Authors: Sara Hadi, MD (EM Resident Physician, SUNY Downstate Medical Center/Kings County Hospital Center) and James Willis, MD (EM Attending Physician, Associate Residency Director, SUNY Downstate Medical Center/Kings County Hospital Center) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Case
A 55-year-old male with a history of hypertension, diabetes mellitus, and hyperlipidemia presents to the ED complaining of intermittent pain in his left chest. He also complains of mild nausea and vomiting. Vital signs are the following: BP 140/90, HR 100, RR 18, T 98.6 F, SpO2 98% on room air. Physical exam shows a well-appearing male; regular rate and rhythm with no murmurs, rubs, or gallops; lungs clear to auscultation; unremarkable abdominal exam; and no extremity edema.
Given this patient’s risk factors and above presentation, you are concerned about Acute Coronary Syndrome (ACS). However, as an astute emergency physician, you realize that not all chest pain is ACS. What should you consider in the ED?
Background
ACS is a disease spectrum that includes acute myocardial infarction (ST elevation myocardial infarction or STEMI, and non-ST elevation myocardial infarction or NSTEMI) and Unstable Angina (UA). The disease affects 790,000 patients in the U.S. each year. More specifically, someone in the U.S. will suffer from a myocardial infarction every 40 seconds [i]. Classic risk factors include age (>45 for men, >55 for women), hypertension, hyperlipidemia, diabetes, smoking, family history, obesity or being overweight, and a sedentary lifestyle. However, these risk factors aren’t always reliable, and there are many other risk factors.
Atherosclerotic coronary artery disease is the main culprit, leading to plaque buildup and ischemia. The classic symptoms of ACS include chest pain (which may radiate down the upper extremities), dyspnea, nausea, and/or vomiting. In UA, patients may present with chest pain that is increasing in frequency or severity and may occur at rest. Patients with UA do not have myocardial necrosis. Women, elderly, and diabetic patients may present more atypically. They may have symptoms such as dyspnea, nausea, or fatigue, with no chest pain [ii, iii]. ECG findings of ACS include ST elevation > 1 mm in two or more contiguous leads, ST depression, T wave inversions, non-specific ST/T changes, or even a normal ECG [iv]. In cases of STEMI and NSTEMI, labs will show evidence of myocardial injury within six hours of symptom onset, or even earlier with the use of high sensitivity troponin assays [v].
ACS Mimics
Every year, there are about 8 million visits to the ED for chest pain. However, only a minority are consistent with ACS [vi-vii]. Many conditions may mimic ACS, from cardiopulmonary pathology to gastrointestinal disease. Listed below are possible mimics to consider in the ED.
Pulmonary Embolism
Acute pulmonary embolism (PE) is potentially life threatening and requires prompt recognition and treatment. Risk factors include prolonged immobilization, hypercoagulable state, history of deep vein thrombosis or PE, and malignancy. Signs and symptoms of PE include chest pain, dyspnea, syncope, hypoxia, tachycardia, and tachypnea. Patients with massive PE may present with hypotension. These signs and symptoms are nonspecific and can mimic ACS [viii]. ECG findings of T wave inversion in precordial and inferior leads and lab findings of troponinemia may be seen in both PE and ACS, further confounding the diagnosis. Others ECG findings include sinus tachycardia, or an unremarkable ECG. Clinical rules for risk stratification include Well’s Criteria and the Geneva Score. An experienced clinician may also use clinical gestalt. In patients with low pre-test probability, pulmonary embolism may be excluded by the PERC criteria or a negative quantitative d-dimer assay. In moderate to high-risk patients, the diagnosis is confirmed by CT pulmonary angiogram [ix]. Initial ED management begins with the ABCs of resuscitation and attention to abnormal vital signs. Hypoxia should be corrected with supplemental oxygen (to above 90%). Hypotensive patients should receive gentle fluid resuscitation (due to potential worsening of RV dilatation) and if necessary, vasopressor support. Anticoagulation remains the mainstay of treatment. Thrombolytics are indicated in patients with massive PE. Patients with contraindication to anticoagulation or patients with recurrent embolism despite anticoagulation are candidates for vena cava filters.

Aortic Dissection
Acute thoracic aortic dissection is a serious condition with a high morbidity and mortality rate if missed and not treated promptly. It is crucial to distinguish aortic dissection from ACS, as the two are managed very differently. Furthermore, improperly diagnosing aortic dissection as ACS can have devastating consequences if the patient were to receive anticoagulation or thrombolytic therapy. Risk factors for dissection include hypertension, Marfan syndrome, Ehlers-Danlos syndrome, Turner syndrome, bicuspid aortic valve, coarctation of aorta, trauma, and aortic valve replacement. The diagnosis is often difficult to make. Patients classically present with sudden onset of severe ripping or tearing chest pain radiating to the back. Physical exam may show elevated blood pressure, blood pressure differentials, a diastolic murmur, and neurologic deficits. A review of the accuracy of history and physical exam in the diagnosis of acute aortic dissection showed most patients have severe pain (pooled sensitivity 90%) of sudden onset (sensitivity 84%) [x]. The absence of sudden pain onset lowers the likelihood of dissection (negative likelihood ratio [LR], 0.3; 95% confidence interval [CI], 0.2-0.5). However, the clinical exam lacks the sensitivity to rule out the diagnosis. Plain chest radiographs are usually abnormal (pooled sensitivity 90%), but nonspecific [x]. Abnormal findings include a widened mediastinum (sensitivity 64%) and abnormal aortic contour (sensitivity 71%). Bedside transthoracic cardiac ultrasound may aid in diagnosis. In one study, the presence of at least one sonographic sign of type A dissection (intimal flap/intramural hematoma, ascending aortic dilatation, aortic valve insufficiency, or pericardial effusion/tamponade) had a sensitivity of 88% [xi]. However, ultrasonography is operator dependent. Diagnosis is confirmed by CT aorta with contrast in stable patients or trans-esophageal echo in unstable patients. Thoracic aortic dissection is a surgical emergency requiring immediate operative repair. Initial ED management entails placement of large bore intravenous catheters and attention to airway, breathing, and circulation. Pain control, followed by heart rate and blood pressure management (usually with opioid medications and beta-blockers, respectively) are essential in reducing aortic wall stress. Control of heart rate, followed by blood pressure control, is required. Despite surgical advances, operative mortality remains between 15-30% [xii].

Esophageal Rupture
Esophageal rupture is a serious injury that is difficult to diagnose and manage. Most cases are iatrogenic (i.e., after endoscopic procedures) but may also result from forceful vomiting (Boerhaave’s syndrome) or trauma. Clinical presentation depends on the location of injury (cervical, intrathoracic, or intraabdominal perforation). Patients may present with chest pain, epigastric pain, neck pain, dyspnea, dysphagia, subcutaneous emphysema, tachycardia, and tachypnea. Hamman’s sign, a rare “mediastinal crunch” heard during cardiac auscultation, is considered to be pathognomonic for pneumomediastinum. One of the most feared complications of intrathoracic perforations is mediastinitis, which results from infiltration of gastric contents into the mediastinum. Rupture and contamination may also extend into the pleural cavity, resulting in pleural effusions. These patients may subsequently develop fever, leukocytosis, sepsis, and shock. Diagnosis is complicated by nonspecific symptoms, which may mimic other conditions. Emergency physicians should have a high index of suspicion for perforation in patients presenting with the above signs and symptoms in the setting of recent instrumentation. Rapid diagnosis is imperative in reducing the overall 20% mortality rate. This may double if treatment is delayed more than 24 hours after perforation [xiii]. Initial diagnostic workup includes plain films of the chest and neck, which may show subcutaneous emphysema, hydrothorax, and soft tissue and mediastinal emphysema [xiv]. Confirmation can be obtained by contrast esophagography. However, a negative test in the setting of high suspicion does not rule out esophageal rupture. Definitive diagnosis may require CT scan and esophagoscopy. Management includes initiation of broad-spectrum antibiotics and emergent surgical consultation. Surgical primary repair is the most successful treatment option and reduces mortality by 50% to 70% compared to other therapies [xiii].
Cardiac Tamponade
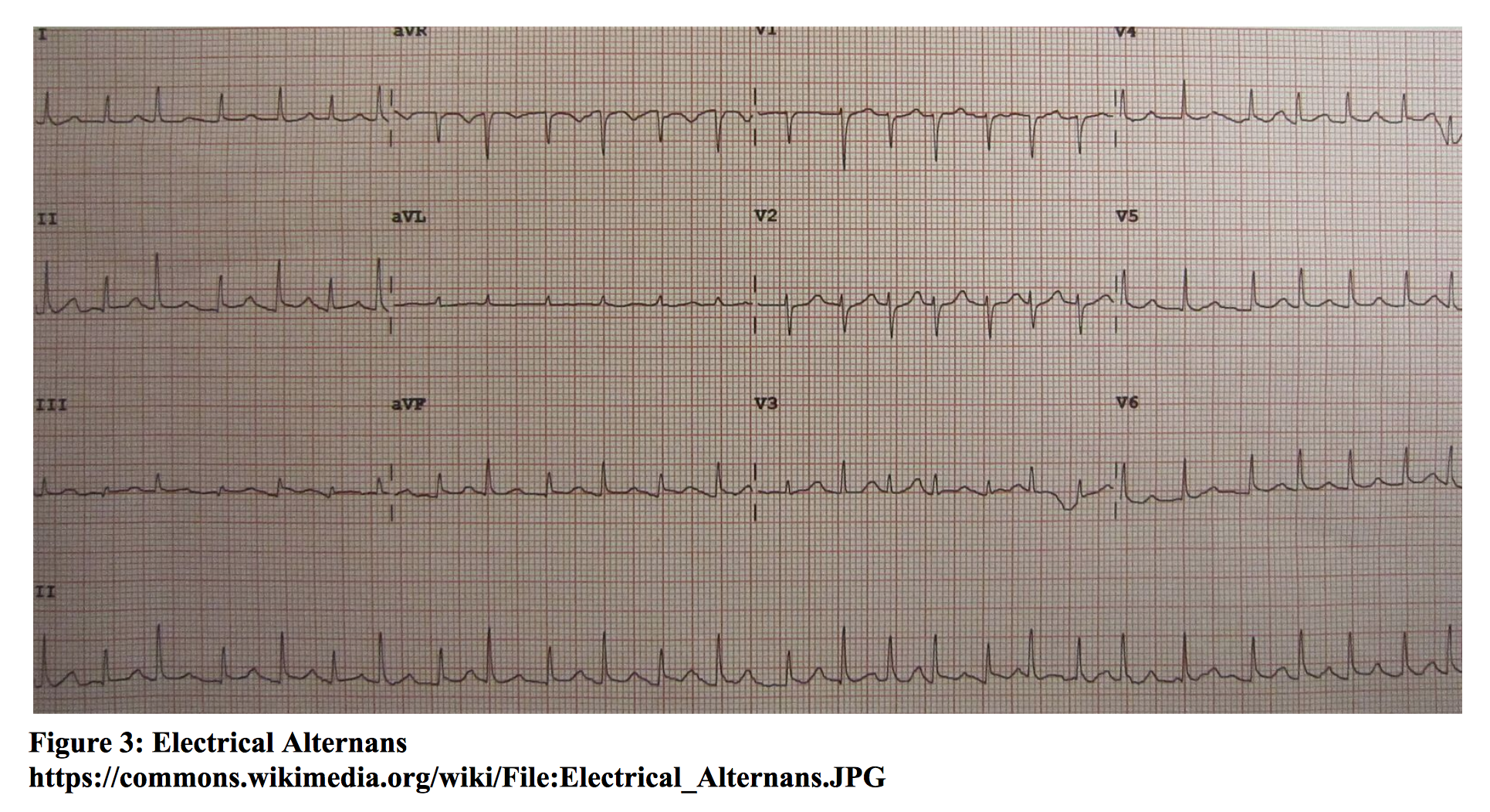
Cardiac tamponade is the accumulation of pericardial fluid in the pericardium. The parietal pericardium is able to stretch and accommodate a certain amount of intrapericardial fluid. Once the pericardium is no longer distensible, continued accumulation of fluid leads to a rise in intrapericardial pressure that is greater than normal filling pressures of the heart. Thus, rate of accumulation is a major factor in the occurrence of tamponade. This leads to compression of the heart chambers, resulting in decreased total venous return, cardiac filling, and cardiac output. Risk factors include trauma, malignancy, pericarditis, uremia, hemorrhage, systemic lupus erythematosus, and radiation therapy [xv]. Signs and symptoms include chest pain, dyspnea, tachycardia, hypotension, jugular venous distention (JVD), pericardial rub, and pulsus paradoxus (fall in systolic blood pressure of >10 mmHg during inspiration). Beck’s triad of JVD, hypotension, and muffled heart sounds is only seen in a minority of patients. ECG may show low voltage QRS complexes and variation in R wave amplitude from beat to beat (electrical alternans). Chest radiograph may or may not show an enlarged cardiac silhouette, depending on the amount of accumulated intrapericardial fluid. Echocardiography is the diagnostic test of choice. Findings include hypoechoic pericardial fluid, right atrial and/or right ventricular collapse, and an enlarged inferior vena cava without inspiratory collapse. Treatment involves drainage of fluid, preferably by pericardiocentesis, or surgical drainage [xv]. For the unstable patient, bedside pericardiocentesis must be performed.
Pericarditis
Pericarditis is inflammation of the pericardium, with many causes. In developed countries, around 80-90% cases are idiopathic [xvi]. Other causes include infections, connective tissue diseases (i.e., SLE), malignancy, uremia, drug-induced, following surgery or radiation, and post myocardial infarction. Patients present with sharp retrosternal or precordial chest pain. Pain may radiate to the neck, back (especially the trapezius ridge), arm, or left shoulder. Pain may worsen with inspiration and lying supine, and improve with sitting upright and leaning forward. Patients may also have low-grade fever, dyspnea, and a viral prodrome. Diagnosis is made if the patient has at least two of the following: pain that is typical of pericarditis, pericardial friction rub, typical ECG changes (new widespread ST elevation or PR depression), and new or worsening pericardial effusion [xv]. The different stages of ECG findings can make it difficult to distinguish pericarditis from myocardial injury. Labs may show elevated inflammatory markers (white blood cell count, ESR, CRP); however, these findings are neither sensitive nor specific. Chest radiograph may show an enlarged cardiac silhouette in the presence of a large pericardial effusion. The most severe complication of acute pericarditis is the development of cardiac tamponade from a large pericardial effusion. Treatment is with nonsteroidal anti-inflammatory drugs (NSAIDs), in conjunction with proton-pump inhibitors for gastric protection. Commonly used agents are ibuprofen, indomethacin, and aspirin. If possible, glucocorticoids should be avoided, as they increase the risk of disease recurrence [xvi]. The benefits of colchicine have also been noted in multiple studies. A recent systematic review of randomized trials has shown the efficacy of colchicine in preventing both primary and recurrent episodes of pericarditis [xvii].

Myocarditis
Myocarditis is an inflammatory disease of the myocardium and is a common cause of dilated cardiomyopathy. The disease can result from infection (viral or bacterial), inflammatory conditions, or medications. It may also be idiopathic. Signs and symptoms in the adult population include fever, myalgia, angina-type chest pain, dyspnea, syncope, tachycardia (especially sinus tachycardia out of proportion to fever), atrial or ventricular arrhythmias, heart failure, hemodynamic instability, and sudden death. These patients may have a pericardial friction rub due to pericarditis, which often accompanies myocarditis. Due to the wide spectrum of clinical manifestations, physicians should consider myocarditis in their differential diagnosis for patients with the above presentation. ECG may show non-specific ST segment and T wave abnormalities and may even mimic an acute myocardial infarction. Labs may show elevated cardiac markers in a minority of patients. One study showed high specificity (89%), but low sensitivity (34%) of Troponin I in the diagnosis of myocarditis [xviii]. Bedside transthoracic ultrasound may be used to assess for other causes of heart failure, as myocarditis lacks specific sonographic findings. Findings include regional wall-motion abnormalities; increased left ventricular wall thickness; and patterns of dilated, restrictive, and ischemic cardiomyopathies [xix]. Endomyocardial biopsy is the gold standard for diagnosis, but this invasive test is not commonly performed. Diagnosis is usually made clinically with supportive testing including ECG, chest radiograph, echocardiography, and cardiac MRI. Treatment of acute myocarditis in the ED is geared toward abating any signs and symptoms of overt heart failure. As always, initial management should be directed toward airway, breathing, and circulation. Hypoxia should be treated with supplemental oxygen. Patients presenting with signs of congestive heart failure should be treated with diuretics and preload/afterload reduction if normotensive. However, inotropic and vasopressor support may be needed.
Hypertrophic Cardiomyopathy
Hypertrophic cardiomyopathy (HCM) is characterized by hypertrophy of the left ventricle (and sometimes the right ventricle), primarily involving the interventricular septum. The disease is hereditary with an autosomal dominant inheritance pattern, affecting 1 in 500 of the general population. The disease carries an annual mortality rate of about 1% and may present during any phase of life [xx]. Patients may present with chest pain, dyspnea on exertion, palpitations, syncope, systolic heart murmur, and cardiac arrhythmias. The diagnosis is usually not made in the ED due to nonspecific symptoms that may mimic other conditions. ECG findings include left ventricular hypertrophy, left atrial enlargement, and deep Q waves in the inferior and lateral leads. Bedside cardiac ultrasound may demonstrate left ventricular hypertrophy. Other diagnostic modalities include cardiac MRI and genetic testing. Initial management in the ED should focus on ABCs and abnormal vital signs. Signs of heart failure should be addressed, and any arrhythmias should be treated accordingly. Of note, beta-blockers are the mainstay of therapy [xxi]. Beta-blockers improve diastolic filling and decreased myocardial oxygen demand. Calcium channel blockers are effective alternatives. Patients with risk factors for sudden death (i.e., prior cardiac prior cardiac arrest or spontaneous sustained ventricular tachycardia; family history of premature HCM- related death; syncope and some cases of near-syncope, particularly when exertional or recurrent) [xxi] may require admission for risk stratification and further management.
Pneumothorax
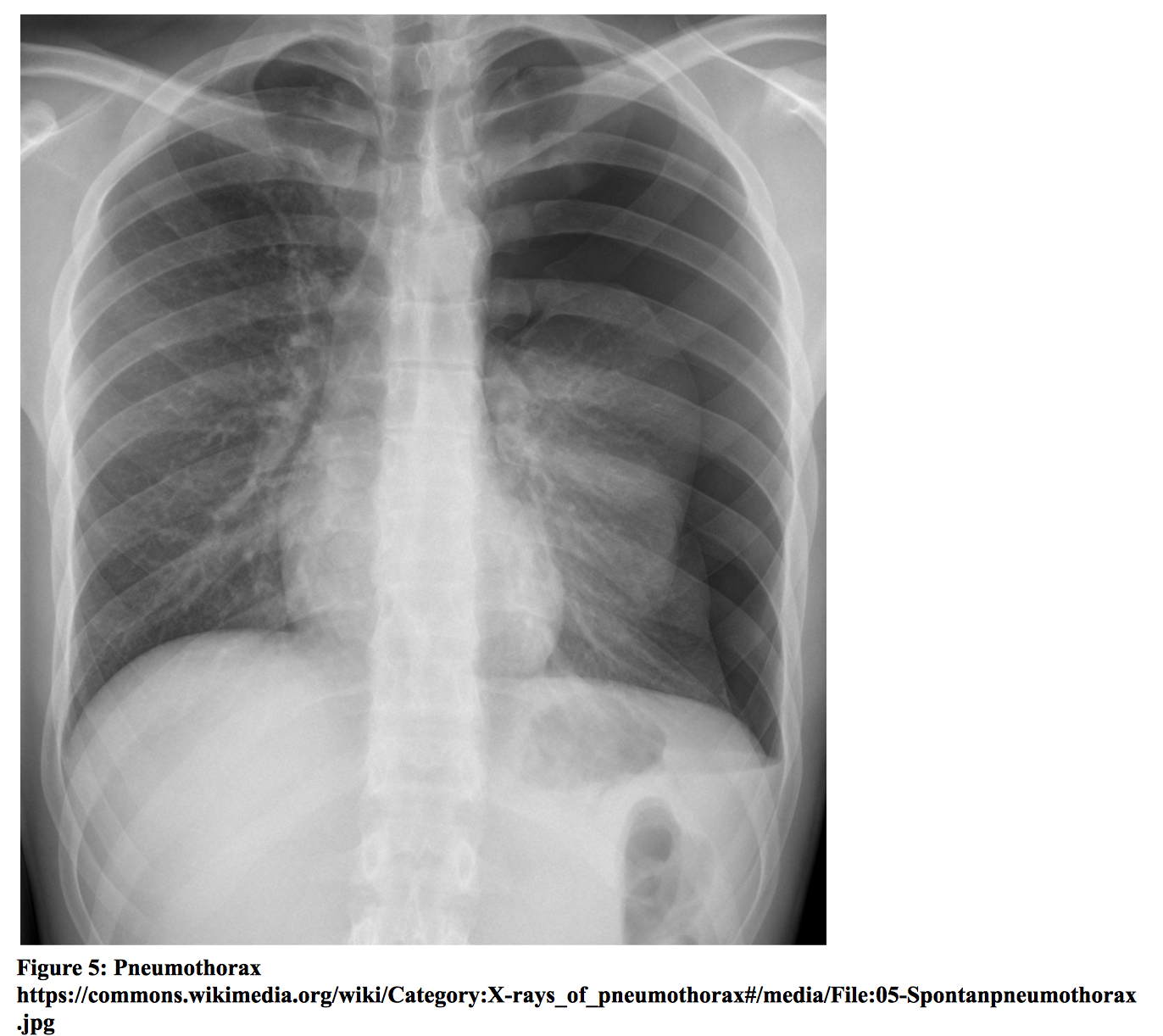
Pneumothorax is defined as air in the pleural cavity. It may occur spontaneously or as the result of underlying lung disease, trauma, or iatrogenic causes. Tension pneumothorax occurs when air accumulates in the pleural space but is unable to exit due to the formation of a one-way valve system. The accumulation of air leads to a rise in intrathoracic pressure, compressing the heart and great vessels, which reduces venous return and cardiac output. Patients may present with sudden onset chest pain, dyspnea, tachycardia, tachypnea, hypotension, hypoxia, tracheal deviation, and decreased breath sounds. Tension pneumothorax is a clinical diagnosis and requires immediate treatment with decompression to prevent hemodynamic collapse. Non-tension pneumothorax may be diagnosed by chest radiograph, which may demonstrate loss of lung markings in the periphery and the presence of a pleural line. Bedside lung ultrasound may be a superior imaging modality. Lung ultrasound will show the absence of lung sliding, which is highly suggestive of a pneumothorax. In one study comparing chest radiography and bedside ultrasound for the diagnosis of traumatic pneumothorax [xxii], ultrasound was shown to have a sensitivity of 98.1% (95% CI, 89.9% to 99.9%) and specificity of 99.2% (95% CI, 95.6% to 99.9%), while sensitivity for chest radiography was 75.5% (95% CI = 61.7% to 86.2%). Chest CT scan may be obtained for confirmation. Tension pneumothorax should be treated emergently with needle decompression followed by subsequent chest tube placement. Treatment for primary spontaneous pneumothorax depends on clinical characteristics, and management varies slightly amongst published guidelines. According to the British Thoracic Society, symptomatic patients with primary spontaneous pneumothorax may be treated with chest tube placement or needle aspiration [xxiii]. A review of simple aspiration versus chest tube drainage for primary spontaneous pneumothorax showed no significant difference in immediate success rate, early failure rate, duration of hospitalization, or one-year success rate [xxiv]. Observation or outpatient management may be appropriate for asymptomatic patients and patients with small pneumothorax (< 2 cm distance from chest wall to visceral pleural line, according to BTS guidelines) without significant symptoms. Patients with secondary spontaneous pneumothorax may require more aggressive management due to underlying lung disease and potential for persistent air leakage. Guidelines include admission with supplemental oxygen and insertion of a small-bore chest tube [xxiii].
Biliary Colic, Cholecystitis, and Cholangitis
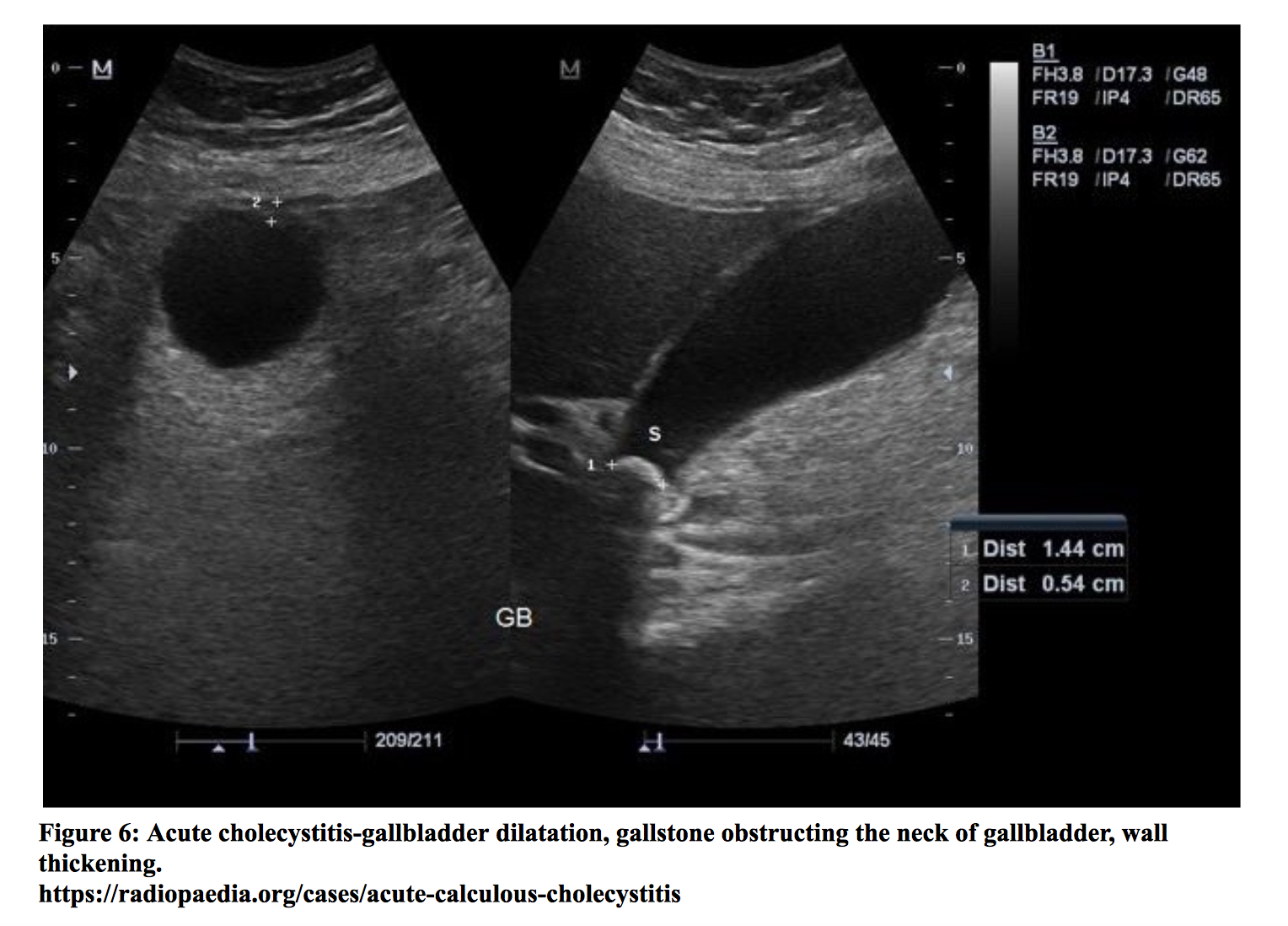
Patients with gallstones may experience intermittent right upper quadrant or epigastric pain with nausea and vomiting. Cholecystitis is inflammation of the gallbladder and usually results from an obstructing gallstone. These patients exhibit symptoms similar to those with gallstones. Physical exam may elicit tenderness in the right upper quadrant or epigastrium. Patients with cholecystitis may have a positive Murphy’s sign (cessation of deep inspiration due to pain upon palpation). It is important to note that there is no single clinical or laboratory finding that can establish or rule out the diagnosis. While fever and leukocytosis are commonly described in acute cholecystitis, they have sensitivities of 35% and 63%, respectively [xxv]. Labs may also show elevated liver function tests, including bilirubin, AST, ALT, and alkaline phosphatase levels. Some patients may develop gangrenous cholecystitis if there is necrosis of the gallbladder wall. One of the most serious complications is gallbladder perforation, which is a surgical emergency. Diagnosis of cholecystitis is most commonly made by ultrasound of the right upper quadrant, which has a reported sensitivity of 81% (95% CI = 75% to 87%) and specificity of 83% (95% CI = 74% to 89%) [xxvi]. Sonographic findings include gallbladder wall thickening, an enlarged gallbladder, a positive sonographic Murphy’s sign, gallstones, and pericholecystic fluid. In the event of a negative ultrasound report and high clinical suspicion, a HIDA scan may be obtained, which has a sensitivity of 96% (95% CI = 94% to 97%). The presence of jaundice in the setting of fever and right upper quadrant pain (Charcot’s Triad) is suggestive of cholangitis, a life-threatening illness that results from obstruction and infection of the biliary tract. In addition to these symptoms, patients may also present with hemodynamic instability and altered mental status (Reynolds’ Pentad). Patients with asymptomatic gallstones do not require treatment. ED management of symptomatic patients with gallstones includes pain control and referral for elective cholecystectomy. NSAIDs are the first line agents for pain control. A recent meta-analysis has shown NSAIDs are comparable with opioid medications in providing pain relief [xxvii]. Management of acute cholecystitis involves initiation of empiric antibiotics and surgical consultation. Patients should also receive analgesia, antiemetics for nausea/vomiting, and replacement of fluids and electrolytes. Patients with cholangitis require fluid resuscitation, treatment and monitoring for sepsis, empiric antibiotic coverage, and biliary decompression (typically with ERCP) [xxviii].
Pancreatitis
Pancreatitis is an inflammatory condition of the pancreas and may involve the surrounding tissues or cause systemic illness. The disease carries an overall mortality of about 2% [xxix]. The most common cause of pancreatitis is gallstones, followed closely by alcohol. Other causes include drugs, trauma, infection, and hypertriglyceridemia. Patients may present with epigastric pain radiating to the back, nausea, vomiting, fever, tachycardia, and diaphoresis. Diagnosis requires at least two of the following: abdominal pain consistent with acute pancreatitis, serum amylase or lipase levels at least 3 times the upper limit of normal, and findings of acute pancreatitis on computed tomography or MRI [xxx]. Of note, lipase is superior to amylase for diagnosis; it is more specific and remains elevated for longer after the onset of symptoms [xxx]. Management in the ED involves intravenous hydration, analgesia, and antiemetics for nausea/vomiting. Guidelines place an emphasis on early aggressive hydration, which is most beneficial during the first 12-24 hours [xxx]. Transabdominal ultrasound should be performed to assess for the presence of gallstones. Patients with signs of organ failure may require admission to a critical care unit.
Peptic Ulcer Disease and Gastritis
Peptic ulcer disease is characterized by ulcerations in the gastric mucosa and proximal duodenum. Most cases are caused by Helicobacter pylori infection or nonsteroidal anti-inflammatory drug (NSAID) use. Gastritis is acute or chronic inflammation of the gastric mucosa and may be caused by medications (NSAIDS, steroids, bile acids), ischemia, shock, trauma, chronic alcohol use, and pernicious anemia [xxxi]. These mucosal disorders may mimic ACS due to overlapping symptoms of vague epigastric pain, pain in the lower half of the chest, nausea, and vomiting. ACS cannot reliably be differentiated from gastrointestinal pathology on the basis of history and physical exam. One study showed the presence of burning pain or indigestion actually predicted ACS (OR 3.0, p = 0.034) [xxxii]. Physical exam may demonstrate epigastric tenderness. A rigid abdomen is highly suspicious for perforation in the setting of peptic ulcer disease. Labs may show anemia in the setting of an acute gastrointestinal hemorrhage. Plain radiographs of the abdomen may show free air in the setting of perforation. In the case of negative radiographs and high clinical suspicion, CT scan should be obtained, as this imaging modality has higher sensitivity for detecting free intra-abdominal air [xxxiii]. Upper GI endoscopy remains the gold standard for diagnosing peptic ulcer disease. Management in the ED entails initiation of a proton-pump inhibitor (PPI) or H2 blocker and primary care follow-up for patients with suspected peptic ulcer disease. Patients with alarm symptoms that suggest significant structural disease or malignancy (family history of malignancy, unintended weight loss, anemia, dysphagia or odynophagia, a palpable mass) should be referred immediately for endoscopy [xxxiv]. Patients with evidence of perforation require initiation of broad-spectrum antibiotics and urgent surgical consultation. Management of bleeding ulcers includes hemodynamic support with intravenous fluids and packed red blood cells, treatment with a PPI, and early endoscopy [xxxi].
Chest Wall Pain
Chest wall pain or musculoskeletal pain is a benign cause of chest pain, though it has not been sufficiently studied in ED settings. Costochondritis (also known as Tietze’s syndrome) is inflammation of the cartilage connecting the upper ribs with the sternum. Pain may be sharp, positional, worse with deep inspiration, and localized to a particular region of the chest. Physical exam may show reproducible chest wall tenderness that is highly localized; however, this is not a specific finding. Though pain reproduced by palpation has been shown to lower the likelihood of ACS [xxxv], this finding (along with any other single clinical or laboratory finding) cannot reliably exclude the diagnosis. The diagnosis of musculoskeletal pain is made clinically, after ruling out other serious conditions.
Panic Disorder
Panic disorder is an anxiety disorder characterized by unexpected, recurrent episodes of intense fear (“panic attacks”). Symptoms include chest pain, shortness of breath, palpitations, nausea, and nervousness. In one study evaluating the prevalence of panic disorder in ED chest pain patients, 25% of patients with chest pain met the criteria for panic disorder. 9% of the patients identified as having panic disorder received an ultimate diagnosis of ACS upon hospital discharge [xxxvi]. Patients presenting with signs and symptoms of panic disorder should receive appropriate risk stratification and testing to exclude a more serious diagnosis. Once the diagnosis of an acute panic attack has been made, physicians should educate and reassure patients regarding their symptoms. Short-term benzodiazepine therapy is also a treatment option [xxxvii]. However, these drugs should be used judiciously in the elderly population. Those with a history of substance abuse or respiratory disorders should likely not be prescribed these agents.
Anemia
Anemia is a broad group of disorders characterized by reduction of red blood cells based on the patient’s age and gender. There are many causes of anemia, including decreased production (B12 or folate deficiency, aplastic or myelodysplastic anemia), increased destruction of red blood cells (sickle cell disease, hemolytic anemia), acute blood loss due to hemorrhage, or slow chronic blood loss [xxxviii]. Patients may present with weakness, fatigue, dyspnea, palpitations, and chest pain (especially patients with pre-existing angina/coronary disease). Physical exam may show skin or mucosal pallor, tachycardia, and hypotension in severe blood loss. CBC will show a reduction in hemoglobin, hematocrit, and red blood cell counts. For symptomatic patients or patients with ongoing bleeding, ED management involves assessment of hemodynamic status and support with packed red blood cell transfusion. Transfusion guidelines vary amongst different societies. Emergency physicians should be aware of the more liberal thresholds for transfusion in patients with ACS and other co-morbid conditions [xxxix]. For more on RBC transfusion thresholds, see: http://www.emdocs.net/8959-2/.
Summary
-The patient with undifferentiated chest pain often presents as a diagnostic challenge in the ED. Patients may present with non-specific symptoms, often mimicking other conditions. Physicians must perform a focused history, physical exam, and risk stratification for appropriate evaluation.
-Remember to think broadly; you cannot make the diagnosis if it is not considered.
-History and physical exam alone cannot reliably rule out ACS. Remember that patients with burning pain or indigestion can very well have ACS!
-Management of the patient with undifferentiated chest pain begins with attention to airway, breathing, and circulation.
References/Further Reading:
- Benjamin, Emelia J., et al. “Heart Disease and Stroke Statistics-2017 Update: A Report from the American Heart Association.” Circulation, American Heart Association, Inc., 1 Jan. 2017, circ.ahajournals.org/content/early/2017/01/25/CIR.0000000000000485. Accessed 8 Aug. 2017.
- Culić, V, et al. “Symptom presentation of acute myocardial infarction: influence of sex, age, and risk factors.” American heart journal, U.S. National Library of Medicine, Dec. 2002, www.ncbi.nlm.nih.gov/pubmed/12486425. Accessed 8 Aug. 2017.
- Milner, K A, et al. “Gender and age differences in chief complaints of acute myocardial infarction (Worcester Heart Attack Study).” The American journal of cardiology, U.S. National Library of Medicine, 1 Mar. 2004, www.ncbi.nlm.nih.gov/pubmed/14996588. Accessed 9 Aug. 2017.
- Welch, MD Robert D. “Prognostic Value of a Normal or Nonspecific Initial Electrocardiogram in Acute Myocardial Infarction.” JAMA, American Medical Association, 24 Oct. 2001, jamanetwork.com/journals/jama/fullarticle/194314. Accessed 9 Aug. 2017.
- Body, Richard, et al. “Rapid Exclusion of Acute Myocardial Infarction in Patients with Undetectable Troponin Using a High-Sensitivity Assay.” Journal of the American College of Cardiology, Journal of the American College of Cardiology, 20 Sept. 2011, www.onlinejacc.org/content/58/13/1332. Accessed 11 Aug. 2017.
- Bhuiya, F A, et al. “Emergency department visits for chest pain and abdominal pain: United States, 1999-2008.” NCHS data brief, U.S. National Library of Medicine, Sept. 2010, www.ncbi.nlm.nih.gov/pubmed/20854746. Accessed 11 Aug. 2017.
- Amsterdam EA, Kirk JD, Bluemke DA, et al. “Testing of low-risk patients presenting to the emergency department with chest pain: a scientific statement from the American Heart Association.” Circulation, American Heart Association, Inc., 25 Oct. 2010, http://circ.ahajournals.org/content/122/17/1756. Accessed 11 Aug. 2017.
- Fedullo, Peter F, and Victor F Tapson. “The Evaluation of Suspected Pulmonary Embolism — NEJM.” New England Journal of Medicine, www.nejm.org/doi/full/10.1056/NEJMcp035442. Accessed 12 Aug. 2017.
- Fesmire, F M, et al. “Critical issues in the evaluation and management of adult patients presenting to the emergency department with suspected pulmonary embolism.” Annals of emergency medicine, June 2011, www.annemergmed.com/article/S0196-0644(11)00097-7/fulltext. Accessed 12 Aug. 2017.
- Klompas, MD Michael. “Does This Patient Have an Acute Thoracic Aortic Dissection?” JAMA, American Medical Association, 1 May 2002, jamanetwork.com/journals/jama/article-abstract/194885. Accessed 13 Aug. 2017.
- Nazerian, P, et al. “Diagnostic performance of emergency transthoracic focus cardiac ultrasound in suspected acute type A aortic dissection.” Internal and emergency medicine, U.S. National Library of Medicine, Sept. 2014, www.ncbi.nlm.nih.gov/pubmed/24871637. Accessed 13 Aug. 2017.
- Trimarchi, Santi, et al. “Contemporary results of surgery in acute type A aortic dissection: The International Registry of Acute Aortic Dissection experience.” Contemporary results of surgery in acute type A aortic dissection: The International Registry of Acute Aortic Dissection experience – Science Direct, www.sciencedirect.com/science/article/pii/S0022522304013212. Accessed 14 Aug. 2017.
- Brinster, C J, et al. “Evolving options in the management of esophageal perforation.” The Annals of thoracic surgery, U.S. National Library of Medicine, Apr. 2004, www.ncbi.nlm.nih.gov/pubmed/15063302. Accessed 14 Aug. 2017.
- Han, S Y, et al. “Perforation of the esophagus: correlation of site and cause with plain film findings.” AJR. American journal of roentgenology, U.S. National Library of Medicine, Sept. 1985, www.ncbi.nlm.nih.gov/pubmed/3875257. Accessed 14 Aug. 2017.
- Adler, Yehuda, et al. “2015 ESC Guidelines for the diagnosis and management of pericardial diseases: The Task Force for the Diagnosis and Management of Pericardial Diseases of the European Society of Cardiology (ESC) Endorsed by: The European Association for Cardio-Thoracic Surgery (EACTS) | European Heart Journal | Oxford Academic.” OUP Academic, Oxford University Press, 7 Nov. 2015, academic.oup.com/eurheartj/article/36/42/2921/2293375/2015-ESC-Guidelines-for-the-diagnosis-and. Accessed 14 Aug. 2017.
- LeWinter, Martin M. “Acute Pericarditis — NEJM.” New England Journal of Medicine, www.nejm.org/doi/pdf/10.1056/NEJMcp1404070. Accessed 1 Sept. 2017.
- Raval, J. et al. “The Role of Colchicine in Pericarditis – A Systematic Review and Meta-Analysis of Randomised Trials.” The Role of Colchicine in Pericarditis – A Systematic Review and Meta-Analysis of Randomised Trials – Science Direct, July 2015, www.sciencedirect.com/science/article/pii/S1443950615000487. Accessed 15 Aug. 2017.
- Smith, S C, et al. “Elevations of cardiac troponin I associated with myocarditis. Experimental and clinical correlates.” Circulation, U.S. National Library of Medicine, 7 Jan. 1997, www.ncbi.nlm.nih.gov/pubmed/8994432. Accessed 15 Aug. 2017.
- Cooper, Leslie T. “Myocarditis — NEJM.” New England Journal of Medicine, 9 Apr. 2009, www.nejm.org/doi/full/10.1056/NEJMra0800028. Accessed 1 Sept. 2017.
- Maron, MD Barry J. “Hypertrophic Cardiomyopathy.” JAMA, American Medical Association, 13 Mar. 2002, jamanetwork.com/journals/jama/fullarticle/194713. Accessed 15 Aug. 2017.
- Gersh, Bernard J., et al. “2011 ACCF/AHA Guideline for the Diagnosis and Treatment of Hypertrophic Cardiomyopathy: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines.” Circulation, American Heart Association, Inc., 1 Jan. 2011, http://circ.ahajournals.org/content/124/24/e783. Accessed 15 Aug. 2017.
- Blaivas, Michael, et al. “A Prospective Comparison of Supine Chest Radiography and Bedside Ultrasound for the Diagnosis of Traumatic Pneumothorax.” Academic Emergency Medicine, Blackwell Publishing Ltd, 28 June 2008, onlinelibrary.wiley.com/doi/10.1197/j.aem.2005.05.005/full. Accessed 15 Aug. 2017.
- MacDuff, A, et al. “Management of spontaneous pneumothorax: British Thoracic Society Pleural Disease Guideline 2010.” Thorax, U.S. National Library of Medicine, Aug. 2010, www.ncbi.nlm.nih.gov/pubmed/20696690. Accessed 15 Aug. 2017.
- Wake, Abel, et al. “Simple aspiration versus intercostal tube drainage for primary spontaneous pneumothorax in adults.” Wiley, John Wiley & Sons, Ltd, 24 Jan. 2007, onlinelibrary.wiley.com/doi/10.1002/14651858.CD004479.pub2/abstract. Accessed 16 Aug. 2017.
- Trowbridge, MD Robert L. “Does This Patient Have Acute Cholecystitis?” JAMA, American Medical Association, 1 Jan. 2003, jamanetwork.com/journals/jama/article-abstract/195707. Accessed 16 Aug. 2017.
- Kiewiet, J, et al. “A systematic review and meta-Analysis of diagnostic performance of imaging in acute cholecystitis.” Radiology, U.S. National Library of Medicine, Sept. 2012, www.ncbi.nlm.nih.gov/pubmed/22798223. Accessed 17 Aug. 2017.
- Long, Brit, and Michael D. April. “Are Nonsteroidal Anti-Inflammatory Drugs Efficacious in Relieving Biliary Colic Pain?” Annals of Emergency Medicine, 2017, doi:10.1016/j.annemergmed.2017.03.012
- Mosler, Patrick. “Management of Acute Cholangitis.” Gastroenterology & Hepatology, Millennium Medical Publishing, Feb. 2011, www.ncbi.nlm.nih.gov/pmc/articles/PMC3061017/. Accessed 31 Aug. 2017.
- Forsmark, Chris, et al. “Acute Pancreatitis — NEJM.” New England Journal of Medicine, www.nejm.org/doi/full/10.1056/NEJMra1505202. Accessed 18 Aug. 2017.
- Tenner, S., et al. “Management of Acute Pancreatitis.” Management of Acute Pancreatitis | American College of Gastroenterology, gi.org/guideline/acute-pancreatitis/. Accessed 18 Aug. 2017.
- Stapczynski, J. and Tintinalli, J. (2016). Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8th Edition. New York: McGraw-Hill Education, [2016], Chapter 78: Peptic Ulcer Disease and Gastritis
- Goodacre, S.W., et al. “Clinical predictors of acute coronary syndromes in patients with undifferentiated chest pain | QJM: An International Journal of Medicine | Oxford Academic.” OUP Academic, Oxford University Press, 1 Dec. 2003, academic.oup.com/qjmed/article/96/12/893/1533186/Clinical-predictors-of-acute-coronary-syndromes-in. Accessed 1 Aug. 2017.
- Saverio, Salomone Di, et al. “Diagnosis and treatment of perforated or bleeding peptic ulcers: 2013 WSES position paper.” World Journal of Emergency Surgery, BioMed Central, 3 Aug. 2014, wjes.biomedcentral.com/articles/10.1186/1749-7922-9-45. Accessed 22 Aug. 2017.
- ASGE, C o, et al. “The role of endoscopy in the management of patients with peptic ulcer disease.” Gastrointestinal endoscopy, U.S. National Library of Medicine, Apr. 2010, www.ncbi.nlm.nih.gov/pubmed/20363407. Accessed 23 Aug. 2017.
- Fanaroff, MD Alexander C. “Does This Patient With Chest Pain Have Acute Coronary Syndrome?” JAMA, American Medical Association, 10 Nov. 2015, jamanetwork.com/journals/jama/article-abstract/2468896. Accessed 24 Aug. 2017.
- “Panic disorder in emergency department chest pain patients: Prevalence, comorbidity, suicidal ideation, and physician recognition.” Panic disorder in emergency department chest pain patients: Prevalence, comorbidity, suicidal ideation, and physician recognition – Science Direct, www.sciencedirect.com/science/article/pii/S0002934396002240. Accessed 25 Aug. 2017.
- Stapczynski, J. and Tintinalli, J. (2016). Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8th Edition. New York: McGraw-Hill Education, [2016], Chapter 289: Mood and Anxiety Disorders
- Stapczynski, J. and Tintinalli, J. (2016). Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 8th Edition. New York: McGraw-Hill Education, [2016], Chapter 231: Anemia
- Tobian, A, et al. “Red blood cell transfusion: 2016 clinical practice guidelines from AABB.” Transfusion, U.S. National Library of Medicine, Oct. 2016, www.ncbi.nlm.nih.gov/pubmed/27739152. Accessed 27 Aug. 2017.






