Author: Erica Simon, DO, MHA (@E_M_Simon, EM Chief Resident at SAUSHEC, USAF) // Edited by: Jennifer Robertson, MD, MSEd and Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW Medical Center / Parkland Memorial Hospital)
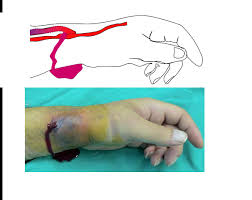
A 78-year-old male with a past medical history of coronary artery disease, hypertension and end stage renal disease presents to the emergency department (ED) with a chief complaint of “can’t stop bleeding.” The patient is seated in triage, holding a blood-soaked towel over his left arm. As you approach him, you scan his intake sheet and note the following VS: Blood pressure (BP) 72/59, heart rate (HR) 127 beats per minute, respiratory rate 18/minute, temperature (T) 102.1°Fahrenheit (F). As you lift the towel, a significant amount of blood pours from his hemodialysis fistula. You instruct the patient to continue holding firm pressure and consider possible etiologies of the bleeding.
You recognize that the patient is hemodynamically unstable, needs blood and an infectious workup, but what is the next step if direct pressure is unsuccessful?
The following review includes a few tips and tricks for identifying and treating common hemodialysis (HD) complications.
Epidemiology of Chronic Kidney Disease
In the United States, approximately 300,000-400,000 chronic kidney disease patients are maintained on HD.1-4 The process of HD requires vascular access through an arteriovenous (AV) fistula, AV graft, or central venous catheter (CVC). Today, as a result of the National Kidney Foundation’s “Fistula First” initiative, nearly 55% of HD patients utilize an AV fistula. .4,5 Significant morbidity and mortality are associated with fistula placement and recurrent cannulation. Because of the possibility of complications involving fistulas, EM physicians should recognize and treat vascular insufficiency, hemorrhage, infection, stenosis, thrombosis, aneurysms, and pseudoaneurysms.
Vascular Insufficiency – Dialysis Associated Steal Syndrome (DASS)
DASS is a complication of AV fistulas and its incidence is reported as high as 8% in the current vascular literature.3 DASS occurs secondary to retrograde flow from the artery distal to the AV anastomosis, and is seen most commonly when a large artery (brachial or superficial femoral) supplies blood through the fistula into a large, low-pressure vein.6,7 Symptoms of DASS progress from a painless cool extremity, to claudication, rest pain, and finally to tissue necrosis.6 While the majority of patients with symptomatic DASS present within one month of AV fistula creation, there are some reports detailing cases as late as one year post procedure.7
Examination of a patient with DASS includes the pathognomonic finding of a diminished or absent distal radial pulse, palpable only with compression of the dialysis access site.6 Although formal ultrasound (US) may demonstrate high-velocity, retrograde flow through the HD fistula, this is not a sensitive indicator of DASS as the diagnosis is based on clinical symptoms.6,7 The gold standard in addressing DASS is ligation of the AV access; therefore, vascular surgical consultation is required urgently or emergently according to symptom severity.7
Hemorrhage
In the HD patient, hemorrhage often arises secondary to platelet dysfunction (uremia or transient thrombocytopenia observed in the ESRD population), supra-therapeutic anticoagulation, or fistula abnormalities (infection, stenosis, aneurysms, pseudoaneurysms).1,6,8
Mechanisms for Obtaining Hemostasis
Direct Pressure
Direct pressure is the primary intervention for controlling hemorrhage. It should be applied to the site of bleeding for 5-10 minutes.1,5,7,8 If hemostasis is achieved, patients should be observed for 1-2 hours to monitor for recurrent bleeding.1,6,8
Note: The majority of hemodialysis literature advises against the application of excessive pressure as it may lead to iatrogenic fistula thrombosis.5,7 How much pressure is too much pressure? This is difficult to determine.
Topical Hemostatic Agents
Gelfoam – While there are no trials or studies specifically assessing the utilization of gelfoam in the setting of vascular access hemorrhage, its use is extensively detailed in emergency medicine literature.1,9,10 Gelfoam is a water-insoluble sponge prepared from purified porcine skin, gelatin granules, and water, which when applied to a bleeding site acts as a mechanical matrix facilitating clot formation.11 Gelfoam may be saturated with sterile saline prior to application or applied directly to the bleeding site until hemostasis is achieved.1,9 Once hemostasis has been attained, a bandage may be applied over the gelfoam (taking precautions to avoid excessive pressure which, as above, predisposes the patient to iatrogenic thrombosis).1
Chitosan – Available as HemCon®, chitosan is a non-toxic, complex carbohydrate derived from chitin. It is known to exhibit a mucoadhesive activity when applied directly to an injury site with active blood extravasation.12 One randomized control trial of 50 HD patients with vascular hemorrhage demonstrated improved time to hemostasis at the four minute treatment point after the application of HemCon® under direct pressure versus the application of direct pressure with plain gauze.13
Thrombin – Recombinant human thrombin (rhThrombin) was approved by the FDA in 2008. It helps attain hemostasis whenever control of bleeding by suture, ligature, or cautery is ineffective or impractical.14 Thrombin plays an essential role in the intrinsic and extrinsic pathways of the coagulation cascade, activating factors XI, VIII, V, XII, and I (fibrinogen), thereby facilitating hemostasis.10 Recombinant human thrombin may be utilized in conjunction with a gel foam product.1,14 The most common adverse reaction to rhThrombin is thrombosis, however, thrombosis occurring secondary to topical application in the setting of fistula hemorrhage has not been evaluated.14 (Thrombosis has been noted to occur in 1% of adult and pediatric burn patients (N=72) after application to obtain hemostasis at graft sites).14
Reversal of Supra-Therapeutic Anticoagulation – Heparin is commonly utilized in dialysis centers. Persistent oozing or bleeding occurring within hours of HD should raise concern for over anticoagulation. Experts recommend the use of intravenous protamine at a dose of 1 mg for every 100mg heparin given during dialysis. If the dose of heparin is unknown, 10-20 mg of protamine should be given in total (estimated sufficient to reverse an average dose of 1,000-2,000 units of heparin).1,7
Pharmacotherapy – Desmopressin (DDAVP) is a synthetic analogue of anti-diuretic hormone (ADH). It is FDA approved for the treatment of bleeding episodes in patients with Hemophilia A and von Willebrand’s disease (Type 1).1,9,10 While the mechanism of action has yet to be elucidated, DDAVP has been demonstrated to decrease the activated partial thromboplastin and bleeding times in uremic patients,15 as well as to prevent bleeding prior to invasive procedures in HD patients.1,16,17 This compound may be utilized by the emergency physician (in consultation with a vascular surgeon) to manage hemorrhage from AV grafts or fistulae.1,16 DDAVP should be given parenterally with a recommended dose of 0.3mcg/kg over ten minutes. It is contraindicated in patients with a history of hyponatremia, unstable angina, or congestive heart failure.1,14
If the above interventions have failed to attain hemostasis, vascular surgery should be consulted immediately for intervention.1,7,9 If ED intervention is required to prevent death, a tourniquet or strong manual pressure may be applied to the fistula site.1,9 Fistula thrombosis and loss of limb may occur in this scenario.1,7,9
Infection
Vascular access infection is a major cause of morbidity and mortality in HD patients.6,18 Current data estimate the rate of AV fistula infection as between 2-5%.10 Given the altered humoral and cell-mediated immunity occurring in chronically uremic HD patients, individuals with an AV fistula infection may lack localized inflammatory findings (rubor, calor, dolor) and present only with intermittent fever, generalized malaise, minimally elevated white blood cell count, and/or hypotension.6 The most common infecting organism in the HD population is Staphylococcus aureus, followed by Staphylococcus epidermidis and gram-negative bacteria.6,19
Suspected fistula infections should be managed aggressively with blood cultures and intravenous antibiotics. Recommended antibiotic therapy includes vancomycin (15mg/kg or 1g IV). Gentamicin (100mg IV initially and after each dialysis treatment) should be given if an infection with gram-negative organisms is suspected.6,19 Patients should be hospitalized until cultures are resulted and susceptibilities are known so that antibiotic therapy can be narrowed (reducing the incidence of vancomycin-resistant organisms).20
Ultrasound should be utilized to differentiate fistula infection from infected thrombus, local abscess, or infected hematoma which require vascular surgery consultation and oftentimes surgical management.20
Fistula Stenosis
Stenosis and thrombosis are the most common complications of AV fistulas.21 Central vein stenosis occurs in 19-41% of HD patients.22 Patients presenting to the ED with fistula stenosis may report distress secondary to upper extremity and chest wall edema. Physical examination is essential in the emergency physician’s evaluation of stenosis and it will vary according to the stenosed segment (inflow versus outflow stenosis).23 Inflow stenosis presents with a weakened radial pulse and a high pitched bruit in the systolic phase of the cardiac cycle at the site of stenosis.23 In contrast, outflow stenosis is identified as the site distal to the stenosis exhibits a bounding pulse and absent thrill. In the ED, Doppler US may be utilized for the assessment of vascular flow. Vascular surgery should be consulted for patients presenting to the emergency department with the aforementioned symptomatology, as percutaneous transluminal angioplasty is the treatment of choice.23
Fistula Thrombosis
Thrombosis is a common problem associated with vascular access.10 AV fistula thrombosis is likely to occur secondary to venous outflow stenosis (venous stenosis increases resistance to blood flow, which in turn results in increased venous pressure, decreased blood flow, and ultimately, thrombosis1). Fistula thrombosis can also occur in hours following dialysis treatment as patients are often relatively hypovolemic (venous stasis) and the fistula access site may be excessively compressed to attain hemostasis.1,8 Erythropoietin therapy, often prescribed to address the chronic anemia encountered in ESRD patients, has been noted to increase the risk of thrombotic complications secondary to increased levels of acute phase reactant proteins and chronic inflammation.7
AV fistula thrombosis is quickly identified by examining the fistula site for the absence of a bruit and thrill.1,7,8 In the case of thrombosis, vascular surgery should be consulted immediately. Management options include surgical thrombectomy versus thrombolysis with streptokinase or tissue plasminogen activator with or without angioplasty.1,8,24
Fistula Aneurysm or Pseudoaneurysm
Aneurysms form in AV fistulas secondary to repetitive cannulation and subsequent weakening of vessels walls.8,10 Patients with aneurysms may present to the emergency department reporting extremity pain, neurologic dysfunction secondary to aneurysmal impingement of surrounding nerves, significant thinning of overlying fistula skin, or hemorrhage secondary to this skin erosion.8,25
Pseudoaneurysms are pulsating extravascular hematomas resulting from dialysis site access. These are rare complications of AV fistula access.7,10 As compared to aneurysms, patients with pseudoaneurysms are more likely to present to the emergency department for vascular hemorrhage or signs and symptoms consistent with infection.7,10,25 Both AV fistula aneurysm and pseudoaneurysms can be identified with the use of Doppler US.7 Vascular surgery should be consulted for all detected vessel irregularities for consideration for operative repair.7,10
Summary
Nearly 400,000 individuals in the United States are maintained on HD therapy.1-4 With incident cases of ESRD reaching nearly 21,000 annually, and vascular access complications accounting for 16 to 25% of hospital admissions,5,7 understanding of the appropriate management of AV fistula complications is paramount for the emergency medicine physician.
Key Pearls
- Fistula complications = 16-25% of hospital admissions for HD patients
- DASS => complication of operative creation of a HD fistula
- Diagnosis based upon presentation and PE
- Cool/painful limb + diminished or absent distal radial pulse, palpable only with compression of the dialysis access site => Vascular Consult
- Hemorrhage
- #1 = direct pressure
- Gel foam, rhThrombin +/- DDAVP in consultation with a specialist are options
- Consider protamine for heparin reversal if recently received dialysis
- Infection
- Common cause of morbidity = cover for Staph and Strep
- Use US to differentiate perivascular cellulitis from local abscess, infected hematoma or infected thrombus.
- Perivascular cellulitis => Vancomycin (+ gentamycin if gram negatives suspected)
- Abscess, hematoma, thrombus => Antibiotics + vascular consult for possible OR
- Fistula Stenosis
- Patients present with extremity pain +/- chest wall edema
- Order Doppler US
- Abnormalities = consult vascular => PTCA
- Fistula Thrombosis
- Fistula has absent bruit or thrill = vascular consult => thrombectomy vs. thrombolysis
- Fistula Aneurysm or Pseudoaneurysm
- Patients present with extremity pain, compression neuropathy, or hemorrhage secondary to skin erosion.
- Doppler US => vascular consult for abnormalities
- Patients present with extremity pain, compression neuropathy, or hemorrhage secondary to skin erosion.
- Order Doppler US
- Patients present with extremity pain +/- chest wall edema
- Diagnosis based upon presentation and PE
- DASS => complication of operative creation of a HD fistula
References / Further Reading
- Larsen C, Weathers B, Schwartzwald M, Barton M. Focus on: dialysis access emergencies. American College of Emergency Physicians Clinical & Practice Management 2010. Available from https://www.acep.org/Clinical—Practice-Management/Focus-On–Dialysis-Access-Emergencies/
- Soi V, Moore C, Kumar L, Yee J. Prevention of catheter-related bloodstream infections in patients on hemodialysis: challenges and management strategies. Int J Nephrol Renovasc Dis. 2016;9:95-103.
- Leake A, Winger D, Leers S, Gupta N, and Dillavou E. Management and outcomes of dialysis access-associated steal syndrome. J Vasc Surg. 2015; 61(3): 754-760.
- KDOQI Clinical Practice Guidelines and Clinical Practice Recommendations for 2006 Updates: Hemodialysis Adequacy, Peritoneal Dialysis Adequacy and Vascular Access. Am J Kidney Dis. 2004; 48:S1-S322.
- United States Renal Data System (USRDS) Annual Data Report. Epidemiology of kidney disease in the United States. 2015. Available from: https://www.usrds.org/adr.aspx
- Dhingra R, Young E, Hulbert-Shearon T, Leavey S, Port F. Type of vascular access and mortality in US hemodialysis patients. Kidney Int. 2001; 60(4):1443-1451.
- Hodde L, Sandroni S. Emergency department evaluation and management of dialysis patient complications. J Emerg Med. 1992; 10:317-334
- Wolfson A, Singer I. Hemodialysis-related emergencies-part I. J Emerg Med. 1987; 5(6):533-543.
- Venkat, A. Challenging and Emerging Conditions in Emergency Medicine. Chichester, West Sussex: Wiley-Blackwell, 2011. Print.
- Tintinalli, J. Tintinalli’s Emergency Medicine: A Comprehensive Study Guide. New York: McGraw-Hill, 2011. Print.
- United States Food and Drug Administration. Gelfoam (absorbable gelatin powder). Device Approvals, Denials, and Clearances. 2000. Available at http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfTopic/pma/pma.cfm?num=n18286s012
- Wedmore I, McManus J, Pusateri A, Holcomb J. A special report on the chitosan-based hemostatic dressing: experience in current combat operations. J Trauma. 2006; 60(3):655-658.
- Bachtell N, Goodell T, Grunkemeier G, et al. Treatment of dialysis access puncture wound bleeding with chitosan dressings. Dialysis Transplant. 2006:35:1-6.
- United States Food and Drug Administration. Recothrom, thrombin topical (recombinant lyophilized powder for solution-for topical use only. Highlights of prescribing Information. 2008. Available from: http://www.fda.gov/downloads/Biologi…/ucm120557.pdf
- Mannucci P, Remuzzi G, Pusineri F, et al. Deamino-8-D-arginine vasopressin shortens the bleeding time in uremia. N. Engl. J. Med. 1983;308:8-12.
- Mannucci PM. Desmopressin (DDAVP) in the treatment of bleeding disorders: the first 20 years. Blood 1997;90:2515-21.
- Lethagen S. Desmopressin (DDAVP) and hemostasis. Ann. Hematol. 1994;69:173-80.
- U.S. Renal Data System: Chapter 3: Hospitalizations. In: USRDS 2012 Annual Data Report: Atlas of Chronic Kidney Disease and End-Stage Renal Disease in the United States, National Institutes of Health. Bethesda, MD, National Institute of Diabetes and Digestive and Kidney Diseases, 2012
- Ball L. Fatal vascular access hemorrhage: reducing the odds. Nephrol Nurs J. 2013; 40(4):297-303.
- Hammes M. Medical complications in hemodialysis patients requiring vascular access radiology procedures. Semin Intervent Radiol. 2004; 21(2):105-110.
- Pirozzi N, Garcia-Medina J, Hanoy M. Stenosis complicating vascular access for hemodialysis: indications for treatment. J Vasc Access. 2014; 15(2):76-82.
- Quaretti P, Galli F, Moramarco L, Corti R, Leati G, et al. Dialysis catheter related superior vena cava syndrome with patent vena cava: long term efficacy of unilateral viatorr stent-graft avoiding catheter manipulation. Korean J Radiol. 2014; 15(3):364-269.
- Vachharajani, T. Diagnosis of arteriovenous fistula dysfunction. Seminars in Dialysis. 2012; 25(4): 445-450.
- Bernal NP, Grammer ME, Mark JR, et al. Surgical thrombectomy remains a standard of care for treatment of thrombosed arteriovenous grafts. J. Surg. Res. 2008;144:362-3.
- Siedlecki A, Barker J, Allon M. Aneurysm formation in arteriovenous grafts: associations and clinical significance. Seminars in Dialysis. 2007; 20(1):73-77.





5 thoughts on “The Dialysis Patient: Managing Fistula Complications in the Emergency Department”
Bleeding from fistula puncture site:
As a retire surgeon who has constructed and managed many dialysis fistulae I can assure you that a mature fistula (several months old),the skin and subq is so tightly adherent to the graft that all you have to do is figure eight the skin puncture site with a superficially placed 5-0 suture.BUT have an assistant pressure occlude proximal AND distal to the puncture site until no more bleeding is seen BEFORE you place the figure of eight stitch.
Because the skin and subq are so tightly adherent you will NOT be creating a contained spreading hematoma.
Bleeding from fistula puncture site:
As a retire surgeon who has constructed and managed many dialysis fistulae I can assure you that a mature fistula (several months old),the skin and subq is so tightly adherent to the graft that all you have to do is figure eight the skin puncture site with a superficially placed 5-0 suture.BUT have an assistant pressure occlude proximal AND distal to the puncture site until no more bleeding is seen BEFORE you place the figure of eight stitch.
Because the skin and subq are so tightly adherent you will NOT be creating a contained spreading hematoma.
Also elevating the arm decreases the pressure in the fistula and helps make the compression more effective
Pingback: Länkar v41-42 | Internmedicin
Pingback: Complications of Hemodialysis Access - County EM