Originally published at Pediatric Emergency Playbook on October 1, 2017 – Visit to listen to accompanying podcast. Reposted with permission.
Follow Dr. Tim Horeczko on twitter @EMTogether
Details in Audio:
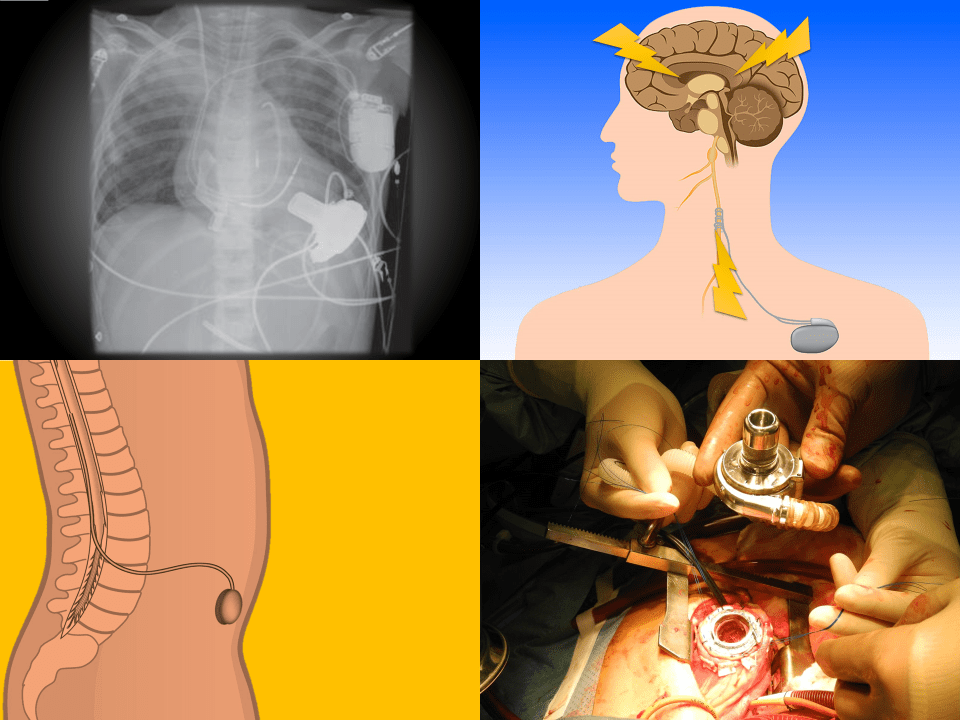
Vagus Nerve Stimulators
For intractable epilepsy; sends retrograde signal up corona radiata
Also may be used in: depression, bulimia, Alzheimer, narcolepsy, addiction, and others
VNS magnets
Are VNS safe in MRI?
Are VNS safe in everyday life?
Intrathecal Pumps
Used to infuse basal rate of drug, usually baclofen for spasticity, but pump may contain morphine, bupivicaine, clonidine. Also used for severe MS, stroke, TBI, chronic pain. Verify the medication and identify the toxidrome if symptomatic.
Ventricular Assist Devices
May be left ventricular assist, right ventricular assist, or biventricular assist device.
References
Vagus Nerve Stimulators (VNS)
Elliott RE, Rodgers SD, Bassani L et al. Vagus nerve stimulation for children with treatment-resistant epilepsy: a consecutive series of 141 cases. J Neurosurg Pediatrics. 2011; 7:491-500.
Groves DA, Brown VJ. Vagal nerve stimulation: a review of its applications and potential mechanisms that mediate its clinical effects. Neuroscience and Biobehavioral Reviews. 2005; 29: 493–500.
Panebianco M, Rigby A,Weston J,Marson AG. Vagus nerve stimulation for partial seizures. Cochrane Database of Systematic Reviews. 2015; 4, Art. No.: CD002896.
Ruffoli R, Giorgi FS, Pizzanelli C et al. The chemical neuroanatomy of vagus nerve stimulation. Journal of Chemical Neuroanatomy; 2011; 42: 288–296.
Intrathecal Pumps
Borowski A, Littleton AG, Borkhuu B et al. Complications of Intrathecal Baclofen Pump Therapy in Pediatric Patients. J Pediatr Orthop. 2010; 30:76–81.
Ghosh D, Mainali G, Khera J, Luciano M. Complications of Intrathecal Baclofen Pumps in Children: Experience from a Tertiary Care Center. Pediatr Neurosurg. 2013; 49:138–144.
Yang TF, Wang JC, Chiu JW et al. Ultrasound-guided refilling of an intrathecal baclofen pump—a case report.Childs Nerv Syst. 2013; 29:347–349.
Yeh RN, Nypaver MM, Deegan TJ, Ayyangar R. Baclofen Toxicity in an 8-year-old with an Intrathecal Baclofen Pump. J Emerg Med. 2004; 26(4): 163–167.
Ventricular Assist Devices
Blume ED, Naftel DC, Bastardi HJ et al. for the Pediatric Heart Transplant Study Investigators. Outcomes of Children Bridged to Heart Transplantation With Ventricular Assist Devices: A Multi-Institutional Study. Circulation. 2006; 113: 2313-2319.
Colón JE, Laborde ME, Nossaman BD. Case Report: Left Ventricular Assist Device in a 12 Year Old Child as a Bridge to Heart Transplantation. Section of Congenital Cardiac Anesthesia, Ochsner Medical Center, New Orleans, Louisiana. 2012.
Fan Y, Weng YG, Huebler M et al. Predictors of In-Hospital Mortality in Children After Long-Term Ventricular Assist Device Insertion. J Amer Coll Cardiol. 2011; 58(11):1183–90
Fraser CD, Jaquiss RDB, Rosenthal DN et al. Prospective Trial of a Pediatric Ventricular Assist Device. N EnglJ Med. 2012;367:532-41.
Gazit AZ, Gandhi SK, Canter CC. Mechanical Circulatory Support of the Critically Ill Child Awaiting Heart Transplantation. Current Cardiology Reviews. 2010; 6: 46-53.
VanderPluym CJ, Fynn-Thompson F, Blume ED. Ventricular Assist Devices in Children Progress With an Orphan Device Application. Circulation. 2014;129:1530-1537.
This post and podcast are dedicated to Joe Bellezzo, MD, FACEP and Zack Shinar, MD, FACEP for bringing us all up to speed. Listen to their fantastic ED ECMO podcast here.