Written by: Robert J. McMickle (@RMcMickl; Medical Student IV, David Geffen School of Medicine at UCLA) and Lilly Bellman MD (@LillyBellman; Pediatric Emergency Medicine and Ultrasound Fellow, Harbor-UCLA Medical Center). // Edited By: Manpreet Singh MD (@MPrizzleER) and Stephen Alerhand MD (@SAlerhand)
Case
A 16 year-old boy with a history of sickle cell disease (SCD) complicated by multiple hospitalizations for pain crises and acute chest syndrome presents to the ED with fever and pain in his arms and legs for two days. He is tachycardic to 115 bpm and demonstrates fever to 38.3° C. His exam is otherwise unremarkable. Empiric antibiotics are administered and the patient is admitted for pain management and sepsis. On hospital day two, he develops respiratory distress, is intubated, and then transferred to the PICU. What complicated this boy’s condition? How can accurate and early diagnosis help identify and potentially prevent further respiratory complications?
Acute Chest Syndrome (ACS)
- Pulmonary complications of SCD (pulmonary hypertension and ACS) are THE leading cause of death in sickle cell patients [1,2].
- ⅓ of all SCD patients will have 1+ episodes in their lifetime and 17% of kids with SCD + fever will have ACS [2].
- Pediatric patients with SCD receive on average of 27 radiographic tests by age 18, most of which are chest X-rays (CXRs) [3].
3 major proposed mechanisms resulting in lung injury and infarction [2]:
- Pulmonary infection 54% (Chlamydia, mycoplasma, viruses, Strep pneumo)
- Bone marrow fat embolization 16%
- Intravascular sequestration of sickled erythrocytes (unknown prevalence)
How do we currently diagnose ACS? New pulmonary infiltrate by CXR* with 1+ of the following:
- Fever >38.5C (most common presenting symptom) [4]
- Chest pain
- Hypoxemia
- Respiratory symptoms (i.e. cough, tachypnea, wheezing, increased work of breathing )
*CXR currently defined as practical gold standard in evaluating for ACS, though sensitivity and specificity of bedside CXR limited to >85% and <60%, respectively when compared directly to CT (theoretical gold standard) in diagnosing ACS [5,6] ACS is SIGNIFICANTLY underestimated in the pediatric population on initial presentation
- Almost ½ of all patients will have a pre-test probability of <25% [5].
- Morris et al. have shown 61% of cases of ACS were not clinically suspected by clinicians before obtaining a CXR; physician sensitivity by history and physical alone were only 39% sensitive in diagnosing ACS [7].
- Current recommendations include repeating CXR’s if initially negative, thus increasing an individual’s cumulative radiation exposure within a single hospitalization [8]. So, why lung ultrasound (LUS)?

- Avoids repeated ionizing radiation (cumulative radiation exposure increases risk of acute lymphoblastic leukemia, especially when exposed to 3+ plain film radiographs) [9]
- LUS is able to identify smaller areas of consolidation (<1 cm) with high specificity [12]. Important given CXR often prone to missing smaller lesions, though clinical significance of subcentimeter nodules remains unclear.
- Decreased ED length-of-stay (average 47 minutes reduction in total ED time) [5] and cheaper in cost
What are we looking for with LUS?
- Scan through 6 total zones in longitudinal and transverse orientations, 3 per side with curvilinear or linear probes [12]
- Anterior along midclavicular line (clavicle to diaphragm)
- Posterior along midclavicular line (clavicle to diaphragm)
- Lateral along mid-axillary line (axilla to diaphragm)
- Pulmonary consolidations
- Loss of A-lines
- Hypoechoic areas of tissue-like echotexture/hepatization of the lung (see below)
- Disruption of the hyperechoic pleural line
- Air bronchograms
- Bright hyperechoic areas in the consolidation representing trapped air

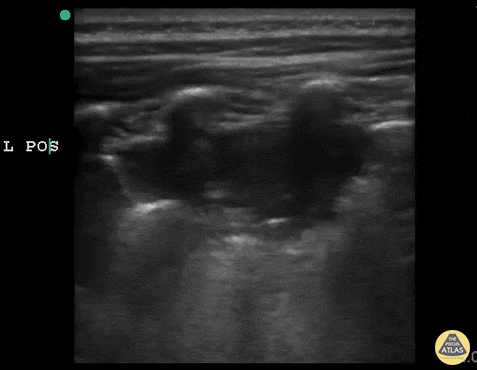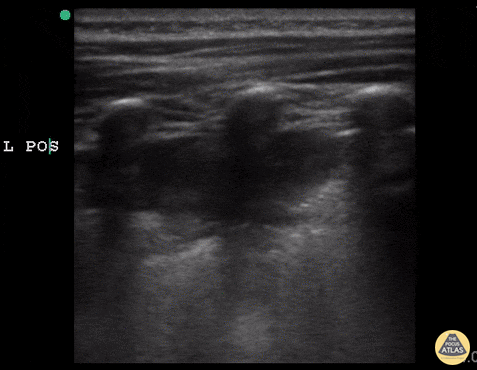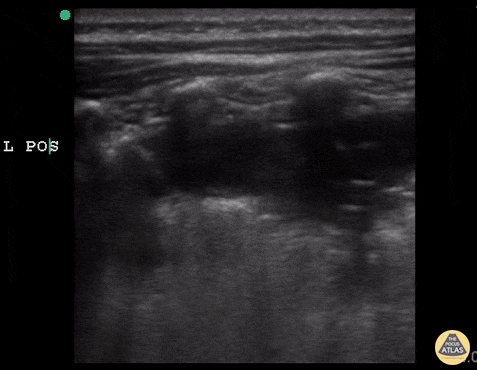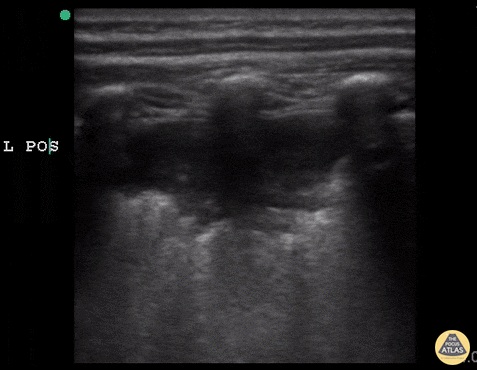
A consolidative process is seen with a hypoechoic region with posterior enhancement greater than 1 cm in an area where normal A-lines should be present. This is highly suggestive of acute chest syndrome given clinical features.” Courtesy of Dr. Sathya Subramanian, Pediatric EM Fellow – Kings Country/SUNY Downstate; access courtesy of POCUS Atlas at http://www.thepocusatlas.com/

“5 year old child with sickle cell disease. Coughing and fever for 3 days. On exam not ill appearing but decreased breath sounds over right lung. POCUS completed to evaluate for pneumonia. Hepatization of the lung clearly demonstrates consolidate process concerning for pneumonia. The beginning of the image demonstrates hepatization in the lung field. The ultrasonography then slides the probe inferiorly over normal lung past the diaphragm to the liver, demonstrating how similar lung hepatizatiion can appear compared to the actual liver.” Dr. Sathya Subramanian, Pediatric EM Fellow – Kings County/SUNY Downstate, accessed courtesy of POCUS Atlas at http://www.thepocusatas.com/

Lung consolidation with air bronchograms and a positive “shred sign” (shredded, irregular pattern at the interface between the consolidated and aerated lung fields). Courtesy of Stephen Alerhand MD.
CAUTION! Potential Pitfalls
Sonographic appearances of spleen, air in the stomach, and thymus can all be easily mistaken for areas of consolidations and can lead to false positives [5,12].

“A false-positive case determined to be operator error as the spleen and air in the stomach were interpreted as consolidation at the left lung base (Daswani et al. 2016)”.

Missed acute chest syndrome due to operator error. A consolidation was identified adjacent to the thymus on blinded expert review (Daswani et al. 2016)”.
What can we do if ACS is suspected in the ED?
Do not wait! Be prepared to manage the airway as patients may rapidly decline. The following are interventions that can be helpful in these patients. Be sure to contact your hematology colleagues.
- Respiratory support
- O2 therapy with goal SpO2 >92%
- PEEP (via High Flow Nasal Cannula, CPAP, BiPAP, or mechanical ventilation)
- Pain control
- Pulmonary toilet
- Bronchodilators
- Incentive spirometry
- Empiric antibiotics
- Fluid resuscitation
- Blood transfusion (in consultation with hematology) for:
- Severe hypoxemia
- Multilobar involvement
- Worsening clinical course
References
- Piel FB, Steinberg MH, Rees DC. Sickle Cell Disease. New England Journal of Medicine. 2017;376(16):1561-1573. doi:10.1056/nejmra1510865.
- Gladwin MT, Vichinsky E. Pulmonary Complications of Sickle Cell Disease. New England Journal of Medicine. 2008;359(21):2254-2265. doi:10.1056/nejmra0804411.
- Vetter CL, Buchanan GR, Quinn CT. Burden of diagnostic radiation exposure in children with sickle cell disease. Pediatric Blood & Cancer. 2014;61(7):1322-1324. doi:10.1002/pbc.24956.
- Chang TP, Kriengsoontorkij W, Chan LS, Wang VJ. Clinical Factors and Incidence of Acute Chest Syndrome or Pneumonia Among Children With Sickle Cell Disease Presenting With a Fever. Pediatric Emergency Care. 2013;29(7):781-786. doi:10.1097/pec.0b013e31829829f7.
- Daswani DD, Shah VP, Avner JR, Manwani DG, Kurian J, Rabiner JE. Accuracy of Point-of-care Lung Ultrasonography for Diagnosis of Acute Chest Syndrome in Pediatric Patients with Sickle Cell Disease and Fever. Academic Emergency Medicine. 2016;23(8):932-940. doi:10.1111/acem.13002.
- Dessap AM, Deux J-F, Habibi A, et al. Lung imaging during acute chest syndrome in sickle cell disease: computed tomography patterns and diagnostic accuracy of bedside chest radiograph. Thorax. 2013;69(2):144-151. doi:10.1136/thoraxjnl-2013-203775.
- Morris C, Vichinsky E, Styles L. Clinician Assessment for Acute Chest Syndrome in Febrile Patients With Sickle Cell Disease: Is It Accurate Enough? Annals of Emergency Medicine. 1999;34(1):64-69. doi:10.1016/s0196-0644(99)70273-8.
- Howard, J. , Hart, N. , Roberts‐Harewood, M. , Cummins, M. , Awogbade, M. , Davis, B. and , (2015), Guideline on the management of acute chest syndrome in sickle cell disease. Br J Haematol, 169: 492-505. doi:10.1111/bjh.13348
- Bartley K, Metayer C, Selvin S, Ducore J, Buffler P. Diagnostic X-rays and risk of childhood leukaemia. Int J Epidemiol 2010;39:1628–37.
- Razazi K, Deux J-F, Prost ND, et al. Bedside Lung Ultrasound During Acute Chest Syndrome in Sickle Cell Disease. Medicine. 2016;95(7). doi:10.1097/md.0000000000002553.
- Cohen SG, Malik ZM, Hagbom R et al. Utility of Lung Ultrasound for Evaluating Acute Chest Syndrome in Pediatric Patients with Sickle Cell Disease. Poster presented at: 2015 American Society of Hematology 57th Annual Meeting & Exposition. December 5, 2015; Orange County, CA.
- Shah VP, Tunik MG, Tsung JW. Prospective Evaluation of Point-of-Care Ultrasonography for the Diagnosis of Pneumonia in Children and Young Adults. JAMA Pediatrics. 2013;167(2):119. doi:10.1001/2013.jamapediatrics.107.






2 thoughts on “US Probe: Ultrasound for Diagnosis of Acute Chest Syndrome in Children with Sickle Cell Disease”
Pingback: LITFL Review 341 – SimWessex
Pingback: LITFL Review 341 | Edwin M. Thames