Authors: Marshall Howell IV, MD (EM Resident Physician, Emory University Department of Emergency Medicine/Grady Memorial Hospital) and Eliot Blum, MD, FACEP (Assistant Professor/Attending Physician, Emory University Department of Emergency Medicine/Grady Memorial Hospital) // Reviewed by: Edward Lew, MD (@elewMD); Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case #1
A 50-year-old male with a history of obesity, hypertension, hyperlipidemia, non-insulin-dependent diabetes mellitus, obstructive sleep apnea, and Roux-en-Y gastric bypass 8 days ago presents with 24 hours of sharp mid-abdominal pain associated with nausea and vomiting. He has been following his prescribed diet and supplementation plan but has been unable to tolerate PO for the past 24 hours due to pain. Initial vitals include T 38.7°C, HR 115, BP 155/95, RR 18, SpO2 99% on room air. On exam, his abdomen is soft and tender to palpation in the epigastrium without rebound or guarding. Surgical incisions are healing well without erythema or purulence.
Case #2
Your next patient is a 56-year-old female with a history of obesity, hypertension, hyperlipidemia, and laparoscopic adjustable gastric banding 2 years ago presenting with non-radiating epigastric pain and nausea for the past 5 days. Her pain is a 6/10 and worst at night. Over the past several weeks, she has been hungrier and has been tolerating more food with meals. Her bariatric surgeon loosened her gastric band at the last office visit due to concerns for overtightening. Initial vitals include T 37°C, HR 80, BP 150/98, RR 16, SpO2 100% on room air. On exam, abdomen is tender in the epigastrium without rebound or guarding. Cardiac exam and pulses are normal. EKG from triage shows sinus rhythm with no signs of ischemia.
Questions
How is your approach to abdominal pain different for bariatric surgery patients? What unique complications are associated with these procedures? What is your threshold to involve your surgery consultants? What imaging studies would be most helpful in diagnosis? Thinking forward, what are these patients’ expected dispositions?
Background
The worldwide prevalence of individuals that are either overweight or obese has doubled since the mid-1980’s.1 Obesity increases an individual’s risk for developing cardiovascular disease2, diabetes mellitus3, several cancers4, and poor mental health.5 Bariatric surgery has risen as a popular and effective weight loss strategy in recent years. The American Bariatric Society established the most recent indications for surgical management of severe obesity to include: BMI > 40kg/m2 or BMI 35.0-39.9kg/m2 with significant cardiopulmonary comorbidities or severe diabetes mellitus.6,7
Bariatric surgery is the most effective proven treatment for severe obesity. A 2020 meta-analysis showed that bariatric surgery was associated with enduring weight loss, decreased rate of all-cause and cardiovascular mortality, and lower incidences of diabetes mellitus type 2, hypertension, dyslipidemia, and ischemic heart disease.8,9 In the US, there was a 44.3% increase in total number of bariatric procedures from 2011 to 2017, with 228,000 cases performed in 2017 alone.10
While these procedures can provide significant weight loss, improved comorbidities, and increased quality of life, they are not without their complications. Roughly 26% of bariatric surgery patients will be re-admitted within their first 2 years post-operatively.11 Many more will present to the emergency department and not require admission. Most bariatric surgery patients present to the ED with abdominal pain (46.2%), emesis (38.5%), and/or dehydration (30.8%).12 Each procedure is different in technique, and the changes in structural anatomy create unique complications not seen in other patients. These anatomical differences and patients’ body habitus also pose difficult challenges in regard to physical exam, assessment, imaging, and management. It is imperative that emergency physicians anticipate, understand, and are prepared to treat the variety of complications associated with these procedures.
Surgical Procedures
There are two ways in which current bariatric surgeries can help patients achieve weight loss: gastric restriction or mixed gastric restriction and malabsorption. Gastric restriction includes sleeve gastrectomy (SG) and laparoscopic adjustable gastric banding (LAGB). The most popular restriction and malabsorption procedure is the Roux-en-Y gastric bypass (RYGB). According to the American Society for Metabolic and Bariatric Surgery, in 2017, the most popular bariatric surgeries performed in the US were SG (59.4%), RYGB (17.8%), and LAGB (2.7%).10 Vertical banded gastroplasty and biliopancreatic diversion +/- duodenal switch are older procedures that have fallen out of favor due to their unacceptably high rates of complications.6 These 3 most popular procedures and their complications will be the focus of this article.

Sleeve Gastrectomy is a partial gastrectomy procedure. The majority of the greater curvature of the stomach is removed and the remnant of the lesser curvature is stapled or sutured to create a smaller, tubular stomach pouch. SG is technically simpler than traditional bypasses and results in lower complication rates, leading to its rise in popularity.

Roux-en-Y Gastric Bypass is performed through a complex gastric diversion. The stomach is divided into a smaller proximal pouch and a larger remnant pouch. The proximal pouch is then connected to the Roux limb, a segment of the jejunum. The stomach remnant, duodenum, and proximal jejunum are then reattached to the Roux limb distally. This procedure causes both gastric restriction as well as malabsorption via bypass of the duodenum and proximal jejunum. Digestion of food begins distal to the JJ anastomosis when the secretions from the biliopancreatic limb enter the Roux limb.
Laparoscopic Adjustable Gastric Banding is performed by surgical placement of an adjustable, 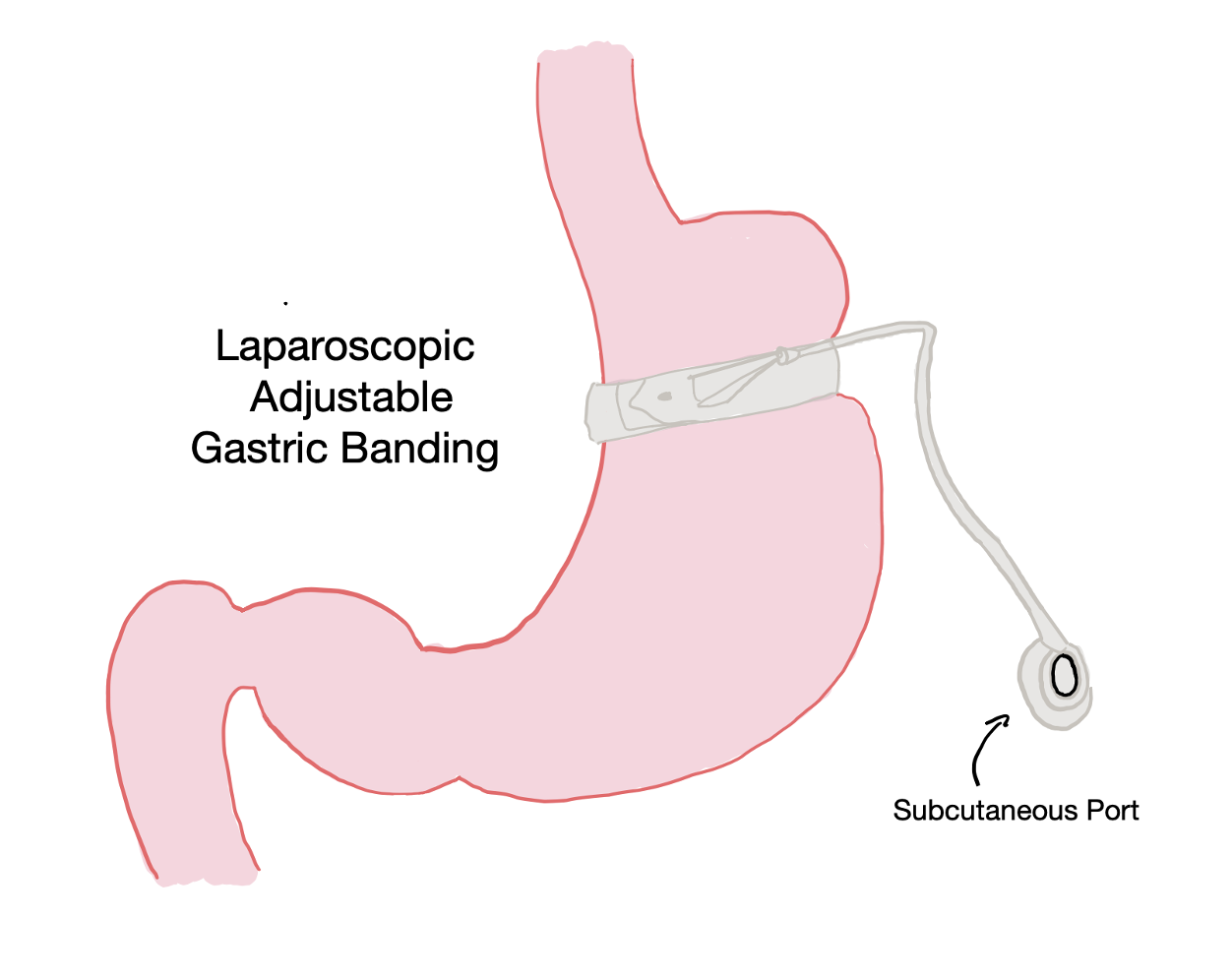 inflatable band around the upper stomach distal to the GE junction. This banding creates a smaller proximal stomach section and induces satiety at smaller food volumes.13 The band is connected to a subcutaneous port which allows for minimally invasive adjustment of the band tightness, and thus the new gastric outlet, by injecting or removing saline. This procedure has declined in popularity over the last decade due to its increased complication rate when compared to sleeve gastrectomy.
inflatable band around the upper stomach distal to the GE junction. This banding creates a smaller proximal stomach section and induces satiety at smaller food volumes.13 The band is connected to a subcutaneous port which allows for minimally invasive adjustment of the band tightness, and thus the new gastric outlet, by injecting or removing saline. This procedure has declined in popularity over the last decade due to its increased complication rate when compared to sleeve gastrectomy.
Evaluation and Management
Assessment of the post-bariatric surgery patient begins with assessment of the ABCs, IV access, monitoring of vital signs, and fluid resuscitation. Fever and tachycardia are often the first signs of anastomotic leak or intraperitoneal infection. Hypotension, tachycardia, and a new oxygen requirement could be signs of distributive shock secondary to infection. Fluid resuscitation is extremely important in these patients. Their PO intake is usually limited to induce weight loss but may be virtually non-existent when in distress.13,14 These patients also have a limited cardiopulmonary physiologic reserve and may deteriorate quickly, especially when bleeding.15
It is important to understand what surgery was performed, when the surgery occurred, who performed the surgery and where the surgery was done. Also ask if the patient is following their prescribed dietary and supplementation regimen. The type and time of the surgery are acutely relevant for developing the differential diagnosis. Patients presenting with complications within the first month post-op are likely to have bleeds, leaks, abscesses, strictures, and/or wound infections.16 Beyond the first month, patients often have infections, obstructions, herniations, ulcerations, reflux, band slippage/erosions, and/or nutritional deficiencies.16
On exam, patients may not exhibit typical findings indicative of serious intraabdominal pathology. Large amounts of abdominal fat may mask signs of peritonitis.15 Abdominal distention may also be difficult to assess when investigating possible obstruction. Abnormal vital signs, even with a benign exam, may indicate a post-surgical complication.17
When beginning the work-up, it is very important to reach out to your surgery consultants early. Diagnostic studies should include CBC, CMP, AST, ALT, amylase, lipase, and lactate in addition to your institution’s standard pre-operative labs and studies in anticipation of likely surgical repair in decompensating patients.16 Specific recommendations regarding imaging modalities will be discussed below with their associated complications. However, have a low threshold for obtaining abdominal CT in these patients given their often-unreliable abdominal exam. Bariatric surgery complications are one of the very few current indications for CT with PO contrast in the ED due to the increased sensitivity in identifying leaks and anastomotic stenoses. To best visualize the stomach, patients should receive a water-soluble contrast prep immediately prior to CT, not the typical 2-3 hours before.
You must maintain a broad differential that includes other surgical and metabolic complications. The incidence of post-operative pulmonary embolism is 2-3 times higher in obese patients.17 DVT/PE is the second leading cause of death in these patients.15 Cholelithiasis is often seen in this population as well due to decreased food intake and cholestasis. Gastric bypass patients are also at risk for nephrolithiasis due to hyperoxaluria.18 Given these patients’ typical comorbidities, also have high suspicion for cardiac disease and ischemia.
Take Home Points – Part 1
- The amount of bariatric procedures being performed in the US is rapidly increasing, and ~26% of these patients will be re-admitted within 2 years post-operatively.
- The most popular bariatric procedures are 1. Sleeve Gastrectomy 2. Roux-en-Y Gastric Bypass 3. Laparoscopic Adjustable Gastric Banding
- Initial management includes ABC’s, careful monitoring of vitals, aggressive fluid resuscitation, early surgical consult, abdominal and pre-op labs, and imaging.
- Bariatric surgery complications are one of the few ED indications for CT with PO contrast. For best results, have the patient drink the contrast on the way to the scanner instead of the typical 2-3 hour prep.
- Within the first month post-op, the most likely complications are bleeds, leaks, abscesses, strictures, and wound infections.
- Patients beyond the first month post-op most often present with infections, obstructions, herniations, ulcerations, reflux, band slippage/erosion, and nutritional deficiencies.
- Keep the differential broad, including PE, gallstones, kidney stones, and heart disease.
Diagnosis and Management based on bariatric procedure
Sleeve Gastrectomy (SG)
Staple line leaks are an early complication of SG that can present within the first few weeks post-operatively.19 The SG procedure creates a high pressure, small volume stomach wedged between two sphincters. This pressure and resulting distention can cause ischemia and delayed healing of the suture or staple line. Patients may have a wide variety of presentations from mild, nagging abdominal pain to peritonitis and hemodynamic instability. Consider a leak in SG patients with unexplained fever, tachycardia, epigastric pain, left flank or referred shoulder pain, and possibly a pleural effusion.20 First line imaging is abdominal CT with PO +/- IV contrast.17,19 Second line is upper GI series or endoscopy.19 For leaks in the first 7 days post-operatively, surgical repair or revision is the typical treatment modality, whereas endoscopic repair is preferred afterward.17 For some stable patients, surgery may be avoided with percutaneous drainage, antibiotics, and TPN until the leak heals.21
Stenosis of the long tubular stomach can cause symptoms of gastric outlet obstruction. The most common sites for stenoses are the incisura angularis and the gastroesophageal junction.22 Patients may present with dysphagia, reflux, nausea, vomiting, dehydration, and inability to tolerate PO.23 A common early cause of stenosis is over-sewing of the staple line intraoperatively with resultant edema or ischemia.22 Later, there can be fibrosis, kinking, or twisting of the tubular gastric pouch.22 The imaging modality of choice for identifying SG stenoses in the emergency department is abdominal CT with PO and IV contrast.17 This is especially the case for patients with fever and concern for associated leak. The surgical literature recommends an upper GI series in more mild progressive presentations that are limited to reflux and minimal dysphagia.17,19 This study will show dilation and accumulation of contrast proximal to the stensosis.17,19 Management includes surgical consult for endoscopic dilation of the region of stenosis.17 If the stenosed area is too long, surgical revision or conversion may be considered.

Roux-en-Y Gastric Bypass (RYGB)
Anastomotic Leaks in RYGB are typically an early complication within the first 10 days post-operatively, similar to staple line leaks in SG.19,24 The most common site for enteric leak is at the GJ anastomosis, but JJ anastomotic leaks may also occur in the early post-operative period.17,24 Patients will most often present with fever, tachycardia, nausea, vomiting, oliguria, abdominal pain +/- rebound, and hypotension.19,25 First line imaging in the ED is CT with PO and IV contrast.25 CT may show pneumoperitoneum, contrast leak, fat stranding, or abscess.26 Second line imaging is UGIS, which will show direct evidence of the leak.17,19,26 When the diagnosis of anastomotic leak is confirmed, broad spectrum antibiotics must be initiated.26 If the patient exhibits peritonitis on exam or is hemodynamically unstable, emergent surgery consult is required for repair along with aggressive fluid resuscitation.19 Similar to SG, stable patients may avoid surgical management with admission for percutaneous drainage, broad spectrum antibiotics, and either TPN or feeding through nasoenteric tube.24
Anastomotic stenosis is a complication that arises in around 6% of RYGB patients.27,28 Stenoses can occur at either the GJ or the JJ anastomosis.17 JJ stenoses occur in the first week post-operatively, while GJ stenoses can be seen several weeks to months later.17 This condition occurs when the stoma is narrowed to <1cm in diameter, likely due to ischemia or ulceration.29 This narrowing causes a bowel obstruction with proximal backup of fluid. Patients with GJ stenoses often present to the ED with epigastric pain, nausea, vomiting, reflux, progressive dysphagia, and inability to tolerate PO, including fluids.17,30 JJ stenoses typically cause dilation of the biliopancreatic limb, not the Roux limb, and result in abdominal pain due to distention but no vomiting.17 Diagnosis is typically made with an upper GI series.25,30 On UGIS, GJ stenosis will show gastric dilation with slow leak of contrast through the anastomosis, while JJ stenosis will show dilation of the biliopancreatic limb and stomach remnant. Management should include surgical consultation for endoscopic balloon dilation of a GJ stenosis or urgent surgical repair of a JJ stenosis.17,27
Marginal Ulceration refers to ulceration around an anastomotic junction, usually the GJ junction. In the normal GI tract, stomach acid is buffered by pancreatic and bilious secretions in the duodenum. After RYGB, the jejunal mucosa is exposed to unbuffered stomach acids, albeit less volume due to the smaller stomach surface area. This causes breakdown of the mucosa and ulceration. Other causes of ulceration include ischemia due to tension at the anastomosis, NSAID use, H. pylori infection, and smoking.30 Patients often present with vague abdominal pain, nausea, vomiting, GI bleeding, hematemesis, and dysphagia.30 Diagnosis and management of marginal ulceration is done via endoscopy. This allows for diagnosis, biopsy for H. pylori, and monitoring of treatment.17 If there is concern for a fistulous tract, a CT with oral contrast may be performed.30 If patient is stable, they may be discharged for endoscopy on an outpatient basis. Discharge medications should include 6 weeks of PPI therapy and sucralfate. Discharge instructions should include counseling on smoking cessation and strict discontinuation of NSAID use. Surgical intervention is required in cases of perforation or continued rebleeding despite maximal medical management.
Small Bowel Obstruction and Internal Hernia require urgent evaluation in RYGB patients. Approximately 60% of SBO in RYGB patients is due to internal hernia.31 Internal hernias can occur at any time post-operatively.19 Patients’ large weight loss after bariatric surgery leads to large potential spaces in the abdomen for herniation and anatomical rearrangement. Presenting symptoms vary widely from vague, diffuse abdominal pain with nausea and vomiting to peritonitis.25 However, obstruction is almost always intermittent, causing distinct periods of abdominal or flank pain in the same region without obvious cause.17 In the most serious cases, the hernia will cause constant pain with incarceration or strangulation of the bowel loop. Bilious emesis following RYGB is indicative of common limb obstruction. These patients need emergent surgical consultation due to the risk of distention of the biliopancreatic limb and stomach remnant rupture. The preferred imaging modality is abdominal CT with PO and IV contrast during a pain episode.17 However, CT alone is not particularly reliable for the diagnosis of internal hernia, sensitivity of 76% with 95% CI 53-90%] and specificity of 60% with 95% CI 39-78%.32 Sensitivity increases to 96% with 95% CI 78-99.8% when combining positive CT scan with neutrophilia findings.32 In a 2015 study, 52 patients underwent operation for suspected internal hernia, and it was found that 1:4 patients with internal hernia on exploration had a negative CT scan.32 Concern for internal hernia in a patient with acute abdominal pain post-RYGB merits emergent surgical consultation and exploration.
Dumping Syndrome refers to a constellation of postprandial gastrointestinal and vasomotor symptoms seen in gastric bypass patients secondary to rapid gastric emptying.33 The incidence of dumping syndrome in post-gastric bypass patients is nearly 75%.33 Symptoms occur due to the bypass or removal of the pylorus, causing rapid movement of stomach contents into the small bowel. When patients eat high carbohydrate meals, the hyperosmolar food bolus enters the intestine and causes bowel distention, fluid shifts across the bowel wall, and GI hormone hypersecretion.25 Patients may experience symptoms in two distinct time periods: early (<30min post-prandial) or late (2-4 hours post-prandial). Patients with early dumping typically present with dehydration, abdominal pain, nausea, diarrhea, dizziness, tachycardia, hypotension, and/or syncope.13,33 Late dumping is caused by a post-prandial insulin surge in response to the high carbohydrate load. This causes hypoglycemia hours after the meal. Patients with late dumping typically present with sweating, faintness, decreased concentration, and altered mental status.33 Primary treatment for dumping syndrome is fluid and electrolyte repletion in addition to dietary modification. Counseling and discharge instructions should include directions to eat small dry meals, separate solids from liquids, avoid concentrated foods, and limit sugars and lactose.13,25 Early dumping is usually self-limited and resolves 7-12 weeks post-operatively.30 For severe cases, studies have shown that octreotide treatments can abate symptoms by slowing gastric emptying.34
Metabolic and nutritional derangements are very important components of the differential diagnosis in bariatric surgery patients, particularly after gastric bypass. It is very important to ask patients if they are keeping up with their vitamin supplementation regimen. RYGB bypasses iron and calcium absorption in the duodenum, leading to these deficiencies.13 The Roux and biliopancreatic limb reconfiguration in RYGB changes intraluminal pH of the duodenum, causing intrinsic factor to remain inactive. This impairs Vitamin B12 absorption.17 Vitamin D absorption is also decreased, pre-disposing patients to early osteoporosis in the absence of supplementation. Perhaps the most important consideration for the emergency provider is the potential for thiamine deficiency. Most complications of bariatric procedures result in vomiting and decreased oral intake. These factors predispose patients to symptomatic thiamine deficiency, including Wernicke-Korsakoff Syndrome.14,35 Always give 100mg thiamine to chronic vomiting patients prophylactically prior to glucose containing fluids. If a post-RYGB patient is presenting with altered mental status, ataxic gait, ophthalmoplegia, or amnesia, consider symptomatic thiamine deficiency and begin supplementation with 500mg IV thiamine.14,36

Laparoscopic Adjustable Gastric Banding (LAGB)
Band slippage is the most common complication of LAGB, occurring in 9-13% of patients.37 In this condition, the stomach migrates upward through the band, creating a larger upstream gastric volume. This larger volume delays feelings of satiety, leading to more distention and more prolapse through the band. Patients often present to the ED with abdominal or chest pain, nausea, vomiting, reflux, and slowed weight loss.32 This diagnosis must be considered in LAGB patients with arrest of weight loss, with or without other symptoms.17 In extreme cases, the patient may have strangulated herniation through the gastric band, leading to gastric necrosis.17 Diagnosis is established with an upper GI series showing dilation of the proximal stomach pouch or a mispositioned band.17,30 Management involves urgent surgical consultation for loosening of the gastric band. This can involve removing fluid from the band via the subcutaneous port or surgical removal.
Band erosion is a later complication of LAGB, typically occurring months to years after the original procedure.38 The gastric band erodes through the wall of the stomach, typically due to overtightening of the band or irritation from band components.39 The erosion itself develops over weeks to months and may progress to gastric perforation. Patients often present to the ED with vague epigastric pain, tachycardia, fever, bleeding, and possibly port site infection.17 They may also experience arrest of expected weight loss. Diagnosis and repair are done via upper endoscopy.32 This typically involves removal of the band. If diagnosis is less certain, UGIS or CT with PO contrast can be considered. Endoscopic management is recommended for stable patients, while those with peritoneal signs from perforation will require surgical repair.
Port Site Infection and malfunction occurs in approximately 1% of LAGB patients.17 The most common source of infection is port access via transcutaneous puncture to tighten or loosen the band.17 Infection may also be a result of spread from band erosion. Patients often present to the ED with pain, purulent drainage from the port site, cellulitis, and/or fluctuant areas concerning for abscess. Cellulitis in the immediate post-operative period is most often treated with oral antibiotics.40 Late port site infections have a higher association with band erosion and do not typically respond well to antibiotics.40 The ports are foreign material inside the body, so treatment of resistant infection involves surgical removal of the port, connector tubing, and possibly the band itself via endoscopy. Once the infection has resolved, a new port, tubing, and band may be placed via laparoscopy.

Take Home Points – Part 2
- Anastomotic or staple line leaks are early complications. Patients will often present with fever, tachycardia, abdominal pain, and decreased PO. Resuscitate and get a CT scan with PO contrast if stable. Consult surgery early.
- Stenoses or gastric band slippage can be later complications. Patients will often experience abdominal pain, reflux, and decreased PO. Get an UGIS for diagnosis. Consult surgery for endoscopic dilation or band adjustment.
- Internal hernia is a high mortality complication of RYGB. Symptoms are often intermittent. CT is the best imaging modality but is still not very sensitive. Suspicion for this diagnosis merits surgical consult for exploration.
- Bypass patients may have low calcium, iron, vitamin D, vitamin B12, and thiamine. Consider these deficiencies, especially thiamine, before initiating electrolyte and fluid replacement.
References/Further Reading:
- Chooi YC, Ding C, Magkos F. The epidemiology of obesity. Metab – Clin Exp. 2019;92:6-10. doi:10.1016/j.metabol.2018.09.005
- Czernichow S, Kengne A-P, Stamatakis E, Hamer M, Batty GD. Body mass index, waist circumference, and waist-hip ratio: which is the better discriminator of cardiovascular disease mortality risk? Evidence from an individual-participant meta-analysis of 82,864 participants from nine cohort studies. Obes Rev Off J Int Assoc Study Obes. 2011;12(9):680-687. doi:10.1111/j.1467-789X.2011.00879.x
- Singh GM, Danaei G, Farzadfar F, et al. The Age-Specific Quantitative Effects of Metabolic Risk Factors on Cardiovascular Diseases and Diabetes: A Pooled Analysis. PLoS ONE. 2013;8(7). doi:10.1371/journal.pone.0065174
- Lauby-Secretan B, Scoccianti C, Loomis D, Grosse Y, Bianchini F, Straif K. Body Fatness and Cancer — Viewpoint of the IARC Working Group. N Engl J Med. 2016;375(8):794-798. doi:10.1056/NEJMsr1606602
- Anstey KJ, Cherbuin N, Budge M, Young J. Body mass index in midlife and late-life as a risk factor for dementia: a meta-analysis of prospective studies. Obes Rev. 2011;12(5):e426-e437. doi:10.1111/j.1467-789X.2010.00825.x
- Franco JVA, Ruiz PA, Palermo M, Gagner M. A Review of Studies Comparing Three Laparoscopic Procedures in Bariatric Surgery: Sleeve Gastrectomy, Roux-en-Y Gastric Bypass and Adjustable Gastric Banding. Obes Surg. 2011;21(9):1458-1468. doi:10.1007/s11695-011-0390-5
- Burguera B, Agusti A, Arner P, et al. Critical assessment of the current guidelines for the management and treatment of morbidly obese patients. J Endocrinol Invest. 2007;30(10):844-852. doi:10.1007/BF03349226
- Farrell TM, Haggerty SP, Overby DW, Kohn GP, Richardson WS, Fanelli RD. Clinical application of laparoscopic bariatric surgery: an evidence-based review. Surg Endosc. 2009;23(5):930-949. doi:10.1007/s00464-008-0217-1
- Wiggins T, Guidozzi N, Welbourn R, Ahmed AR, Markar SR. Association of bariatric surgery with all-cause mortality and incidence of obesity-related disease at a population level: A systematic review and meta-analysis. PLOS Med. 2020;17(7):e1003206. doi:10.1371/journal.pmed.1003206
- Estimate of Bariatric Surgery Numbers, 2011-2018. American Society for Metabolic and Bariatric Surgery. Published June 26, 2018. Accessed August 28, 2020. https://asmbs.org/resources/estimate-of-bariatric-surgery-numbers
- Telem DA, Talamini M, Gesten F, et al. Hospital Admissions Greater Than 30 Days Following Bariatric Surgery: Patient and Procedure Matter. Surg Endosc. 2015;29(6):1310-1315. doi:10.1007/s00464-014-3834-x
- Gonzalez-Sánchez JA, Corujo-Vázquez O, Sahai-Hernández M. Bariatric surgery patients: reasons to visit emergency department after surgery. Boletin Asoc Medica P R. 2007;99(4):279-283.
- Luber SD, Fischer DR, Venkat A. Care of the Bariatric Surgery Patient in the Emergency Department. J Emerg Med. 2008;34(1):13-20. doi:10.1016/j.jemermed.2007.03.017
- Swaminathan A, Venkat A. Complications after Bariatric Surgery. Accessed September 10, 2020. https://www.emrap.org/episode/emrap2020/complications
- Koyfman A. Management of the Post-Bariatric Surgery Patient. emDOCs.net – Emergency Medicine Education. Published December 18, 2014. Accessed September 12, 2020. http://www.emdocs.net/power-review-management-post-bariatric-surgery-patient/
- Miller A, Kim J, Moustarah F, Papasavas P, Rogers A. BARIATRIC EXAMINATION, ASSESSMENT, and MANAGEMENT in the Emergency Department. American College of Emergency Physicians. Published 2018. Accessed September 12, 2020. https://www.acep.org/patient-care/beam/
- Contival N, Menahem B, Gautier T, Le Roux Y, Alves A. Guiding the non-bariatric surgeon through complications of bariatric surgery. J Visc Surg. 2018;155(1):27-40. doi:10.1016/j.jviscsurg.2017.10.012
- Sinha MK, Collazo-Clavell ML, Rule A, et al. Hyperoxaluric nephrolithiasis is a complication of Roux-en-Y gastric bypass surgery. Kidney Int. 2007;72(1):100-107. doi:10.1038/sj.ki.5002194
- Altieri MS, Wright B, Peredo A, Pryor AD. Common weight loss procedures and their complications. Am J Emerg Med. 2018;36(3):475-479. doi:10.1016/j.ajem.2017.11.050
- Csendes A, Braghetto I, León P, Burgos AM. Management of Leaks After Laparoscopic Sleeve Gastrectomy in Patients with Obesity. J Gastrointest Surg. 2010;14(9):1343-1348. doi:10.1007/s11605-010-1249-0
- Casella G, Soricelli E, Rizzello M, et al. Nonsurgical Treatment of Staple Line Leaks after Laparoscopic Sleeve Gastrectomy. Obes Surg. 2009;19(7):821-826. doi:10.1007/s11695-009-9840-8
- Cottam D, Qureshi FG, Mattar SG, et al. Laparoscopic sleeve gastrectomy as an initial weight-loss procedure for high-risk patients with morbid obesity. Surg Endosc Interv Tech. 2006;20(6):859-863. doi:10.1007/s00464-005-0134-5
- Dapri G, Cadière GB, Himpens J. Laparoscopic Seromyotomy for Long Stenosis After Sleeve Gastrectomy with or Without Duodenal Switch. Obes Surg. 2009;19(4):495-499. doi:10.1007/s11695-009-9803-0
- Ballesta C, Berindoague R, Cabrera M, Palau M, Gonzales M. Management of Anastomotic Leaks After Laparoscopic Roux-en-Y Gastric Bypass. Obes Surg. 2008;18(6):623-630. doi:10.1007/s11695-007-9297-6
- Tintinalli J, Ma O, Yealy D, Meckler G, Stapczynski J, Cline D, eds. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide, 9th Edition. McGraw-Hill Education; 2020.
- Ashley S, Soybel D, Chen W. Postgastrectomy complications. UpToDate. Published 2019. Accessed September 7, 2020. https://www.uptodate.com/contents/postgastrectomy-complications?search=roux%20en%20y%20anastomotic%20leak&source=search_result&selectedTitle=2~150&usage_type=default&display_rank=2
- Baumann AJ, Mramba LK, Hawkins RB, et al. Endoscopic Dilation of Bariatric RNY Anastomotic Strictures: a Systematic Review and Meta-analysis. Obes Surg. 2018;28(12):4053-4063. doi:10.1007/s11695-018-3491-6
- Schneider BE, Villegas L, Blackburn GL, Mun EC, Critchlow JF, Jones DB. Laparoscopic gastric bypass surgery: outcomes. J Laparoendosc Adv Surg Tech A. 2003;13(4):247-255. doi:10.1089/109264203322333575
- Ukleja A, Afonso BB, Pimentel R, Szomstein S, Rosenthal R. Outcome of endoscopic balloon dilation of strictures after laparoscopic gastric bypass. Surg Endosc. 2008;22(8):1746-1750. doi:10.1007/s00464-008-9788-0
- Ellsmere J, Jones D, Chen W. Late complications of bariatric surgical operations. UpToDate. Published 2019. https://www.uptodate.com/contents/late-complications-of-bariatric-surgical-operations?search=bariatric%20surgery%20complications&source=search_result&selectedTitle=1~150&usage_type=default&display_rank=1#H30
- Geubbels N, Lijftogt N, Fiocco M, Leersum NJ van, Wouters MWJM, Brauw LM de. Meta-analysis of internal herniation after gastric bypass surgery. BJS Br J Surg. 2015;102(5):451-460. doi:10.1002/bjs.9738
- Altieri MS, Pryor AD, Telem DA, Hall K, Brathwaite C, Zawin M. Algorithmic approach to utilization of CT scans for detection of internal hernia in the gastric bypass patient. Surg Obes Relat Dis. 2015;11(6):1207-1211. doi:10.1016/j.soard.2015.02.010
- Berg P, McCallum R. Dumping Syndrome: A Review of the Current Concepts of Pathophysiology, Diagnosis, and Treatment. Dig Dis Sci. 2016;61(1):11-18. doi:10.1007/s10620-015-3839-x
- Arts J, Caenepeel P, Bisschops R, et al. Efficacy of the Long-Acting Repeatable Formulation of the Somatostatin Analogue Octreotide in Postoperative Dumping. Clin Gastroenterol Hepatol. 2009;7(4):432-437. doi:10.1016/j.cgh.2008.11.025
- Coupaye M, Puchaux K, Bogard C, et al. Nutritional Consequences of Adjustable Gastric Banding and Gastric Bypass: A 1-year Prospective Study. Obes Surg. 2009;19(1):56-65. doi:10.1007/s11695-008-9571-2
- Cook CCH, Hallwood PM, Thomson AD. B VITAMIN DEFICIENCY AND NEUROPSYCHIATRIC SYNDROMES IN ALCOHOL MISUSE. Alcohol Alcohol. 1998;33(4):317-336. doi:10.1093/oxfordjournals.alcalc.a008400
- Brac B, Rebibo L, Lemouel J-P, Regimbeau J-M. Laparoscopic Treatment of a Large Gastric Pouch Following Gastric Band Slippage. Obes Surg. 2016;26(12):3084-3085. doi:10.1007/s11695-016-2388-5
- Abu-Abeid S, Szold A. Laparoscopic Management of Lap-Band® Erosion. Obes Surg. 2001;11(1):87-89. doi:10.1381/096089201321454169
- Brown WA, Egberts KJ, Franke-Richard D, Thodiyil P, Anderson ML, OʼBrien PE. Erosions After Laparoscopic Adjustable Gastric Banding: Diagnosis and Management. Ann Surg. 2013;257(6):1047-1052. doi:10.1097/SLA.0b013e31826bc21b
- Eid I, Birch DW, Sharma AM, Sherman V, Karmali S. Complications associated with adjustable gastric banding for morbid obesity: a surgeon’s guide. Can J Surg. 2011;54(1):61-66. doi:10.1503/cjs.015709