Today on the emDOCs cast with Brit Long, MD (@long_brit), we cover abscess.
Episode 60: Abscess
Background:
- There over 6 million annual ED visits for cellulitis and abscess in the U.S.
- Skin abscess may occur in healthy individuals with no predisposing conditions.
- Risk factors for abscess: Edema secondary to impaired lymphatic drainage or venous insufficiency, obesity, skin inflammation (eczema, radiation therapy, psoriasis), skin trauma (abrasion/laceration, ulcers, bites, intravenous drug use), immunosuppression, and pre-existing skin conditions.
Pathophysiology:
- An abscess is a collection of purulent material located within the dermis or subcutaneous space that is painful.
- Develops as a result of the body trying to fight off a bacterial infection. WBCs at collect at the site of infection, commonly at a hair follicle. The purulent material that develops is an accumulation of dead WBCs and bacteria and is surrounded by a fibrous capsule.
-
Staphylococcus aureus is the predominant microbe in abscesses, accounting for 60-75% of cases, with up to 70% of these being MRSA. Beta-hemolytic streptococcal species account for less than 5%. Patients with injection drug use-related abscesses often have polymicrobial infections, which can include oral streptococci and anaerobic species.
- MRSA can be classified as either healthcare-associated MRSA (HA-MRSA) or community-acquired MRSA (CA-MRSA).
- HA-MRSA is defined as a MRSA infection occurring greater than 48 hours after hospitalization or infection occurring within 12 months of healthcare exposure. CA-MRSA is an infection occurring with no healthcare exposure.
- MRSA possesses several unique attributes increasing its ability to cause infection.
- MRSA frequently colonizes the nares, oropharynx, rectum, groin, and axilla, increasing the risk of subsequent infection.
- MRSA also produces a biofilm on invasive devices, which can enhance microbial survival and reproduction.
- Most CA-MRSA strains possess genes for production of the Panton-Valentine leucocidin cytotoxin, which increase the microbe’s virulence.
- Risk factors for MRSA-specific abscesses include recent hospitalization, living in a care facility, recent surgery, hemodialysis, HIV infection, intravenous drug use and sharing needles, recent antibiotic use, incarceration, military employment, sharing sports equipment. However, many MRSA carriers have no risk factors.
Clinical Presentation:
- An abscess will present as a fluctuant, pyogenic focus with a surrounding rim of erythema. Spontaneous drainage of purulent material can occur as the abscess extends towards the skin surface.
- On examination, abscess are often “fluctuant” and “indurated”. Fluctuant means a boggy-like feeling and indurated means firm and hardened.
- Surrounding cellulitis may be present.
- Significant tenderness is typically present.
- Systemic symptoms including fever, chills, and lymphadenopathy are unusual.
- If present, consider sepsis, necrotizing infection, and lymphadenitis.
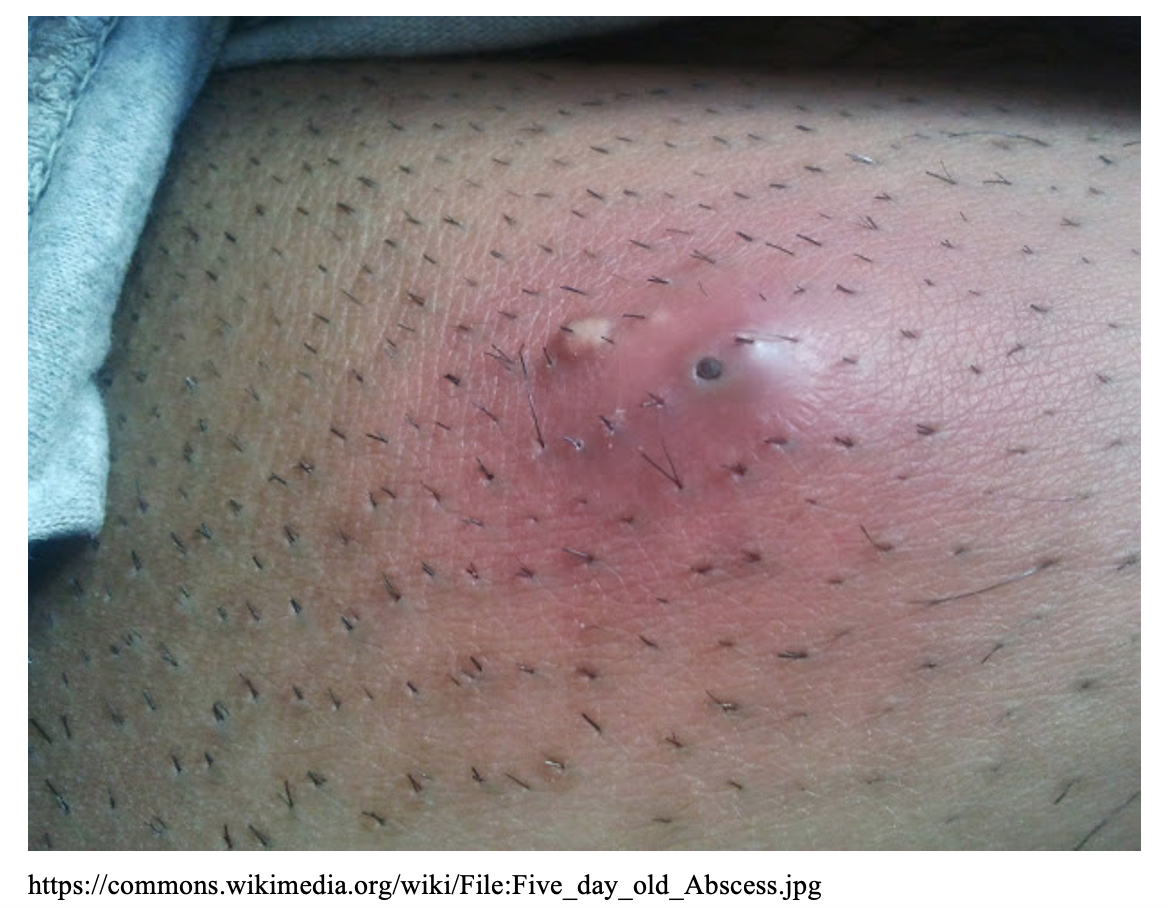
Complications:
- If left untreated, complications include bacteremia and sepsis.
- There are multiple types of abscesses aside from the commonly seen cutaneous abscess, with some having their own distinct complications and treatment protocols (only subcutaneous abscesses are discussed in this article):
- Breast abscess
- Different organ (liver, pulmonary, renal, splenic) abscesses
- Perirectal/perianal abscess
- Periodontal abscess
- Pilonidal abscess
- Bartholin’s cyst abscess
Evaluation:
- Abscess is a clinical diagnosis. Laboratory analysis is typically necessary if the patient is otherwise well appearing. If labs are obtained, they are nonspecific.
- CBC – possible leukocytosis
- ESR/CRP – possible elevation
- The IDSA does recommend obtaining a culture of the purulent fluid in patients with abscesses. However, as MRSA is the predominant cause of abscess, culture of purulent material from an abscess is unlikely to change management.
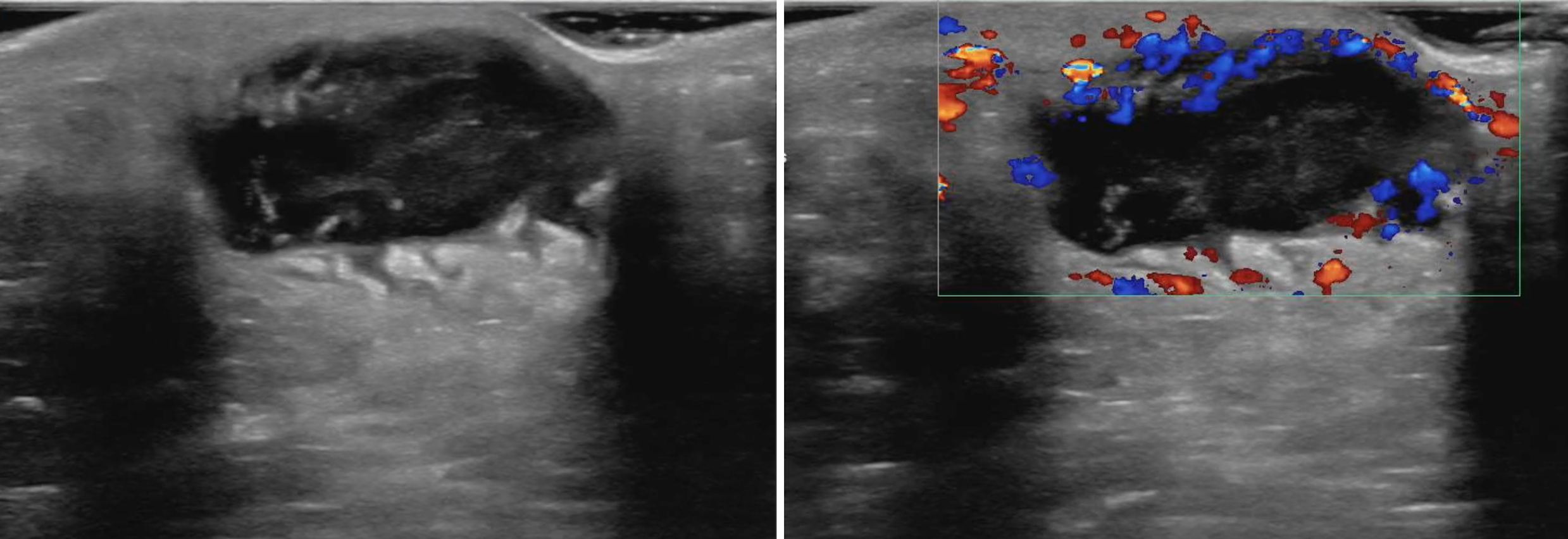
- Ultrasound (picture below) can be used as a mass with heterogenous hypoechoic debris will be seen just beneath the skin. POCUS is highly accurate, sensitive, and specific for abscess diagnosis.
- Assess for fluid collection and swirl within the collection. POCUS can identify the preferred location to perform the incision and drainage, as well as avoiding the incision of pseudoaneurysms, which will display flow on doppler imaging

- If patient has evidence of toxicity or systemic infection, obtain blood cultures, lactate, CBC, renal and liver function, and electrolytes.
Differential Diagnoses:
- Cellulitis
- Erysipelas
- Folliculitis
- Hidradenitis suppurativa
- Lymphangitis
- Necrotizing fasciitis
- Epidermoid cyst
Management:
- Patients with fluctuant masses that are concerning for abscesses should undergo an incision and drainage and potentially receive antibiotic therapy.
- Standard incision and drainage steps:
- Prepare the surface of the abscess and surrounding skin with a cleaning agent such as iodine or chlorhexidine. Using sterile towels, drape around the abscess.
- Use lidocaine to perform a field block with a 25-30g needle by injecting the lidocaine in a ring around the abscess approximately 1 cm peripheral to the erythematous border. Do not inject local anesthetics directly into the abscess to avoid infiltration of the abscess down into the tissue or up toward you. Abscesses are acidic which causes lidocaine to lose effectiveness; IV hydromorphone hydrochloride or morphine sulfate may be utilized in addition. Wait approximately 5 minutes for the lidocaine to take effect.
- Before incising, make sure to use PPE to avoid self-contamination. Make a linear incision with a number 11 or 15 scalpel deep enough to penetrate the abscess. Cut the entire length of the abscess, as not making a wide enough incision could lead to inadequate drainage of the abscess.
- Once the abscess cavity has been reached, purulent material will begin to drain. Have 4×4 gauze on hand. Allow the purulent material to drain from the abscess. Gently probe the abscess with curved hemostats to break up loculations.
- Despite irrigation of the cavity commonly be referred to as standard practice, literature has shown that irrigation does not improve (or worsen) treatment success.
- Packing is controversial. It does not reduce treatment failure but may increase pain scores. Packing may be helpful for patients with abscess over 5 cm. If packing material is warranted, insert the packing in with hemostats or forceps. Dress the wound with sterile gauze and tape.
- The patient should be told to keep the wound clean, dry, and covered. Packing should be removed every one to two days and repacked until the cavity is resolved.
- Loop drainage technique: newly utilized incision and drainage that has been found to be equally as effective in adults, superior in children, and less painful.
- Make two small (4-5mm incisions) at the periphery of the abscess.
- Use a hemostat to break up the loculations, allowing the pus to drain.
- Insert a vessel loop and pull through the other incision, tie ends together allowing for slack to avoid skin tension.
- Remove loop drain when the drainage has stopped, usually 7-10 days.


- While antibiotics are commonly prescribed for cellulitis, their role in abscess management is less clear. A systematic review and meta-analysis of 2,406 patients found that adjunctive antibiotics reduced treatment failure by half (16.1% vs 7.7%), with a statistically significant reduction in new lesion formation (15.3% vs 6.2%).
- Other studies found patients who received TMP-SMX had higher cure rates after treatment compared to placebo (80.5-83 versus 69-73.6%).
- Any benefit with antibiotics must be weighed against the risk of adverse events and the potential for increasing antimicrobial resistance.
- Antibiotic therapy is warranted if:
- Single abscess >2cm
- Multiple abscesses
- Surrounding or overlying cellulitis
- Immunosuppressed patient or patient with comorbidities
- Systemic symptoms
- Indwelling medical device is present
- High risk for transmission of staph aureus to others
- Antibiotic choices include (mg/kg is for pediatric dosing):
- TMP/SMX DS 1-2 tablets (5mg/kg) twice daily x7 days
- Clindamycin 450mg (10mg/kg) PO three times daily x7 days
- Doxycyline 100 mg PO twice daily
- Inpatient
- Parenteral therapy should be utilized if unable to tolerate PO antibiotics, rapid progression of erythema, severe infection, proximity of the lesion to an indwelling medical device.
- Antibiotics
- Preferred
- Vancomycin 20mg/kg IV twice daily8
- Alternate
- Daptomycin 4mg/kg IV once daily13
- Less commonly used
- Linezolid 600mg IV twice daily
- Tedizolid 200mg IV once daily
- Delafloxacin 300mg IV twice daily
- Omadacycline 100mg IV once daily
- Ceftaroline 600mg IV twice daily
- Dalbavancin 1500mg once
- Oritavancin 1200mg IV once
- Telavancin 10mg/kg once daily13
- Preferred
Disposition:
- Discharge unless significantly ill patients or the abscess requires surgical intervention.
- If discharged, follow up within 2-3 days for wound check.
Key Points:
- S. aureus is the most common cause of abscess.
- Abscess, like cellulitis, is a clinical diagnosis. Labs and imaging are not usually needed in evaluating or managing an abscess.
- POCUS can assist with abscess identification and drainage.
- Incision and drainage is the treatment of choice. Some patients should be treated with antibiotics.
- Admit the patient for parenteral therapy when not tolerating PO antibiotics, rapid progression of erythema, severe infection, and/or proximity of the lesion to an indwelling medical device.
Further Reading
Further FOAMed Reading:
- http://www.emdocs.net/r-e-b-e-l-em-abscess-management-the-reformation-of-an-antibiotic-nihilist/
- http://www.emdocs.net/update-on-the-management-of-skin-abscesses-in-the-emergency-department/
- https://emergencymedicinecases.com/skin-soft-tissue-infections/
References:
- Long B, Gottlieb M. Diagnosis and Management of Cellulitis and Abscess in the Emergency Department Setting: An Evidence-Based Review. J Emerg Med. 2022 Jan;62(1):16-27.
- Quirke M, Ayoub F, McCabe A, Boland F, Smith B, O’Sullivan R, Wakai A. Risk factors for nonpurulent leg cellulitis: a systematic review and meta-analysis. Br J Dermatol. 2017 Aug;177(2):382-394. DOI: 10.1111/bjd.15186. Epub 2017 Jul 25. PMID: 27864837.
- Frazee B, Lynn J, Charlebois E, Lambert L, Lowery D, Perdreau-Remington F. High Prevalence of Methicillin-Resistant Staphylococcus aureus in Emergency Department Skin and Soft Tissue Infections. Academic Emergency Medicine. 2004;11(5):552-552. DOI: 10.1197/j.aem.2004.02.164.
- Stevens D, Bisno A, Chambers H, Dellinger E, Goldstein E, Gorbach S, Hirschmann J, Kaplan S, Montoya J, Wade J. Practice guidelines for the diagnosis and management of skin and soft tissue infections: 2014 update by the infectious diseases society of America. Clinical Infectious Diseases. 2014;59(2). DOI:10.1093/cid/ciu296.
- Summanen P, Talan D, Strong C, McTeague M, Bennion R, Thompson Jr J, Vaisanen M, Moran G, Winer M, Finegold S. Bacteriology of skin and soft-tissue infections: comparison of infections in intravenous drug users and individuals with no history of intravenous drug use. Clinical Infectious Diseases. 1995;20. DOI: 10.1093/clinids/20.supplement_2.s279
- Raff AB, Kroshinsky D. Cellulitis: A Review. JAMA. 2016 Jul 19;316(3):325-37. doi: 10.1001/jama.2016.8825. PMID: 27434444.
- Chinnock B, Hendey G. Irrigation of Cutaneous Abscesses Does Not Improve Treatment. Annals of Emergency Medicine. 2015; 67 (3). DOI: 1016/j.annemergmed.2015.08.007.
- Tsoraides S, Pearl R, Stanfill A, Lizabeth W, Vegunta R. Incision and loop drainage: a minimally invasive technique for subcutaneous abscess management in children. Journal of Pediatric Surgery. 2010; 45(3). DOI: 1016/j.jpedsurg.2009.06.013
- Levine, Brian J. EMRA Antibiotic Guide. EMRA, 2020.
- Talan D, Moran G, Krishnadasan A, Abrahamian F, Lovecchio F, Karras D, Steele M, Rothman R, Mower W. Subgroup Analysis of Antibiotic Treatment for Skin Abscesses. Annals of Emergency Medicine. 2018; 71(1):21-30. DOI: 10.1016/j.annemergmed.2017.07.483.
- Daum R, et al. A Placebo-Controlled Trial of Antibiotics for Smaller Skin Abscesses. New England Journal of Medicine. 2017; 376 (26). PMID: 28657870
- O’Malley G, et al. Routine packing of simple cutaneous abscesses is painful and probably unnecessary. Academic Emergency Medicine. 2009; 16(5). PMID: 19388915
- Kessler D, Krantz A, Mojica M. Randomized trial comparing wound packing to no wound packing following incision and drainage of superficial skin abscesses in the pediatric emergency department. Pediatric Emergency Care Journal. 2012; 28(6). PMID: 22653459
- Rybak M, Le J, Lodise T, et al. Therapeutic Monitoring of Vancomycin for Serious Methicillin-Resistant Staphylococcus Aureus Infections: A Revised Consensus Guideline and Review by the American Society of Health-System Pharmacists, the Infectious Diseases Society of America, the Pediatric Infectious Diseases Society, and the Society of Infectious Diseases Pharmacists. American Journal of Health- System Pharmacy. 2020; 77(835).
- Gottlieb M, DeMott JM, Hallock M, Peksa GD. Systemic Antibiotics for the Treatment of Skin and Soft Tissue Abscesses: A Systematic Review and Meta-Analysis. Ann Emerg Med. 2019 Jan;73(1):8-16.





