyema (0.43%).5,6
History and Physical Exam
History is critical for evaluating patients presenting to the ED with neurosurgical complications. It is necessary to obtain data including the surgery date, indication(s) for surgery, and hospital course. Information regarding the chief complaint can help focus the physician on any perioperative or postoperative complications including elevated intracranial pressure (ICP), intracranial hemorrhage (ICH), CSF leak, acute onset hyponatremia, or surgical site infections. An updated medication reconciliation should be performed including any corticosteroids, antibiotics, analgesics, antiplatelets or anticoagulants, and any other medications the patient may be taking.
A thorough neurologic exam should be conducted and compared with any documented pre- and post-operative findings. Exam should be focused towards uncovering focal neurologic deficits and any signs of ICH. Profound ICH can result in a herniation syndrome (Figure 1), but patients with elevated ICP typically have more subtle signs and symptoms. These include vomiting, altered sensorium, decreasing level of consciousness, and diplopia.

Differential Diagnosis
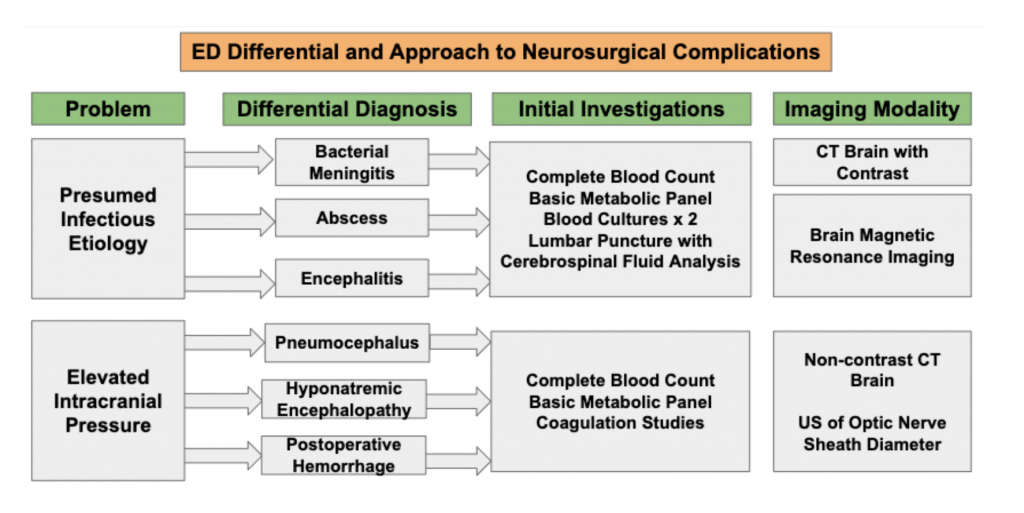
Most neurosurgical procedure complications are not life-threatening. However, “can’t miss” sequelae may result from elevated intracranial pressure and/or infectious causes. Pneumocephalus can cause acute elevations in intracranial pressure followed by rapid subsequent clinical deterioration. Superficial post-craniotomy infections may progress to meningitis, encephalitis, and abscess formation. Not all postoperative complications are neurological in nature: systemic causes of acute decompensation in the days following a neurosurgical procedure should be considered.
Intracranial Hypertension
There are many potential causes of increased intracranial pressure in the post-craniotomy patient. Intracranial hypertension may result from the original pathologic indication for surgery, (i.e. central nervous system (CNS) tumors or hydrocephalus), as a complication of the procedure itself, or secondary to instrumentation of neurological parenchyma or vasculature.11 The most dangerous among post-neurosurgical complications include intracranial hemorrhage, either parenchymal or extra-axial.11 While severe, these complications are also relatively common: a review of 3109 cases found approximately 1.9% resulted in post-craniotomy hemorrhage within 30 days of their initial operation significant enough to require evacuation, particularly for post-traumatic craniectomies.17 A study sample of 62 patients found that 100% of patients with subarachnoid hemorrhage and 79% of patients who received craniectomies for traumatic decompression developed post-operative extra-axial collections. Further, 85% of these extra-axial collections developed in the setting of ventricular CSF drainage.18
Pneumocephalus
Some volume (~9.3-11 mL) of intracranial air is expected after craniotomy.19-21 In a review of postoperative cranial imaging, pneumocephalus was found in 100% of patients following supratentorial craniotomy on imaging 2 days post-op and persisted in 25% of patients by the third week.22 Pneumocephalus is typically asymptomatic and computed tomography (CT) findings may be similar to those of non-tension pneumocephalus.23 However, tension pneumocephalus should be considered if radiographic evidence of air in the subdural or intraventricular space co-occurs with altered or declining mental status up to 10 weeks following surgery.24,25 Similar to tension pneumothorax, tension pneumocephalus is a life-threatening emergency in which one-way air inflow into the subdural space compresses and ultimately induces herniation of the brain.26 As intracranial pressure rises, subdural air trapped in the anterior cranial fossa separates the thin frontal dura mater closely adherent to bone from the arachnoid mater.21 This phenomenon produces the pathognomonic “Mount Fuji” sign on axial CT, in which posterior displacement of the frontal lobes occurs with widening of the interhemispheric space (Figure 2).21

Hyponatremic encephalopathy
Physiological and pharmacological causes of ICP should also be considered in these patients: many neurosurgical patients, particularly those undergoing treatment for malignancies, may be on chronic high-dose corticosteroids, raising the risk for adrenal crisis should they suddenly discontinue use.27 Cortisol and glucocorticoid analogs directly inhibit ADH secretion. Adrenal insufficiency secondary to corticosteroid withdrawal can result in increased ADH, causing free water retention and hyponatremia, as can syndrome of inappropriate antidiuretic hormone (SIADH) secondary to surgical injury or functional malignancies.28,29 Acute severe hyponatremia rapidly widens the osmotic gradient across the blood-brain barrier, promoting fluid shift into brain cells and causing cerebral edema with rapid neurological deterioration.30Acute hyponatremia is common in patients suffering from any type of brain insult, including subarachnoid hemorrhage, brain tumors, and/or surgical procedures.31 Hyponatremia is a particularly frequent complication of intracranial procedures, seen in as many as 20% of patients undergoing pituitary mass resection.31
Infection
As with any invasive procedure, neurosurgical operations pose risks for infection. Surgical site infections are relatively common, occurring in nearly 2% of 132,000 cases evaluated by the National Surgical Quality Improvement Program Analysis which included superficial, deep, and organ/space infections.32 Postoperative CNS infections may be even more frequent, occurring in between 5-7% of cases in large retrospective analyses.32
Meningitis
The 2017 Infectious Diseases Society of America (IDSA) Clinical Practice Guidelines recommend that patients presenting with new headache, fever, evidence of meningeal irritation, seizures, and/or worsening mental status in the setting of recent trauma or neurosurgery should raise suspicion for healthcare associated ventriculitis or meningitis (HAVM), or abscess indicating further workup.33 However, the IDSA provides the caveat that initial CSF analyses of cell count, glucose, and/or protein may reliably indicate infection in post-neurosurgical patients given recent CNS manipulation and/or trauma.33 Meningitis may present with or without focal changes on imaging studies, with subtle evidence of meningeal enhancement being the only indicator.
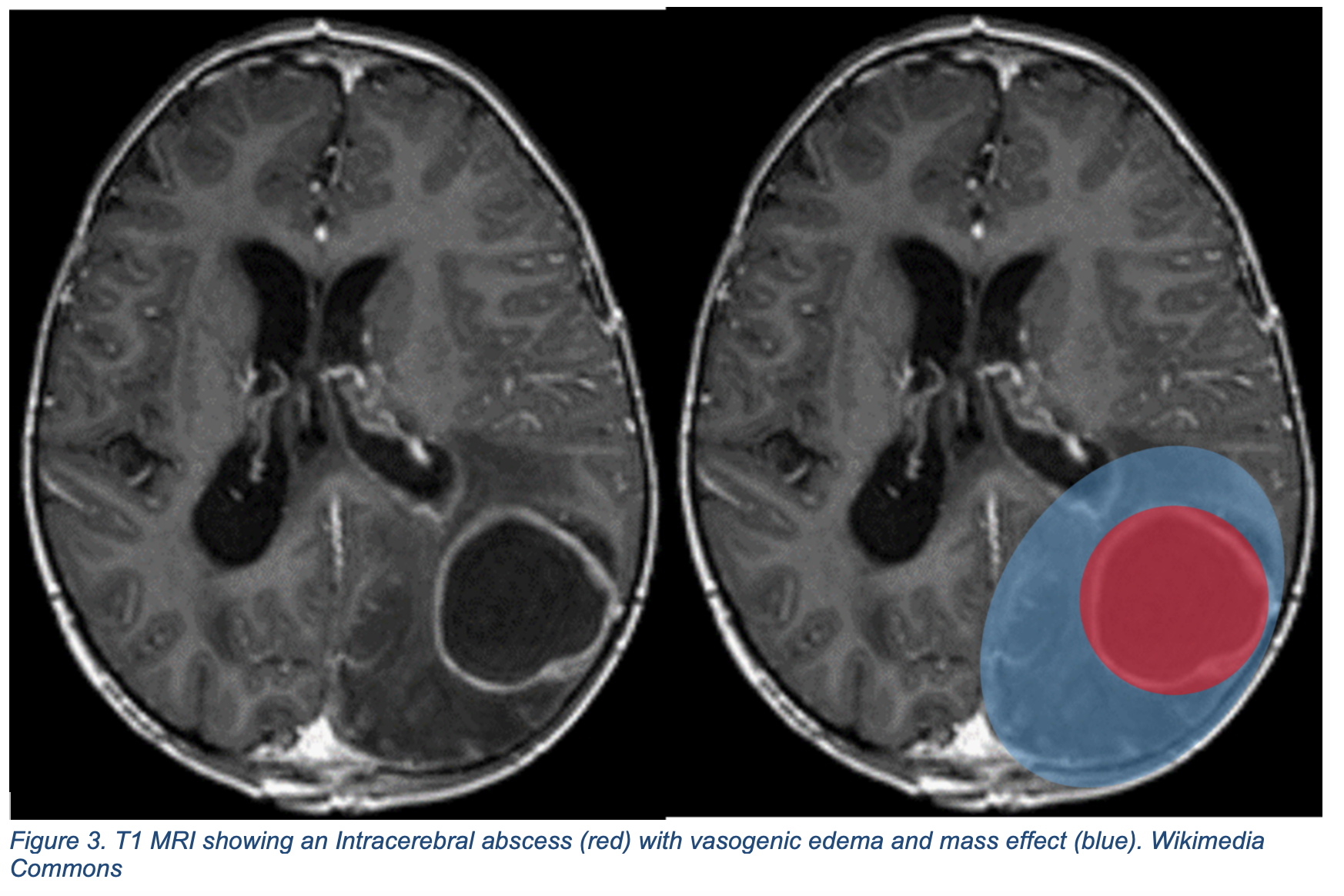
Abscess
Similar to meningitis, the neurosurgical postoperative period poses high risk for bacterial cerebral abscess.34 While development of postoperative intracranial abscess are rare, they portend high mortality at approximately 16%.35Intracranial abscesses can arise due to direct inoculation during instrumentation or via contiguous spread from a local or distant nidus of infection. Signs and symptoms of intracranial abscesses overlap with meningitis; particularly fever, progressively altered sensorium, headache, and seizure. However, new onset focal neurologic signs are more suggestive of abscess versus meningitis.36 In the presence of these new symptoms, immediate neuro-imaging studies such as CT or magnetic resonance imaging (MRI), are recommended to determine presence of bacterial brain abscess (Figure 3).
Encephalitis
Although less common than meningitis and abscess, post-operative encephalitis poses significant risk. Untreated, encephalitis mortality can be as high as 70%.37,38 Even with adequate treatment, mortality approaches 30%.37 Herpes simplex virus (HSV1/2) is the most common viral encephalitis cause overall as well as the most common cause of postoperative encephalitis.39 Herpes simplex encephalitis can theoretically result from direct inoculation secondary to a break in sterile technique. However, approximately two-thirds of HSV encephalitis cases are believed to occur as a result of reactivation rather than primary infection, particularly in the setting of CNS injury and/or immunosuppression which are often necessary aspects of the intra- and/or perioperative care of the neurosurgical patient.38
Diagnostic Considerations
Laboratory Testing
Patient clinical presentation, history, and underlying comorbidities guide laboratory testing. At a minimum, labs should include a complete blood count and basic metabolic panel, with consideration for lumbar puncture (LP) and blood cultures to evaluate for CNS and/or systemic infection.33
LP and CSF Evaluation
IDSA guidelines for initial evaluation of HAVM strongly recommend LP and CSF analysis.33 Evaluation of CSF is often complicated given the inherent invasiveness of craniotomies and other intensive neurological procedures.33 Cell counts may be obscured by blood leakage into the CSF, or may cause an inflammatory reaction, sometimes called “chemical meningitis.”33 Signs and symptoms of infection such as fever, rigors, chills, and elevated serum lactate increase the likelihood of infection.33
LP findings indicative of or highly suspicious for HAVM include CSF with a high leukocyte count, (at least 11 WBCs/mm3 and ≥50% neutrophils) and elevated CSF protein concentrations.40 Early procurement of CSF cultures can guide later treatment, particularly if cultures result positive for Staphylococcus aureus, aerobic gram-negative bacilli, or fungal pathogens.33 However, normal CSF cell count, glucose, and protein may not reliably exclude infection in patients with HAVM, and a negative CSF Gram stain does not exclude the presence of infection, especially in patients who have received previous antimicrobial therapy. CSF lactate is a good marker for active CNS infection. A CSF lactate cutoff of 4 mmol/L is 88% sensitive and 98% specific for diagnosing post-neurosurgical bacterial meningitis.41
Imaging Studies
Point of Care Ultrasound is a rapid and inexpensive tool that can be utilized to evaluate intracranial hypertension via measurement of the optic nerve sheath diameter (ONSD).42 Average ONSD >5 mm suggests elevated intracranial pressure.43 Increased ONSD has been reported to show correlation with elevated ICP in patients following decompressive craniectomy.44 A CT brain without contrast or MRI can reveal hemorrhage, edema, simple or tension pneumocephalus, and brain herniation. In the setting of acute intracerebral hemorrhage (ICH), non-contrast CT is the most widely used study as the presence of blood is evident almost immediately. CT has a sensitivity of 89.8% for detecting hemorrhagic lesions.45 Regarding MRI, results from the large-scale Hemorrhage and Early MRI Evaluation (HEME) study showed that MRI can detect acute ICH with up to 100% sensitivity and accuracy when read by experienced radiologists.46 While MRI and CT may be used for detection of ICH, CT is the gold standard when evaluating a patient for pneumocephalus and has been described in the literature as having high sensitivity and specificity.47-49
If infection is suspected, a non-contrast CT brain should be obtained first to rule out hemorrhage, followed by a CT brain with contrast, as non-contrast CT is often normal and less sensitive for detecting infection.33,50 A study assessing imaging sensitivity for encephalitis noted abnormal cranial CT findings in 38% of patients, while MRI revealed pathological changes in 90.6-95.5%.51 MRI is more sensitive for evaluation of intracranial infection and ischemia but is contraindicated in post-operative patients with non-MRI compatible surgical clips and/or implants for ICP monitoring or CSF drainage.22
Treatment
As with any unstable patient, emergency care of the post-neurosurgical patient rests on principles of maintaining airway, breathing and circulation. The most rapidly lethal complications of neurosurgical sequelae derive from critically elevated ICP.4 Urgent involvement of the surgical team is essential upon recognition of potentially fatal ICH. Standard resuscitation techniques should be instituted with attention towards maintenance of head elevation >30° reduce ICP.52 In any patient with life-threatening elevations of ICP, sedation, intubation, and targeted hyperventilation to a PCO2 of 26 to 30 mmHg is essential.53 Though the protective effects of targeted hyperventilation are transient, this approach is recommended in the setting of critically elevated ICP.54 Additional treatment should be tailored to the severity and underlying pathophysiological causes of ICP elevation.
Superficial wound infections warrant immediate attention with antibiotics, as they can progress to extradural abscesses and/or subdural empyema if left untreated.9,55 Post-neurosurgical cerebral abscesses, subdural empyema, and bone flap infections usually require another neurosurgical procedure to remove pus and debris.55 Empiric antibiotics for intracranial abscess should cover the most common bacteria found in neurosurgical site infections, including Staphylococcus aureus including MRSA, Staphylococcus epidermidis, Enterobacteriaceae, and Pseudomonas aeruginosa.33
Hypertonic saline vs. mannitol
Emergent management of ICH should be tailored towards temporizing the patient’s underlying insult until definitive surgical therapy can be provided. Osmotic diuretic therapy remains a mainstay of treatment for managing elevated ICP. Mannitol 20% solution has historically been the agent of choice for osmotic therapy in patients with increased ICP.56Mannitol is administered as a bolus dose of 1 g/kg, with a repeat of 0.25 to 0.5 g/kg every 6-8 hours. Effects of mannitol typically last from 4-24 hours, peaking around 1 hour following administration.57 Mannitol is well-known, and its efficacy as a robust osmotic agent is well described in the literature.58-61 Hypertonic saline (HTS) has replaced mannitol in many institutions, and a growing body of evidence suggests superiority of HTS in reducing ICP in the acute setting.60,62 HTS is typically administered in bolus doses fluid to acutely lower ICP. HTS concentrations range from 2%-23.5%.63 Randomized controlled trials have shown that a 100 mL bolus of 7.5% saline can significantly reduce ICP.64-66 A meta-analysis concluded that continuous infusion or bolus administration of HTS may be more effective than mannitol to reduce acute ICH.62 Furthermore, a case-control study reported significantly lower durations of increased ICP and decreased cerebral perfusion pressure when using HTS in comparison to mannitol.67 In all patients receiving osmotic therapy, it is important to carefully evaluate risks and benefits. A basic metabolic panel can exclude hyponatremia in consideration of preventing central pontine myelinolysis.68 However, given these patients’ critically status, time may be limited for laboratory studies to result before treatment is administered. In patients receiving mannitol, a Foley catheter should be placed and IV fluid rehydration administered matching urine output.
Disposition
Disposition of the sick post-neurosurgical patient depends on the ED setting. In tertiary care centers with neurological surgery available, early consultation with the operative team can assist in determination for inpatient readmission by the ED physician and/or urgent neurosurgical revision. Patients requiring reoperation should be admitted to the ICU following intervention. This may be applicable for patients with symptomatic pneumocephalus, persistent pneumocephalus lasting more than one week, or extensive intracranial hemorrhage. Simple pneumocephalus is managed conservatively with bed rest and high flow oxygen. Such patients may be admitted to the floor or intermediate care units/ICU step-down for further observation, depending on facility capabilities.69 The decision to admit patients with meningitis to the ICU should be based on any acute changes in consciousness, severe sepsis/septic shock, or meningococcal origin due to its known rapid progression.70 In the community ED setting without access to neurosurgical management, early assessment of acuity and need for urgent/emergent intervention, and prompt transfer to a neurosurgical center – preferably where the patient received initial operative management – can greatly improve patient outcomes.
Case Conclusion:
An emergent unenhanced CT scan of the brain was performed and showed no extra-axial blood, but instead a “Mt. Fuji sign”- large volume of intracranial air causing compression and separation of both frontal lobes away from the falx cerebri in the midline (Figure 3). Neurosurgery was consulted, and the patient proceeded to the operating room where the neurosurgeon created bifrontal burr holes and durotomies. Although no audible air rush was heard, the patient’s consciousness improved rapidly to GCS 14/15 (E4 V4 M6), and he was returned to the ward to recover. Over the next 48 hours, the patient made a steady recovery in the intensive care unit and was subsequently discharged home.
Key Points
- Approximately 13-27% of patients experience major complications following craniotomies.
- Normal post-neurosurgical findings include surgical wounds such as burr holes, bleeding in the form of small hematomas, pneumocephalus without clinical deterioration, and post-operative radiologic enhancement.
- The most common complications of neurosurgical procedures include hemorrhage (incidence of 10.8%), soft tissue and bone flap infection (6.8%), tension pneumocephalus (2.5%), extradural abscesses and rarely, subdural empyema (0.43%).
- If infection is suspected, a non-contrast CT brain should be ordered first to rule out hemorrhage, followed by a CT brain with contrast, as non-contrast CT is often normal and less sensitive for detecting infection.
- Empiric antibiotics for intracranial abscess should cover the most common bacteria found in neurosurgical site infections, including Staphylococcus aureus including MRSA, Staphylococcus epidermidis, Enterobacteriaceae, and Pseudomonas aeruginosa.
References / Further Reading
- Kuge A, Tsuchiya D, Watanabe S, Sato M, Kinjo T. Endoscopic hematoma evacuation for acute subdural hematoma in a young patient: a case report. Acute Med Surg. 2017;4(4):451-453.
- Lonjaret L, Guyonnet M, Berard E, et al. Postoperative complications after craniotomy for brain tumor surgery. Anaesth Crit Care Pain Med. 2017;36(4):213-218.
- Goel NJ, Mallela AN, Agarwal P, et al. Complications Predicting Perioperative Mortality in Patients Undergoing Elective Craniotomy: A Population-Based Study. World Neurosurgery. 2018;118:e195-e205.
- Chughtai KA, Nemer OP, Kessler AT, Bhatt AA. Post-operative complications of craniotomy and craniectomy. Emerg Radiol. 2019;26(1):99-107.
- Coughlan F, Lam A, Honeybul S. Tension Pneumocephalus Following Bilateral Craniotomies. Cureus. 2017;9(6):e1358.
- Chughtai KA, Nemer OP, Kessler AT, Bhatt AA. Post-operative complications of craniotomy and craniectomy. Emergency Radiology. 2019;26(1):99-107.
- Jiang X, Ma J, Hou F, Li J, Li R, Lang H. Neurosurgical Site Infection Prevention: Single Institute Experience. Turkish neurosurgery. 2016;26(2):234-239.
- Shi ZH, Xu M, Wang YZ, et al. Post-craniotomy intracranial infection in patients with brain tumors: a retrospective analysis of 5723 consecutive patients. British journal of neurosurgery. 2017;31(1):5-9.
- Hlavin ML, Kaminski HJ, Fenstermaker RA, White RJ. Intracranial suppuration: a modern decade of postoperative subdural empyema and epidural abscess. Neurosurgery. 1994;34(6):974-980; discussion 980-971.
- Mollman HD, Haines SJ. Risk factors for postoperative neurosurgical wound infection. A case-control study. J Neurosurg. 1986;64(6):902-906.
- Seifman MA, Lewis PM, Rosenfeld JV, Hwang PY. Postoperative intracranial haemorrhage: a review. Neurosurgical review. 2011;34(4):393-407.
- Vassilouthis J, Anagnostaras S, Papandreou A, Dourdounas E. Is postoperative haematoma an avoidable complication of intracranial surgery? British journal of neurosurgery. 1999;13(2):154-157.
- Fukamachi A, Koizumi H, Nagaseki Y, Nukui H. Postoperative extradural hematomas: computed tomographic survey of 1105 intracranial operations. Neurosurgery. 1986;19(4):589-593.
- Taylor WA, Thomas NW, Wellings JA, Bell BA. Timing of postoperative intracranial hematoma development and implications for the best use of neurosurgical intensive care. Journal of neurosurgery. 1995;82(1):48-50.
- Agarwal R, Mohapatra S, Rath GP, Kapil A. Active Surveillance of Health Care Associated Infections in Neurosurgical Patients. Journal of clinical and diagnostic research: JCDR. 2017;11(7):Dc01-dc04.
- Adams RD, Victor M, Ropper AH. Principles of neurology. 6th ed. New York: McGraw-Hill, Health Professions Division; 1997.
- Desai VR, Grossman R, Sparrow H. Incidence of Intracranial Hemorrhage After a Cranial Operation. Cureus. 2016;8(5):e616.
- Nalbach SV, Ropper AE, Dunn IF, Gormley WB. Craniectomy-associated Progressive Extra-Axial Collections with Treated Hydrocephalus (CAPECTH): redefining a common complication of decompressive craniectomy. J Clin Neurosci. 2012;19(9):1222-1227.
- Reasoner DK, Todd MM, Scamman FL, Warner DS. The incidence of pneumocephalus after supratentorial craniotomy. Observations on the disappearance of intracranial air. Anesthesiology. 1994;80(5):1008-1012.
- Brandstrom H, Sundelin A, Hoseason D, et al. Risk for intracranial pressure increase related to enclosed air in post-craniotomy patients during air ambulance transport: a retrospective cohort study with simulation. Scand J Trauma Resusc Emerg Med. 2017;25(1):50.
- Dabdoub CB, Salas G, Silveira Edo N, Dabdoub CF. Review of the management of pneumocephalus. Surg Neurol Int. 2015;6:155.
- Sinclair AG, Scoffings DJ. Imaging of the post-operative cranium. Radiographics. 2010;30(2):461-482.
- Hoz SS, Baban K, Sabah M, Aktham A, Narvaez-Rojas AR, Moscote-Salazar LR. Delayed Progressive Intraparenchymal Tension Pneumocephalus after Craniotomy for Recurrent Pituitary Macroadenoma: A Case Report. Bulletin of emergency and trauma. 2018;6(2):174-177.
- Rathore AS, Satyarthee GD, Mahapatra AK. Post-Traumatic Tension Pneumocephalus: Series of Four Patients and Review of the Literature. Turkish neurosurgery. 2016;26(2):302-305.
- Rasouli M, Honeybul S. Delayed tension pneumocephalus following decompressive craniectomy. Journal of Clinical Neuroscience. 2018;58:205-206.
- Sinclair AG, Scoffings DJ. Imaging of the Post-operative Cranium. RadioGraphics. 2010;30(2):461-482.
- Broersen LHA, Pereira AM, Jørgensen JOL, Dekkers OM. Adrenal Insufficiency in Corticosteroids Use: Systematic Review and Meta-Analysis. The Journal of Clinical Endocrinology & Metabolism. 2015;100(6):2171-2180.
- Lamas C, del Pozo C, Villabona C. Clinical guidelines for management of diabetes insipidus and syndrome of inappropriate antidiuretic hormone secretion after pituitary surgery. Endocrinologia y nutricion : organo de la Sociedad Espanola de Endocrinologia y Nutricion. 2014;61(4):e15-24.
- Goldvaser H, Rozen-Zvi B, Yerushalmi R, Gafter-Gvili A, Lahav M, Shepshelovich D. Malignancy associated SIADH: Characterization and clinical implications. Acta Oncologica. 2016;55(9-10):1190-1195.
- Rajagopal R, Swaminathan G, Nair S, Joseph M. Hyponatremia in Traumatic Brain Injury: A Practical Management Protocol. World Neurosurgery. 2017;108:529-533.
- Hannon MJ, Thompson CJ. Neurosurgical Hyponatremia. J Clin Med. 2014;3(4):1084-1104.
- Karhade AV, Cote DJ, Larsen AM, Smith TR. Neurosurgical Infection Rates and Risk Factors: A National Surgical Quality Improvement Program Analysis of 132,000 Patients, 2006-2014. World Neurosurg. 2017;97:205-212.
- Tunkel AR, Hasbun R, Bhimraj A, et al. 2017 Infectious Diseases Society of America’s Clinical Practice Guidelines for Healthcare-Associated Ventriculitis and Meningitis. Clin Infect Dis. 2017.
- McClelland S, 3rd, Hall WA. Postoperative central nervous system infection: incidence and associated factors in 2111 neurosurgical procedures. Clin Infect Dis. 2007;45(1):55-59.
- Yang KY, Chang WN, Ho JT, Wang HC, Lu CH. Postneurosurgical nosocomial bacterial brain abscess in adults. Infection. 2006;34(5):247-251.
- Patel K, Clifford DB. Bacterial brain abscess. Neurohospitalist. 2014;4(4):196-204.
- Chaudhuri A, Kennedy PG. Diagnosis and treatment of viral encephalitis. Postgrad Med J. 2002;78(924):575-583.
- Jaques DA, Bagetakou S, L’Huillier AG, et al. Herpes simplex encephalitis as a complication of neurosurgical procedures: report of 3 cases and review of the literature. Virol J. 2016;13:83.
- Lund M. Herpes simplex virus reactivation and encephalitis after topectomy. J Pediatr Health Care. 2011;25(5):323-327.
- Sundbarg G, Nordstrom CH, Soderstrom S. Complications due to prolonged ventricular fluid pressure recording. Br J Neurosurg. 1988;2(4):485-495.
- Leib SL, Boscacci R, Gratzl O, Zimmerli W. Predictive value of cerebrospinal fluid (CSF) lactate level versus CSF/blood glucose ratio for the diagnosis of bacterial meningitis following neurosurgery. Clin Infect Dis. 1999;29(1):69-74.
- Toscano M, Spadetta G, Pulitano P, et al. Optic Nerve Sheath Diameter Ultrasound Evaluation in Intensive Care Unit: Possible Role and Clinical Aspects in Neurological Critical Patients’ Daily Monitoring. Biomed Res Int. 2017;2017:1621428.
- Bhandari D, Udupi Bidkar P, Adinarayanan S, Narmadhalakshmi K, Srinivasan S. Measurement of changes in optic nerve sheath diameter using ultrasound and computed tomography scan before and after the ventriculoperitoneal shunt surgery in patients with hydrocephalus – A prospective observational trial. British Journal of Neurosurgery. 2019;33(2):125-130.
- Wang J, Li K, Li H, et al. Ultrasonographic optic nerve sheath diameter correlation with ICP and accuracy as a tool for noninvasive surrogate ICP measurement in patients with decompressive craniotomy. 2019:1.
- Gentry LR, Godersky JC, Thompson B, Dunn VD. Prospective comparative study of intermediate-field MR and CT in the evaluation of closed head trauma. AJR American journal of roentgenology. 1988;150(3):673-682.
- Fiebach JB, Schellinger PD, Gass A, et al. Stroke magnetic resonance imaging is accurate in hyperacute intracerebral hemorrhage: a multicenter study on the validity of stroke imaging. Stroke. 2004;35(2):502-506.
- Gorissen Z, Hakvoort K, van den Boogaart M, Klinkenberg S, Schijns O. Pneumocephalus: a rare and life-threatening, but reversible, complication after penetrating lumbar injury. Acta Neurochir (Wien). 2019;161(2):361-365.
- Dabdoub CB, Salas G, Silveira EdN, Dabdoub CF. Review of the management of pneumocephalus. Surg Neurol Int. 2015;6:155-155.
- Schirmer CM, Heilman CB, Bhardwaj A. Pneumocephalus: case illustrations and review. Neurocritical care. 2010;13(1):152-158.
- Foerster BR, Thurnher MM, Malani PN, Petrou M, Carets-Zumelzu F, Sundgren PC. Intracranial infections: clinical and imaging characteristics. Acta radiologica (Stockholm, Sweden : 1987). 2007;48(8):875-893.
- Kastrup O, Wanke I, Maschke M. Neuroimaging of infections. NeuroRx. 2005;2(2):324-332.
- Geeraerts T, Velly L, Abdennour L, et al. Management of severe traumatic brain injury (first 24hours). Anaesth Crit Care Pain Med. 2018;37(2):171-186.
- Oliver Z. Intracranial Hemorrhage. SAEM. Available at https://www.saem.org/cdem/education/online-education/m4-curriculum/group-m4-neurology/intracranial-hemorrhage. Published 2015. Accessed 07/15/2019, 2019.
- Godoy DA, Seifi A, Garza D, Lubillo-Montenegro S, Murillo-Cabezas F. Hyperventilation Therapy for Control of Posttraumatic Intracranial Hypertension. Frontiers in neurology. 2017;8:250.
- Dashti SR, Baharvahdat H, Spetzler RF, et al. Operative intracranial infection following craniotomy. 2008;24(6):E10.
- Wise BL, Chater N. The value of hypertonic mannitol solution in decreasing brain mass and lowering cerebro-spinal-fluid pressure. Journal of neurosurgery. 1962;19:1038-1043.
- Jafar JJ, Johns LM, Mullan SF. The effect of mannitol on cerebral blood flow. Journal of neurosurgery. 1986;64(5):754-759.
- Gasco J, Sendra J, Lim J, Ng I. Linear correlation between stable intracranial pressure decrease and regional cerebral oxygenation improvement following mannitol administration in severe acute head injury patients. Acta neurochirurgica Supplement. 2005;95:73-77.
- Sakowitz OW, Stover JF, Sarrafzadeh AS, Unterberg AW, Kiening KL. Effects of mannitol bolus administration on intracranial pressure, cerebral extracellular metabolites, and tissue oxygenation in severely head-injured patients. The Journal of trauma. 2007;62(2):292-298.
- Abughazaleh RD, Jancik JT, Paredes-Andrade E, Solid CA, Rockswold GL. Safety of intravenous hypertonic saline administration in severe traumatic brain injury. Ann Pharmacother. 2012;46(10):1441-1442.
- Rockswold GL, Solid CA, Paredes-Andrade E, Rockswold SB, Jancik JT, Quickel RR. Hypertonic saline and its effect on intracranial pressure, cerebral perfusion pressure, and brain tissue oxygen. Neurosurgery. 2009;65(6):1035-1041; discussion 1041-1032.
- Mortazavi MM, Romeo AK, Deep A, et al. Hypertonic saline for treating raised intracranial pressure: literature review with meta-analysis. Journal of neurosurgery. 2012;116(1):210-221.
- Mangat HS. Hypertonic saline infusion for treating intracranial hypertension after severe traumatic brain injury. Crit Care. 2018;22(1):37.
- Francony G, Fauvage B, Falcon D, et al. Equimolar doses of mannitol and hypertonic saline in the treatment of increased intracranial pressure. Critical care medicine. 2008;36(3):795-800.
- Strandvik GF. Hypertonic saline in critical care: a review of the literature and guidelines for use in hypotensive states and raised intracranial pressure. Anaesthesia. 2009;64(9):990-1003.
- Boone MD, Oren-Grinberg A, Robinson TM, Chen CC, Kasper EM. Mannitol or hypertonic saline in the setting of traumatic brain injury: What have we learned? Surg Neurol Int. 2015;6:177-177.
- Mangat HS, Wu X, Gerber LM, et al. Hypertonic Saline is Superior to Mannitol for the Combined Effect on Intracranial Pressure and Cerebral Perfusion Pressure Burdens in Patients With Severe Traumatic Brain Injury. Neurosurgery. 2019.
- Rangel-Castilla L, Gopinath S, Robertson CS. Management of intracranial hypertension. Neurol Clin. 2008;26(2):521-541, x.
- Dexter F, Reasoner DK. Theoretical assessment of normobaric oxygen therapy to treat pneumocephalus. Anesthesiology. 1996;84(2):442-447.
- Fernandes D, Gonçalves-Pereira J, Janeiro S, Silvestre J, Bento L, Póvoa P. Acute bacterial meningitis in the intensive care unit and risk factors for adverse clinical outcomes: Retrospective study. Journal of Critical Care. 2014;29(3):347-350.