Authors: David Cisewski, MD (@dhcisewski, EM Resident Physician, Icahn SoM at Mount Sinai), Courtney Cassella, MD (@Corablacas, EM Resident Physician, Icahn SoM at Mount Sinai), and Kaushal Shah, MD (@KShah74, Associate Professor and EM Residency Program Director, Icahn SoM at Mount Sinai) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UT Southwestern Medical Center / Parkland Memorial Hospital) and Brit Long, MD (@long_brit, EM Attending Physician, SAUSHEC, USAF)
Case
A 42-year-old female with a history of lupus on prednisone and HIV compliant on HAART (CD4 537, viral load undetectable) presents to the ED with new-onset neck and back pain that started one day prior to arrival. The pain is episodic in nature, 6/10 in severity at peak, non-radiating. Associated symptoms include subjective fever, intractable hiccups, mild retrosternal burning radiating up the chest, a single episode of nausea and vomiting (non-bloody, non-bilious), and generalized fatigue. She denies chest pain, shortness of breath, headache, numbness, tingling, saddle anesthesia, bladder/bowel incontinence, diarrhea, or urinary complaints. Non-smoker and denies illicit drugs. Her vital signs are within normal limits, but she appears anxious and mildly uncomfortable. Pertinent exam findings include no midline spinal or paraspinal tenderness to palpation, pain or limitation of range of motion of the neck, or overlying erythema or skin changes. Additional exam including neurologic examination is unremarkable. Providers were concerned about spinal or pulmonary infection. The patient’s pain improved with Maalox, Pepcid, and cyclobenzaprine. Work-up including CXR, MRI, and WBC were normal. HEART score of 2 (negative troponins, no EKG findings, non-suspicious history, and 2 risk factors) calculated a 45 day risk of MACE of 0.9-1.7%. The patient was discharged with instructions for 2 day follow-up with her primary care physician.
Patient returned the following evening with sudden-onset sharp chest pain radiating to left shoulder. EKG should significant ST-elevations in anterolateral leads and elevated troponins. Patient rushed to cath lab where large LAD occlusion was identified and stented.
Final diagnosis – ST-Elevation MI c/b atypical ACS presentation
Typically Atypical ACS
Of the approximately 8 million patients presenting to the ED with chest pain each year, only 10-15% will be diagnosed with acute coronary syndrome (ACS) (1-6). Alternatively, 2% of acute MI’s are missed during the initial visit and instead discharged with an incorrect diagnosis (3). One of the most cited reasons for the missed diagnosis is the absence of chest pain and associated features – commonly referred to as ‘atypical’ ACS. These patients have delayed treatment and diagnosis that result in in-hospital mortality rates more than twice their counterparts who present with chest pain. (7)
As our understanding of ACS presentations evolve and our ability to diagnose patients advances, we are beginning to see that the terms ‘typical’ and ‘atypical’ are misleading in their use. Rather we should recognize that different subsets of the patient population – age, sex, race – result in unique presentations that must be respected and anticipated in the workup of individual patients. To say it best, “when non-classic presentations are common [within certain groups] the term atypical becomes misleading” (1). Additionally, we must recognize the pitfalls that lead to these misdiagnoses. Thus the focus of this review is on the pearls and pitfalls of ‘atypical’ ACS and specific ways it can present, including common errors that we should avoid in assessing patients for risk of ACS.
Typical vs. Atypical ACS
Classic (typical) ACS presentations include squeezing retrosternal chest pain or chest tightness, aggravated by activity and relieved by rest. As the pain from cardiac ischemia results from triggered visceral nerve fibers mapped to pathways of overlying somatic fibers in the thoracic and abdominal cavities, the presentation may include shoulder/neck/back pain, nausea, vomiting, diaphoresis, and/or dyspnea (1,8). Chest pain duration, as well as patients ability to recall time of inception, is often highly variable and a less reliable typical feature.
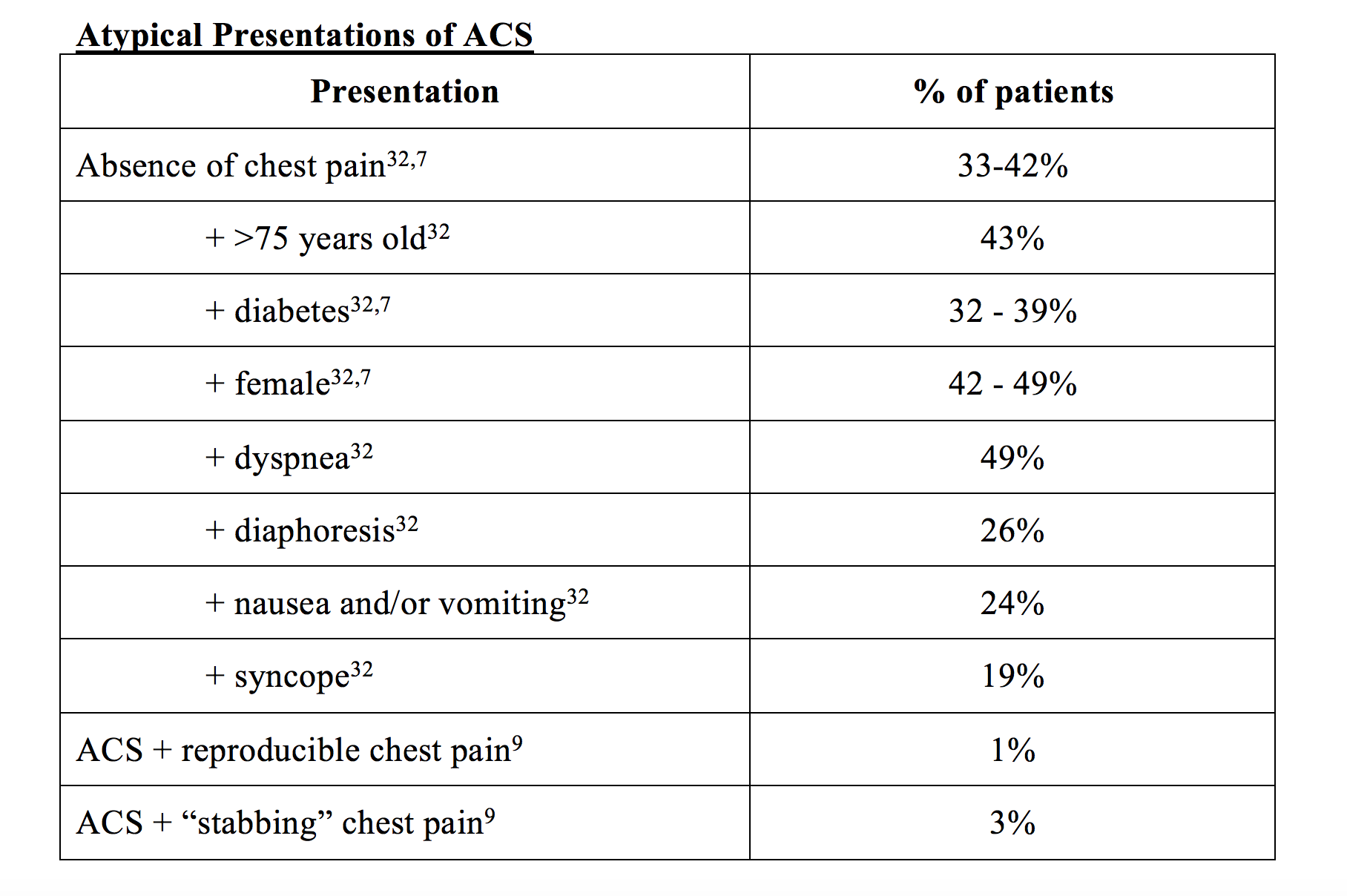
Atypical ACS presentations include the absence of chest pain, positional and reproducible pain, sharp or stabbing pain, or a presentation consisting solely of abdominal pain, nausea/vomiting, neck pain, or jaw pain. In addition, with age and under certain pathologic conditions, visceral innervation pathways become desensitized to pain acuity resulting in vague symptomatology such as generalized fatigue, particularly in older women. (9)
Absence of chest pain has been found in approximately one-third of acute MI’s and as high as 42% of female ACS patients (12% more than their matched male counterparts – 30%) (7, 22). Interestingly, studies also shown that as the population aged, the sex differences of ACS presentation between men and women decreased, indicating that age – and not sex – may play a prominent role in atypical presentation (22). Essentially, men present with ACS at a younger age than women on average, giving the misperception that atypical presentation is an innate quality of a women’s ACS presentation. When controlling for age using a multivariate analysis, women present with similar rates of typical ACS symptoms per event, but accompanied by more atypical symptoms than men, leading us to incorrectly attribute this to an ‘atypical’ presentation (22).
Another important difference between men and women is the experience of prodromal symptoms leading to an ACS event, which are experienced more often in women. One study on MI’s in women showed that 95% of the subjects experienced prodromal symptoms including fatigue (71%), sleep disturbance (48%), shortness of breath (42%), and chest discomfort (30%). (23). Though follow-up studies were inconclusive as to whether males or females experienced a higher prevalence of prodromal symptoms, studies still demonstrated that the atypical prodrome experience was significantly more pronounced (higher intensity, higher number of prodromal symptoms) in females versus males (24). This highlights the importance of sex-based assessment when determining risk for ACS.
1 – Atypical patients
The classic triad of atypical ACS patients include diabetics, elderly, women (a helpful medical school mnemonic – “sweet, old, ladies”). However, there are other high-risk groups that must not be overlooked including psychiatric (1,10,11), altered, HIV (25-27), CKD (31), SLE (28,29), and chronic glucocorticoid (29, 30) patients. For more information on atypical ACS risk factors see emDocs post Non-Traditional ACS Risk Factors: A Snake in the Grass.
2 – Atypical H&P
Sensitivity to cultural and language barriers is an essential when asking the patient to describe the quality of pain. Different terminology can be used in different languages and often the same translated word can have distinct differences in meaning among different cultures. One particular study demonstrated that 45% of the atypical presentations involved the description of “sharp” or “stabbing” chest pain, lowering the likelihood of AMI based on the ‘classic’ presentation. (12). Though this may be an ‘atypical’ presentation, in many languages this may be synonymous with the words used to described angina or an MI.
3 – Atypical EKG
When dealing with an equivocal history and physical, we often resort to the EKG as our indirect visualization of cardiac function. Increasing in popularity is the HEART score which utilizes EKG changes as one of the 5 major components in assessing the patient’s risk for future major adverse cardiac events. Typical EKG patterns of ACS include T-wave inversions and ST changes, consistent with arterial distribution. Yet studies have shown that less than 50% of patients with ACS will have a truly diagnostic EKG (14-17). As a result, the HEART score should be used in conjunction with clinical gestalt. This can be remembered by the acronym “CHEATeR”, the “C” standing for common sense; in other words, if something doesn’t seem right, don’t dismiss it!
4 – Atypical interpretation and assessment
One must also be aware of several biases when diagnosing ACS. Triage cueing occurs when EMS or nursing staff provide a diagnosis as opposed to a chief complaint. When given a diagnosis of “GERD/reflux” or “”shoulder pain”, for example, you may have an alternative mental algorithm that lowers your suspicion for ACS. In addition, premature diagnostic closure results from not being diligent to complete an H&P when a seemingly obvious diagnosis is at hand. Be wary of the patient with a history of peptic ulcers presenting with burning epigastric pain. Though Sutton’s Law may advise we consider the most obvious choice, and Occam’s Razor indicates we choose the diagnosis with the fewest assumptions, we must not forget Hickam’s Dictum: “Patient can have as many disease as they damn well please” (33)!
It is essential that we use the presenting symptom as a starting point to conversation, but that we not underestimate further signs and symptoms, nor make the assumption that the order in which the patient relays the symptoms follows any hierarchy of importance. One large study demonstrated that 47% of patients with AMI presented with chief complaints other than chest pain such as dyspnea at rest, diaphoresis, generalized weakness, light-headedness, of arm/jaw discomfort, highlighting the importance of formulating a broad differential diagnosis (11).
Patients with negative EKG’s, single negative troponins, or pain relieved by antacids are not necessarily non-ACS patients. Pain relief following treatment is often coincidental. Correlation does not necessarily imply causation – when your gut tells you something just isn’t right, go with it!
Summary
- “You can’t make the diagnosis if it’s not on the differential”; always consider atypical presentations of ACS
- Atypical presentations include nausea, vomiting, absence of chest pain, neck and back pain, or general fatigue
- Atypical risk factors include psychiatric/AMS, SLE, CKD, HIV, chronic glucocorticoid use
- Only 50% of EKGs are diagnostic in ACS
- CHEATeR = HEART + “Common sense”
- Beware of triage cueing and premature diagnostic closure when making a differential diagnosis
- If you suspect cardiac involvement, get an EKG +/- troponins and consider placing patient on observation
This post is sponsored by www.ERdocFinder.com, a supporter of FOAM and medical education, who with their sponsorship are making FOAM material more accessible to emergency physicians around the world.
References / Further Reading:
1 – Judith E. Tintinalli, et. al. (2015). Tintinalli’s Emergency Medicine, McGraw Hill.
2 – Owens PL, Barrett ML, Gibson TB, Andrews RM, Weinick RM, Mutter RL: Emergency department care in the United States: a profile of national data sources. Ann Emerg Med 56: 150, 2010. [PMID: 20074834]
3 – Pope JH, Aufderheide TP, Ruthazer R, et al: Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med 342: 1163, 2000. [PMID: 10770981]
4 – Pines JM, Isserman JA, Szyld D, Dean AJ, McCusker CM, Hollander JE: The effect of physician risk tolerance and the presence of an observation unit on decision making for ED patients with chest pain. Am J Emerg Med 28: 771, 2010. [PMID: 20837253]
5 – Fleischmann KE, Goldman L, Johnson PA, et al: Critical pathways for patients with acute chest pain at low risk. J Thromb Thrombolysis 13: 89, 2002. [PMID: 12101386]
6 – Roger VL, Go AS, Lloyd-Jones DM, et al: Heart disease and stroke statistics—2011 update: a report from the American Heart Association. Circulation 123: e18, 2011. [PMID: 21160056]
7 – Canto JG, Shlipak MG, Rogers WJ, et al: Prevalence, clinical characteristics, and mortality among patients with myocardial infarction presenting without chest pain. JAMA 283: 3223, 2000. [PMID: 10866870]
8 – Swap CJ, Nagurney JT: Value and limitations of chest pain history in the evaluation of patients with suspected acute coronary syndromes. JAMA 294: 2623, 2005. [PMID: 16304077]
9 – Lee TH, Juarez G, Cook EF, et al: Ruling out acute myocardial infarction. A prospective multicenter validation of a 12-hour strategy for patients at low risk. N Engl J Med 324: 1239, 1991. [PMID: 2014037]
10 – Canto JG, Goldberg RJ, Hand MM, et al: Symptom presentation of women with acute coronary syndromes: myth vs reality. Arch Intern Med 167: 2405, 2007. [PMID: 18071161]
11 – Gupta M, Tabas JA, Kohn MA: Presenting complaint among patients with myocardial infarction who present to an urban, public hospital emergency department. Ann Emerg Med 40: 180, 2002. [PMID: 12140497]
12 – Summers RL, Cooper GJ, Carlton FB, Andrews ME, Kolb JC: Prevalence of atypical chest pain descriptions in a population from the southern United States. Am J Med Sci 318: 142, 1999. [PMID: 10487403]
13 – Mangili A, Gerrior J, Tang AM, et al: Risk of cardiovascular disease in a cohort of HIV infected adults: a study using carotid intima-media thickness and coronary artery calcium score. Clin Infect Dis 43: 1482, 2006. [PMID: 17083026]
14 – Kontos MC, Diercks DB, Kirk JD. Emergency department and office-based evaluation of patients with chest pain. Mayo Clin Proc. 2010;85(3):248–99.
15 – Brady WJ, Roberts D, Morris F. The nondiagnostic ECG is the chest pain patient: normal and nonspecific initial ECG presentation of acute MI. Am J Emerg Med. 1999;4(17):394–7.
16 – Lee TH, Goldman L. Evaluation of the patient with acute chest pain. N Engl J Med. 2000;342(16):1187–95.
17 – Forest RS, Shofer FS, Sease KL, et al. Assessment of the standardized reporting guidelines ECG classification system: the presenting ECG predicts 30-day outcomes. Ann Emerg Med. 2004;44(3):206–12.
18 – Pope JH, Aufderheide TP, Ruthazer R, et al. Missed diagnoses of acute cardiac ischemia in the emergency department. N Engl J Med. 2000;342(16):1163–70.
19 -O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. J Am Coll Cardiol. 2013;61(4):e78–140.
20 – Boden WE, Kleiger RE, Gibson RS, et al. Electrocardiographic evolution of posterior acute myocardial infarction: importance of early precordial ST-segment depression. Am J Cardiol. 1987;59(8):782–7.
21 – Brady W, Erling B, Pollack M, et al. Electrocardiographic manifestations: acute posterior wall myocardial infarction. J Emerg Med. 2001;20(4):391–401.
22 – Canto, J. G., et al. (2014). “Time to standardize and broaden the criteria of acute coronary syndrome symptom presentations in women.” Can J Cardiol 30(7): 721-728.
23 – McSweeney JC, Cody M, O’Sullivan P, et al. Women’s early warning symptoms of acute myocardial infarction. Circulation 2003;108:2619-23.
24 – Lovlien M, Johansson I, Hole T, Schei B. Early warning signs of an acute myocardial infarction and their influence on symptoms during the acute phase, with comparisons by gender. Gender Med 2009;6:444-53.
25 – Tan, B., et al. (2017). Wellens syndrome in HIV-infected patients: Two case reports.” Medicine (Baltimore) 96(24): e7152.
26 – Klein, D., et al. (2002). “Do protease inhibitors increase the risk for coronary heart disease in patients with HIV-1 infection?” J Acquir Immune Defic Syndr 30(5): 471-477.
27 – Barbaro, G., et al. (2001). “HIV-associated cardiovascular complications: a new challenge for emergency physicians.” Am J Emerg Med 19(7): 566-574.
28 – Manzi S, Meilahn EN, Rairie JE, et al. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am J Epidemiol 1997;145:408-415
29 – Asanuma, Y., et al. (2003). “Premature coronary-artery atherosclerosis in systemic lupus erythematosus.” N Engl J Med 349(25): 2407-2415.
30 – Souverein, P. C., et al. (2004). “Use of oral glucocorticoids and risk of cardiovascular and cerebrovascular disease in a population based case-control study.” Heart 90(8): 859-865.
31 – Sarnak, M. J., et al. (2003). “Kidney disease as a risk factor for development of cardiovascular disease: a statement from the American Heart Association Councils on Kidney in Cardiovascular Disease, High Blood Pressure Research, Clinical Cardiology, and Epidemiology and Prevention.” Circulation 108(17): 2154-2169.
32 – Brieger, D., et al. (2004). “Acute coronary syndromes without chest pain, an underdiagnosed and undertreated high-risk group: insights from the Global Registry of Acute Coronary Events.” Chest 126(2): 461-469.
33 – Amal Mattu, M. A. S. C., MD; Stuart Swadron, MD, Carrie D. Tibbles, MD; Dale P. Woolridge, MD (2010). Avoiding common errors in the emergency department.







2 thoughts on “Atypical ACS Presentations: How Can We Improve?”
It doesn’t change the risk stratification here but why is the HEART score 2 and not 1?
Hx non-suspicious = 0
EKG no findings = 0
Age <45 = 0
Risk factors 2 = +1*
Troponins negative = 0
SUM = +1
* In regards to risk factors: +2 for 3 or more OR known atherosclerotic dz, which in a sense could be the case since SLE is known to cause accelerated atherosclerosis. However, that's not mentioned as the basis for a HEART score of 2 though, rather "two risk factors" are.
Pingback: emDOCs.net – Emergency Medicine EducationACS Mimics: ED Differential Diagnosis and Approach to Management - emDOCs.net - Emergency Medicine Education