Originally published on Ultrasound G.E.L. on 1/7/19 – Visit HERE to listen to accompanying PODCAST! Reposted with permission.
Follow Dr. Michael Prats, MD (@PratsEM), Dr. Creagh Bougler, MD (@CreaghB), and Dr. Jacob Avila, MD (@UltrasoundMD) from Ultrasound G.E.L. team!
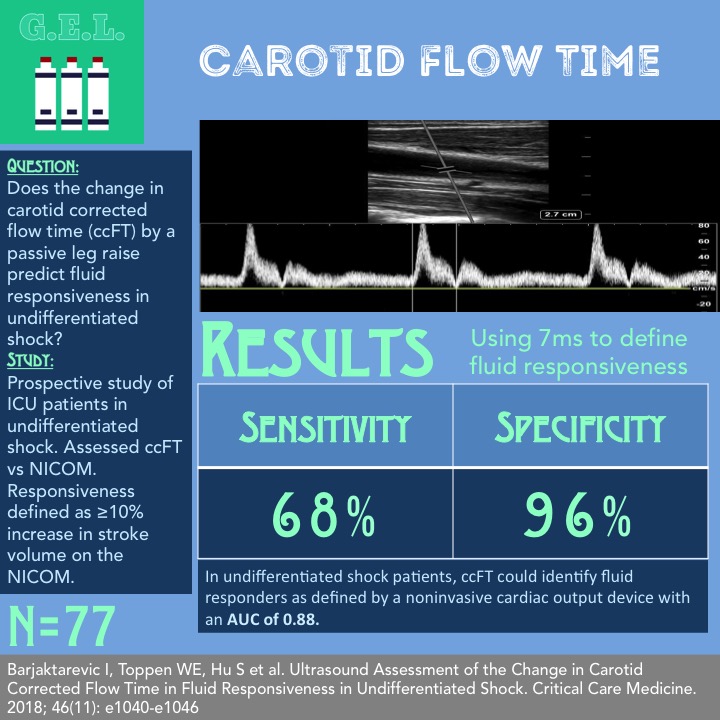
Ultrasound Assessment of the Change in Carotid Corrected Flow Time in Fluid Responsiveness in Undifferentiated Shock
Critical Care Medicine Nov 2018 – Pubmed Link
Take Home Points
1. In a population of undifferentiated shock patients, corrected carotid flow time can identify fluid responders as defined by a noninvasive cardiac output device with an AUC of 0.88.
2. Carotid flow time is one piece of the puzzle and should be interpreted in the clinical context as well as with consideration for contractility and afterload.
Background
In case you haven’t been reading at any POCUS fluid responsiveness literature for the past 5 years – don’t worry, nothing has changed. We are still looking for that perfect non-invasive test to tell us when we should give fluids and when we should use vasopressors for that patient in shock. We know that IVC ultrasound has a lot of problems (not as accurate in spontaneously breathing, limited utility it patients with increased right sided pressures). Left ventricular outflow track VTI has its own problems as well (takes a little more skill, questionable reproducibility). So a few years ago, they came up with the carotid flow time, which was supposed to fix all the problems. The idea is that you are measuring the time is takes for one systole seen on doppler in the common carotid artery. You correct the time for heart rate, hence corrected carotid flow time. Since the carotids are easy to find, it should be easy to do. Since it is based on time, the angle of insonation shouldn’t matter. Well everything sounded great and there were a few promising studies. (like this one, this one, and also this one). These were then followed by a study or two that said, “nevermind, it doesn’t work.” like this one and this one. So now we just don’t know. This study takes another crack at it – using actual patients in shock (unlike many of the prior studies) and using the passive leg raise.
Question
Does the change in carotid corrected flow time (ccFT) induced by a passive leg raise maneuver predict fluid responsiveness in undifferentiated shock?
Population
Single academic quaternary care center in United States
Inclusion:
- Adults
- < 24 hours of undifferentiated shock
- Admitted to medical or surgical ICU with persistent vasopressor requirements despite > 1 L of IV fluids
These are the patients we ask the question “should I give more fluids”
Exclusion:
- History of left or right heart failure
- Pulmonary hypertension
- Non-sinus cardiac rhythm
- Significant peripheral vascular disease
- Suspected or known increased intracranial pressure
- Recent abdominal surgery
- Recent history of venous thromboembolism
- BMI < 15 or greater than 40 kg/m^2
Design
Prospective
Enrolled May 2016 to April 2017
Patients in undifferentiated shock were assessed with POCUS and NICOM (a noninvasive bioreactance cardiac output monitoring device) simultaneously.
Took one measurement at baseline, patient in semirecumbent position, 45º head of bed elevation for at least 10 minutes
Then measured during the passive leg raise maneuver: patient in supine position for 3 minutes with legs passively supported by an inflated wedge at 45º elevation. ccFT measured after 3 minutes.
Fluid responsiveness defined as ≥10% increase in stroke volume on the NICOM.
ccFT calculated using Wodey’s formula (apparently works better for fast heart rates). Many other studies use Bazet’s formula. If you are curious about this debate – you will absolutely love this fight night Wodey’s formula vs. Bazett’s formula
Second investigator re-measured the images to assess inter-user variability
Main outcome was predictor of fluid responsive status expressed as receiver operating characteristics (ROC).
Who did the ultrasounds?
Article states, “trained physician sonographer”. Not clear if one person or multiple.
The Scan
High frequency linear transducer

Here’s how they did it:
- Obtained a long axis view of common carotid
- Pulsed wave doppler in vessel
- Measured from beginning of upstroke to the trough of the incisural notch on a pulse waveform
- Measured one cardiac cycle to correct for heart rate
Here is a nice video on Carotid ultrasound for volume responsiveness assessmentby the folks at MGH
5 Min Sono – Fluid Responsiveness using LVOT VTI
5 Min Sono – Ultrasound in Shock
The Image Atlas – Carotid vs IJ
Results
314 assessed for eligibility, 235 excluded
2 excluded for problems during PLR (hypoxia, atrial fibrillation)
Patients with complete exams n = 77
- Mean age 60.6 years old
- 51% Female
- Mean BMI 24
- 42% end-stage renal disease or dialysis
- Mean total fluids 8 liters
- 59% on mechanical ventilation, 47% of those has passive ventilation
- 71% on norepinpehrine, 14% on combination of pressors, a few on dopamine, vasopressin, phenylephrine
- Mean MAP 60 mmHg
- Mean Acute physiology and chronic health evaluation (APACHE) II score 24.5 (~50% mortality)
70.1% of patients were fluid responsive.
Primary Outcome
Increase in ccFT after PLR
Fluid responsive 14.1 ms(±19). +4.8% from baseline
NonResponsive -4.0 (±8 ms) -1.4% from baseline
ROC curve analysis
AUC 0.8784 (considered “good”)
Using 7 ms to define fluid responsiveness
Specificity of 96%
Sensitivity of 68%
Additional Findings
Mechanical ventilation, respiratory rate, PEEP > 5 cm H20 had no significant impact on test performance
Good agreement between investigators
PLR was able to be completed in 97.5% of patients.
Limitations
Small study. A lot of people were excluded, would have been nice to know what was the main reason for exclusion based on their criteria. Since they exclude anyone who would be hurt by PLR, we are limited in our external validity.
Possible Funding Bias? I’m wondering if this was an auto-measurement of ccFT. They don’t say it specifically in the methods, but they mention that this was done with the GE LOGIQ e and during the discussion they contrast their measurement with “manual measurement” and state that manual measurement can lead to misinterpretation or measurement bias. This is suspicious for some influence of the GE funding for this research.
How good of a standard reference is the NICOM? Questionable to say the least. At the end of the day, it’s just another surrogate.
Discussion
Authors bring up some problems with FT in general. It depends on heart rate, inotropy, afterload. So in order for the change in FT to reflect the preload, the afterload and contractility must stay the same, and you have to correct for the heart rate.This (somewhat hilarious) paper explains why this concept should not work. On the other hand, why would those things change during a passive leg raise?
Is this just a fad? I think that there is some merit to it. It certainly is a lot easier to do than serial LVOT VTIs. Like all tests, I think knowing that it is not perfect, and using it in the context of the patient’s afterload and contractility, might make it useful clinically. However, I am not counting on this test any more than I count on IVC or LVOT VTI.
Take Home Points
1. In a population of undifferentiated shock patients, corrected carotid flow time can identify fluid responders as defined by a noninvasive cardiac output device with an AUC of 0.88.
2. Carotid flow time is one piece of the puzzle and should be interpreted in the clinical context as well as with consideration for contractility and afterload.
Our score

Expert Reviewer for this Post

Nik Theyyunni, MD @Heydrnik
Nik Theyyunni is the ultrasound director at the University of Michigan, Department of Emergency Medicine
Nik Theyyunni has received research funding from GE Healthcare for work on point of care ultrasound.
Reviewer’s Comments
ccFT is a great example of an ultrasound measure that we don’t quite understand yet. In this study the authors are using a measurement that measures an interaction between many factors including preload, afterload, inotropy, and heart rate, and use change in this marker as a marker for change in cardiac output. As the G.E.L. team notes, the data is mixed as to whether this is a good tool for making decisions about fluid. Further complicating this, the study uses a criterion standard that itself has had mixed results predicting cardiac output. That being said, a validation cohort using this derived cutoff would be a great next step in showing this is a useful tool. Of note, another study was just published that showed carotid flow time with an AUC of .51, literally a coin toss! It is also important to note the poor sensitivity at all cutoffs. That’s atypical, for most US measures, you can kind of scale and find a sensitive cutoff and a specific cutoff. Here, if the ccFT is less than zero you still don’t know if a patient WON’T be fluid responsive. An important limitation for ED providers is the ICU setting – the mean fluid administered was 8 liters +- 5 liters, which is not entirely comparable to what most of us will experience in the ED.