Author: Thomas Schaeffer, MD (Emergency Medicine Resident, UT Southwestern, Dallas, TX) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Welcome to EM@3AM, an emDOCs series designed to foster your working knowledge by providing an expedited review of clinical basics. We’ll keep it short, while you keep that EM brain sharp.
A 65-year-old male with a history of type 2 diabetes, hypertension, and a 40 pack-year smoking history presents with altered mental status and right sided weakness to both upper and lower extremities. Symptoms began approximately two hours earlier while eating dinner when the patient began to complain of a severe headache. Since then, the patient has become more confused and is now unable to walk normally. No prior history of similar symptoms. Endorses nausea. Denies fever, chest pain, shortness of breath, abdominal pain. Seemingly healthy immediately prior to symptom onset.
Vital Signs: BP 202/98, HR 89, RR 12, T 36.7, SpO2 98% RA.
Physical exam reveals an uncomfortable gentleman who is able to state his name, but unable to give the correct date or his location. He has profound right sided weakness in both upper and lower extremities. While speaking with the patient, he has an episode of NBNB vomiting.
What’s your next step?
Answer: Spontaneous Intracerebral Hemorrhage (Hemorrhagic Stroke); needs emergent head imaging and serum glucose assessment
Background: Injury or rupture of blood vessel into brain parenchyma resulting in bleeding that causes reduced blood flow to a particular region of the brain or other part of the CNS. Typically causes neurological impairment and long term disability.
Pathophysiology: Vessel rupture secondary to small vessel disease, long standing hypertension, AVM, amyloid angiopathy, or sympathomimetic substance abuse. Injury, of course, can be augmented with concomitant anticoagulant use. In addition to reduced blood flow, mass effect from the initial bleed may cause injury. Activation of the coagulation cascade, inflammatory cytokines, and disruption of the blood brain barrier can also result in additional intra-parenchymal edema and exacerbate injury.
Anatomy: Blood flow to the brain is divided into the anterior and posterior circulations. Arterial supply to the anterior circulation originates from the carotid arteries and supplies 80% of brain tissue. The internal carotid artery branches in to the ophthalmic artery (optic nerve and retina), anterior cerebral artery (medial aspects of cerebral hemispheres and anterior aspect of parietal lobe), and middle cerebral artery (frontoparietal lobe, anterotemporal lobe, branches to basal ganglia). Posterior circulation originates from the bilateral vertebral arteries that join to form the basilar artery. The basilar artery then bifurcates into the posterior cerebral arteries. The posterior circulation supplies the brainstem, cerebellum, and occipital cortex. Anterior and posterior circulation meet at the Circle of Willis. Extent of injury depends on location of hemorrhage and available collateral circulation.
History and Exam:
- Usually sudden onset and include symptoms of increased intracranial pressure, such as headache, nausea, vomiting, and altered mental status.
- Assess blood pressure, as most patients have history significant for long-standing hypertension and present with significantly elevated pressures.
- May also have focal neurological deficits, which commonly include sensory and motor dysfunction contralateral to site of injury. However, cannot reliably distinguish from ischemic stroke based on physical exam alone.
- Symptoms can evolve and worsen over minutes to hours after initial insult. Seizures, coma, and death can occur as a result of significant mass effect or increase in intracranial pressure or brainstem herniation.
Differential: Ischemic stroke, traumatic injury, tumor (primary or metastases) or other intracranial mass, migraine headache, seizure disorder with possible Todd’s paralysis, hypertensive or Wernicke’s encephalopathy, hypoglycemia, electrolyte disorder, infection, drug toxicity.
Diagnostics:
- Upon initial assessment, providers must determine time of last known normal and active medications (especially blood thinners) that the patient is taking. The patient should also be assigned a value on the NIH Stroke Scale, (which can be found at MD Calc to help guide treatment.
- If initial suspicion for intracranial hemorrhage (or stroke) is high, the single most important diagnostic test is a non-contrast CT Head, which can reliably detect acute intracerebral hemorrhages and should be ordered without delay.
- Other diagnostic tests can be utilized to evaluate for ICH mimics:
- CMP
- CBC
- Coagulation Studies
- Blood Glucose
- Urine Tox
- EKG
- Troponins
- ECHO
- Advanced Intracranial Imaging
- Airway management. In setting of AMS, should patient become somnolent or unable to protect airway, intubate. May use short acting post-intubation sedative so as to preserve ability to perform serial neurological examinations.
- Blood Pressure Reduction. Balance between preventing ongoing bleed and preserving cerebral perfusion. Current guidelines suggest that for:
- SBP>200 or MAP>150 treat with continuous IV infusion of BP lowering agent.
- SBP>180 or MAP>130 with evidence of increased intracranial pressure, treat with continuous IV infusion or intermittent doses to preserve cerebral perfusion pressure to >60-80 mmHg.
- SBP>180 or MAP>130 with no evidence of increased intracranial pressure, treat to goal BP of approximately 160/90 or MAP of 110. Has also been shown that SBP may be lowered to 140 with no increase in adverse events.
- Suggested agents selected for short half-lives and ease of titratability include:
- Labetalol- 10-20 mg IV over 1-2 minutes. Repeat dose every 10-15 mintues to a maximum of 300 mg
- Esmolol- 1 mg/kg bolus over 30 seconds. Followed by 150 mcg/kg/min continuous infusion (may titrate up to 300 mcg/kg/min)
- Nicardipine- continuous infusion starting at 5 mg/hr to maximum of 15 mg/hr
- Avoid hypotension, as SBP < 120 mm Hg is associated with worse outcomes.
- Reversal of Coagulopathy. Many patients are on anticoagulation agents at initial presentation.
- Warfarin- Check coagulation studies
- Vitamin K- 10 mg IV
- Fresh Frozen Plasma: 2-4 units
- Prothrombin Complex Concentrate: 50 units/kg (dosing may vary)
- Heparin
- Protamine- 1 mg per 100 units of heparin given in past 2-3 hours to a maximum dose of 50 mg
- Dabigitran
- Idarucizumab- 2.5 g IV given twice over 10-15 minutes
- Antiplatelet Agents (Aspirin, Clopidogrel, etc.)
- See recent PATCH trial, for which platelets were associated with increased mortality
- Consider desmopressin
- Consider platelet transfusion for levels < 30,000-50,000 in consultation with neurosurgery
- Reduce ICP
- Elevate head of bed
- Hyperventilation as temporizing measure
- Mannitol- 0.25 – 1 g/kg
- Hypertonic Saline- 3%; intermittent boluses; titrate to sodium of 145-155 mEq
- Neurosurgical consult for possible evacuation
- Treat seizures with antiepileptic medications
- Avoid hyperthermia
- Glucose control PRN with insulin
- Consider naloxone
- Warfarin- Check coagulation studies
Complications:
- Perihematomal Edema- May develop in the first few days to weeks after initial injury as a result of inflammation, thrombin breakdown, and blood brain barrier disruption. Can result in further increased intracerebral pressure, mass effect, and midline shift.
- Seizure- 16% of patients will have clinical seizure within one week of injury. However, prophylactic anti-seizure medications are not recommended.
- Prognosis- Mortality rate of 40% at one month.
Disposition: Patients will need to be admitted to an intensive care ward with neurosurgery team in close consult.

An 82-year-old man presents with progressive altered mental status and vomiting that started 2 hours ago. On examination, he will open his eyes slightly to painful stimuli but will not follow commands or speak. He has a left gaze deviation. He is intubated shortly after arrival for airway protection. Head CT shows a large intracerebral hemorrhage with midline shift. What ECG finding is most likely to be seen in this patient?
A) Deep inverted T waves in V1-V6
B) Prolonged PR interval
C) Shortened QT interval
D) Sinus tachycardia
Answer: A
The classic presentation for intracerebral hemorrhage is acute onset of headache, vomiting, neurologic deficits and elevated blood pressure. Almost half of patients have significant growth in the size of hemorrhage within the first few hours on onset, making continual assessment of mental status and airway a key component in managing these patients. In addition to airway management and neuroimaging, an electrocardiogram is indicated in all patients with signs and symptoms of stroke. Classic ECG findings in patients with intracerebral hemorrhage and resultant increased intracranial pressure includes widespread giant T wave inversions (“cerebral T waves”), QT prolongation and bradycardia.
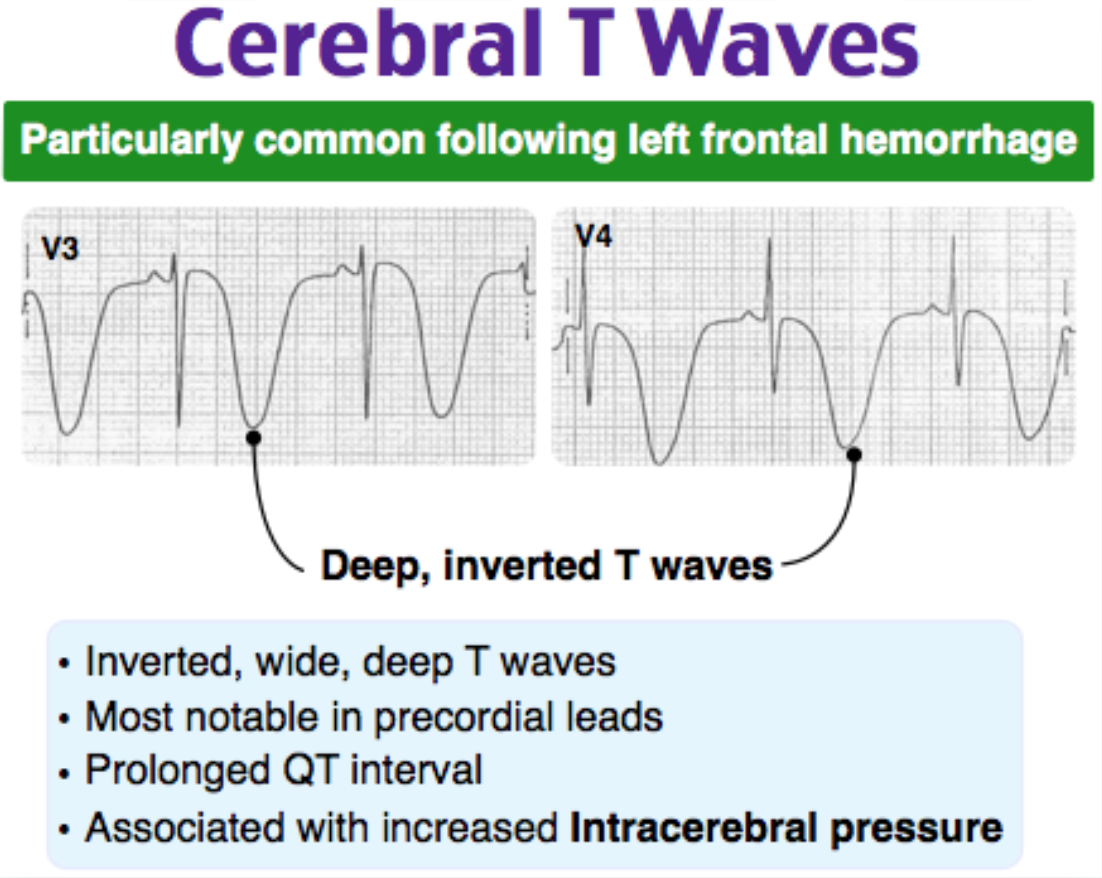
A prolonged PR interval (B) indicates delayed conduction through the AV node. Intracerebral hemorrhage and increased intracranial pressure do not typically affect the PR interval. QT prolongation is typically seen, not a shortened QT interval (C). Cushing reflex – bradycardia, hypertension and respiratory depression – is a sign of impending brain herniation due to increased intracranial pressure. While sinus tachycardia (D) can be seen, it is less common.
Further Reading:
Dastur, C. K. & Yu, W. (2017). Current management of spontaneous intracerebral haemorrhage. Stroke and Vascular Neurology; e000047. DOI: 10.1136/svn-2016-000047.
Heit JJ, Iv M, Wintermark M. Imaging of Intracranial Hemorrhage. Journal of Stroke. 2017;19(1):11-27. doi:10.5853/jos.2016.00563.
Marx, J. A., & Rosen, P. (2018). Rosen’s emergency medicine: Concepts and clinical practice (9th ed.). Philadelphia, PA: Elsevier/Saunders.
Tintinalli, J. E., Stapczynski, J. S., Ma, O. J., Yealy, D. M., Meckler, G. D., & Cline, D. (2016). Tintinalli’s emergency medicine: A comprehensive study guide (Eighth edition.). New York: McGraw-Hill Education.
Venkatasubramanian, C., Mlynash, M., Finley-Caulfield, A., Eyngorn, I., Kalimuthu, R., Snider, R. W., & Wijman, C. A. (2011). Natural history of perihematomal edema following intracerebral hemorrhage measured by serial magnetic resonance imaging. Stroke; a Journal of Cerebral Circulation, 42(1), 73–80. http://doi.org/10.1161/STROKEAHA.110.590646






1 thought on “EM@3AM: Spontaneous Intracerebral Hemorrhage”
Pingback: December FOAMed - FRCEM Success