Authors: Brit Long, MD (@long_brit, EM Attending Physician at SAUSHEC, USAF) and Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW Medical Center / Parkland Memorial Hospital) // Edited by: Jamie Santistevan, MD (@Jamie_Rae_EMdoc, Admin and Quality Fellow at UW, Madison, WI)
A 42-year-old female presents with pleuritic chest pain and dyspnea with exertion. Her VS include HR 102, RR 21, O2 sat 98% on RA, and T 98. Her ECG and chest X-ray are normal, as well as laboratory studies including troponin and BNP. You obtain a CTA chest, which demonstrates a right segmental PE. Does this patient require admission? What anticoagulation options do you have?
Pulmonary embolism (PE) is a common disease, with an incidence approaching 56 per 100,000 patients. This increases with age with 500 per 100,000 in patients over 80 years.1-6 Over 100,000 deaths occur annually in the U.S from PE.4-6 Diagnosis of PE has increased with improved technology; however, the mortality of PE has remained similar despite increased diagnosis.7-9
Testing
Increased testing, including D-dimer and CTA, has resulted in more PEs diagnosed.7-9 Instead of reducing morbidity and mortality, physicians may actually be increasing patient risk in an attempt to diagnose PE.11,12 One study demonstrates that PE testing prevents 6 deaths and 24 major PE-related events, while causing 36 deaths and 37 PE-related harms such as renal failure due to contrast, major hemorrhage, and cancer due to radiation from CT.13-15
Strategies have been suggested to decrease this potential patient harm. Risk scores can be used to assist providers in evaluation, in association with D-dimer and imaging.10-12 Another option is the use of risk stratification to determine which patients are appropriate for discharge home with treatment, as opposed to inpatient admission and treatment.10-12
Inpatient Versus Outpatient Management History
Patients with VTE have historically been admitted for treatment and monitoring. Over 90% of EDs admit patients with PE in the U.S., with therapy including heparin and warfarin.10,16-19 The advent of low molecular weight heparins (LMWHs) and fondaparinux made home treatment possible, specifically for DVT.20-22 These medications are safe and efficacious, while not requiring regular monitoring. These attributes make home treatment feasible.
Close to one third of these patients with DVT have an associated PE.23 In Canada, studies in the early 2000’s demonstrated the safety of outpatient treatment for PE, with 50% of patients safely treated at home.20,24,25 In the U.S. this is not common, as well as other parts of the world.26-28 Even in the era of novel oral anticoagulants, over 98% of patients with PE are admitted for inpatient treatment.29 Despite this trend in the U.S., studies suggest close to 50% of patients are appropriate for outpatient management, specifically patients categorized as low risk for adverse outcome.10,30 The recently updated American College of Chest Physicians 2016 guidelines provide a Grade 2B recommendation for outpatient management for patients with low-risk PE.10
Outpatient Treatment Barriers
The majority of centers in the U.S. admit patients with PE. One issue is the uncertainty in identifying patients at low risk for adverse outcome appropriate for discharge.29,31 Many are not comfortable with the use of criteria for outpatient therapy.32-37
Outpatient Treatment Benefits
Several benefits exist for providing outpatient care of PE. Potential improvements in quality of life, social function, and physical activity are possible with outpatient care.20,29,31,36 Outpatient therapy is associated with decreased length of stay and reduction in overall cost. Estimates demonstrate a potential savings of $7 million per year.37,38 Not only can outpatient treatment reduce cost, but it is safe with proper risk stratification.
Controversy
The controversy surrounding outpatient PE therapy centers on three questions:
- Is outpatient treatment for PE inferior to inpatient treatment?
- Is the risk of harm greater with outpatient versus inpatient therapy?
- Finally, what tools are present for patient risk stratification?
Outpatient versus Inpatient Therapy
The literature suggests outpatient therapy is not inferior to inpatient therapy. A study in 2011 finds that of 344 patients with acute PE, one patient in the outpatient group versus no patients in the inpatient group experienced recurrent VTE, statistically noninferior.39 One patient out of 173 outpatients in this study developed recurrent VTE, with one death in the inpatient group and outpatient group.39 A study by Fang et al. from 2015 conducted an evaluation of low risk PE patients based on the PE Severity Index (PESI), with 494 of 5927 patients treated as outpatient. Investigators find no deaths within 30 days, with two deaths at 90 days with outpatient therapy.40 However, a Cochrane review suggests the current literature is not sufficient to assess efficacy and safety of outpatient versus inpatient therapy for PE due to small sample sizes.41 Though a higher evidence level would be helpful, current literature suggests outpatient management is safe, feasible, and efficacious for a significant percentage of patients with acute PE.
Outpatient Therapy Outcomes
Literature does suggest safety with outpatient treatment. Two studies demonstrate that patients with non-massive PE, hemodynamic stability, and no oxygen requirement treated with LMWH and warfarin have extremely low risk of adverse outcome, with no patients dying of PE at 3 months, and 13 of nearly 600 patients experiencing recurrent VTE.25,42 A study by Kovacs et al. evaluated 639 patients, of which 314 were low risk and managed as outpatient. Less than 1% of patients experience hemorrhage (3 patients) or recurrent VTE (3 patients) in this cohort.25 Erkens et al. investigated 473 patients, with 55% treated as outpatient.42 No deaths due to PE occur in this cohort, with 0.4% recurrent VTE rate in the outpatient group within two weeks. No bleeding occurred at two weeks.42
The American College of Chest Physicians (ACCP) and European Society of Cardiology (ESC) indicate that risk tools may be used to identify patients at low risk for adverse event and early mortality.10,34 Patients at low risk may be discharged home for treatment.
Zondag et al. finds recurrent VTE, major bleeding, and all-cause mortality to not be significantly different between acute PE patients treated as outpatient versus inpatient.43 Aujesky et al. finds that treatment of acute PE patients at low risk using the PESI score, defined by classes 1 or 2, demonstrates efficacy and safety.39 This study did exclude patients with active bleeding, high risk for bleeding, poor social situation, renal failure, hypoxemia, pregnancy, and extreme obesity. Per these results, VTE, bleeding, and overall mortality rates are noninferior at 14 and 90 days at follow-up.39 Vinson et al. conducted a systematic review of studies evaluating patients with acute PE and PESI 1 or 2 treated initially with enoxaparin and oral warfarin.44 Those deemed appropriate for outpatient treatment do not demonstrate significant adverse event rate difference with the admitted group.44
A recent study released in 2015 evaluated the use of rivaroxaban 15 mg by mouth twice daily for 21 days, followed by 20 mg once per day. Investigators used modified Hestia Exclusion Criteria, discussed later, to identify patients at low risk for adverse outcome.45 Results suggest this option is safe. In this cohort of 106 patients discharged with VTE, 28% have PE%, 67% have DVT, and 5% have combined DVT and PE. No patients experience new VTE, while three patients experience recurrent VTE after treatment discontinuation.45
Risk Stratification and Low-Risk Patients
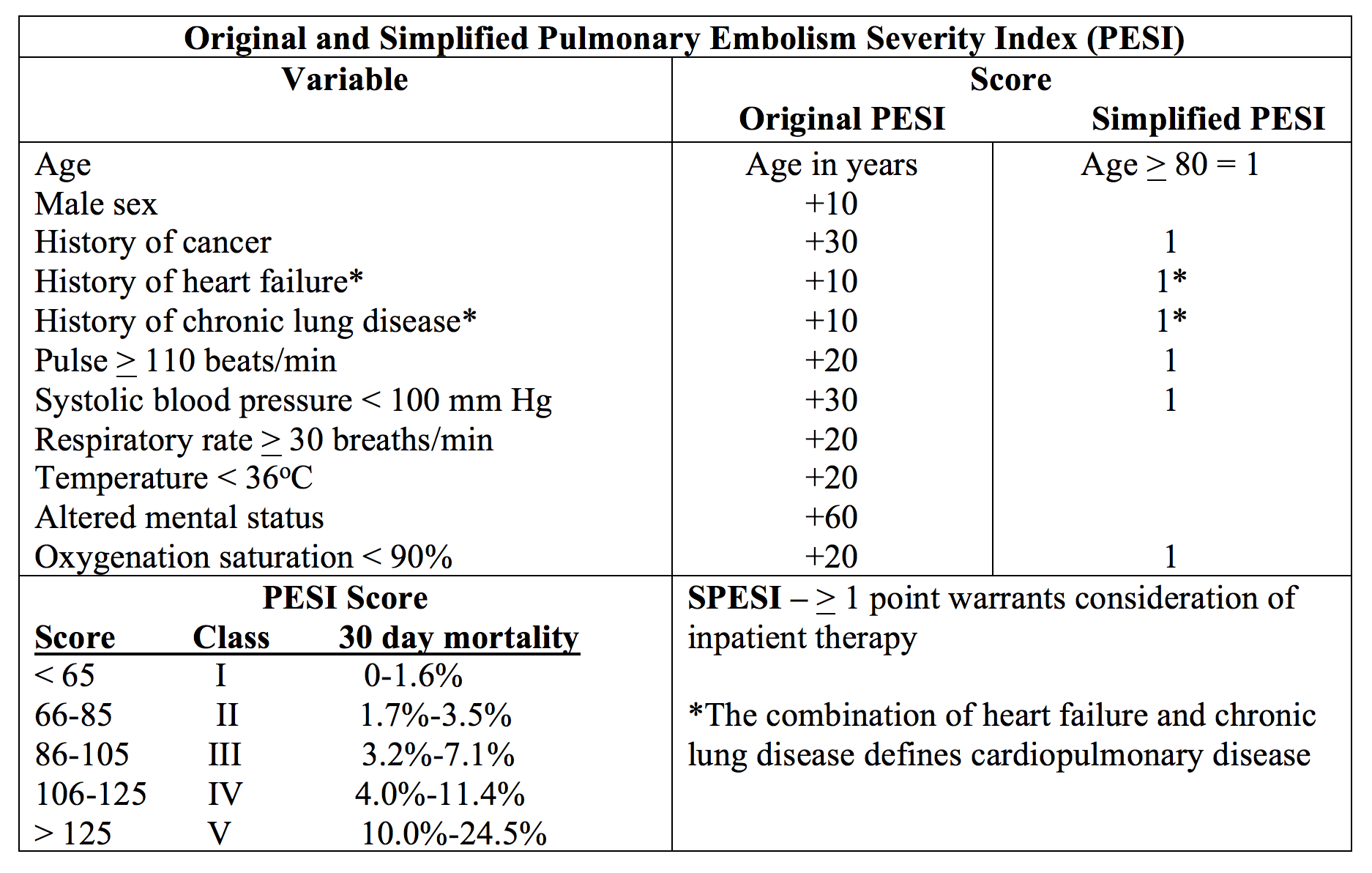
Studies demonstrate outpatient PE treatment is feasible and safe for patients at low risk for PE mortality. ACCP guidelines recommend outpatient treatment for low-risk patients with adequate social situation.10 However, there is no consensus on which rule/score to use. Investigators have sought a rule or score that can identify patients at low risk for PE-related mortality. These include the Pulmonary Embolism Severity Index (PESI), original and simplified versions, Geneva Prognostic Score (GPS), Global Registry of Acute Coronary Events (GRACE), Hestia Criteria, and Aujesky score.
PESI was originally developed to estimate mortality at 30 days in patients with acute PE utilizing eleven factors.46,47 Studies have evaluated PESI to identify patients with PE who are appropriate for discharge if low risk. Sensitivity approaches 89%, with specificity 49%, positive predictive value (PPV) 11%, and negative predictive value (NPV) 98%.46-49 Of patients with suspected PE, approximately 45% of patients meet low risk criteria.46-49
Simplified PESI uses 7 factors from the original PESI. Any item positive in the index places the patient at higher risk for adverse event.50,51 The simplified index possesses sensitivity 96.1%, specificity 38%, PPV 11%, and NPV 99%. The simplified PESI demonstrates similar prognostic accuracy, as well as similar NPV and PPV.50,51 However, the simplified PESI is easier to use and does not require calculation, which reduces complexity, as well as placing 35% of patients at low-risk. Over 25,000 patients have undergone analysis with this rule.50,51 A post-hoc analysis of the EINSTEIN PE study released in 2015 finds that patients with sPESI of < 1 treated with rivaroxaban as an outpatient to have low incidence of major adverse events within 30 days. Scores of 0 demonstrate a recurrent VTE rate of 0.8%, while scores of 1 have a recurrent VTE rate of 1.0%.52 All-cause mortality is also low. Patients with scores > 2 possess greater rates of recurrent VTE (4.3%), all-cause mortality (10.2%), PE-related mortality (1.7%), and major bleeding (4.0%).50-52
The Hestia Criteria utilizes eleven clinical markers, shown below. Any positive criteria warrants consideration of treatment as inpatient. If none are present, the patient may be treated as an outpatient. A study by Zondag et al. including 496 patients finds a sensitivity of 82% (95% CI 0.52-0.95) and specificity of 56% (95% CI 0.52-0.61).43 A three month follow up period reveals approximately 1% mortality rate, though none from PE. Recurrent VTE occurred in 2.0% of the outpatient treatment group. Major bleeding occurs in less than 1% of patients.43,45 Close to 55% of patients are categorized as low risk based on these criteria.43,45 A 2015 study by Beam et al. utilized the Hestia Criteria to risk stratify patients and finds those at low risk to have no recurrent VTE or major bleeding while on anticoagulation.43,45 The Hestia Criteria can be quickly used at the bedside to risk stratify patients.

The Geneva Prognostic Score (GPS) is comprised of six variables including clinical, laboratory, and ultrasound findings.53 Low risk patients demonstrate a rate of adverse outcomes of 2.2%-5%, with sensitivity ranging from 40% to 85% and NPV of 95% to 98%.53 Studies demonstrate that use of this score can place 76% to 88% of patients as low risk.53,54 Validations of this score have not yielded similar results, with high mortality and lower sensitivity.54

The European Society of Cardiology (ESC) guidelines on the diagnosis and management of acute PE stratify patients into several levels of risk for death.34 Per ESC guidelines, this stratification scheme should only be used in patients with suspected PE. Low risk is defined by negative RV strain on biomarkers and imaging. Sensitivity approaches 88%, with 23% to 36% of patients meeting low-risk criteria.34,54

The Global Registry of Acute Coronary Events (GRACE) has a high diagnostic ability for adverse outcomes in acute coronary syndrome (ACS); however, investigators have sought to use this score for PE risk stratification.55 The components of this score can be complex (http://www.mdcalc.com/grace-acs-risk-and-mortality-calculator/). Sensitivities approach 99%, with specificities of 27%. Only 22% of patients meet low risk criteria based on this score.54,55 This score can be difficult to use in the ED, and evidence support is low.
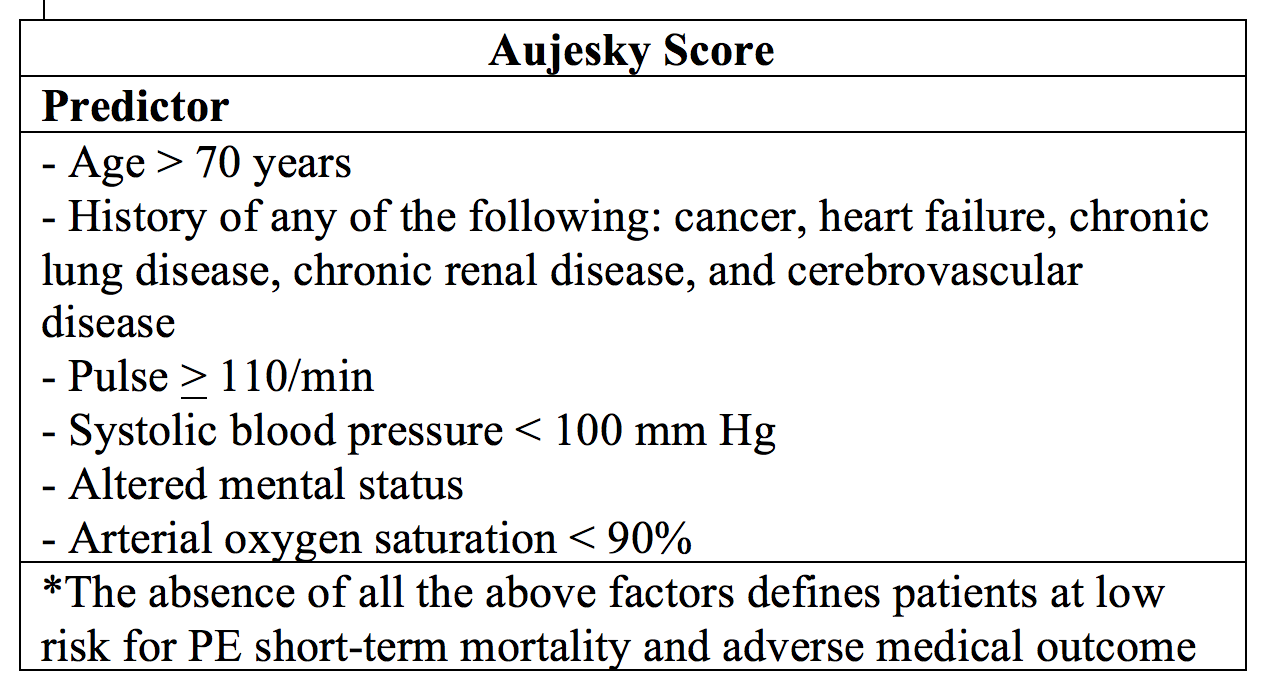
The Aujesky score, first published in 2006, takes into account factors similar to PESI and sPESI.46 These include 10 patient factors and clinical variables. Use of this rule shows 30-day mortality rates for low risk patients 0.6%, 1.5%, and 0% in the derivation, internal validation, and external validation studies, respectively.46,47,48,58 Sensitivities approach 99%; however, upon pooled analysis, only 22% of patients meet low risk criteria.
How do the scores compare?
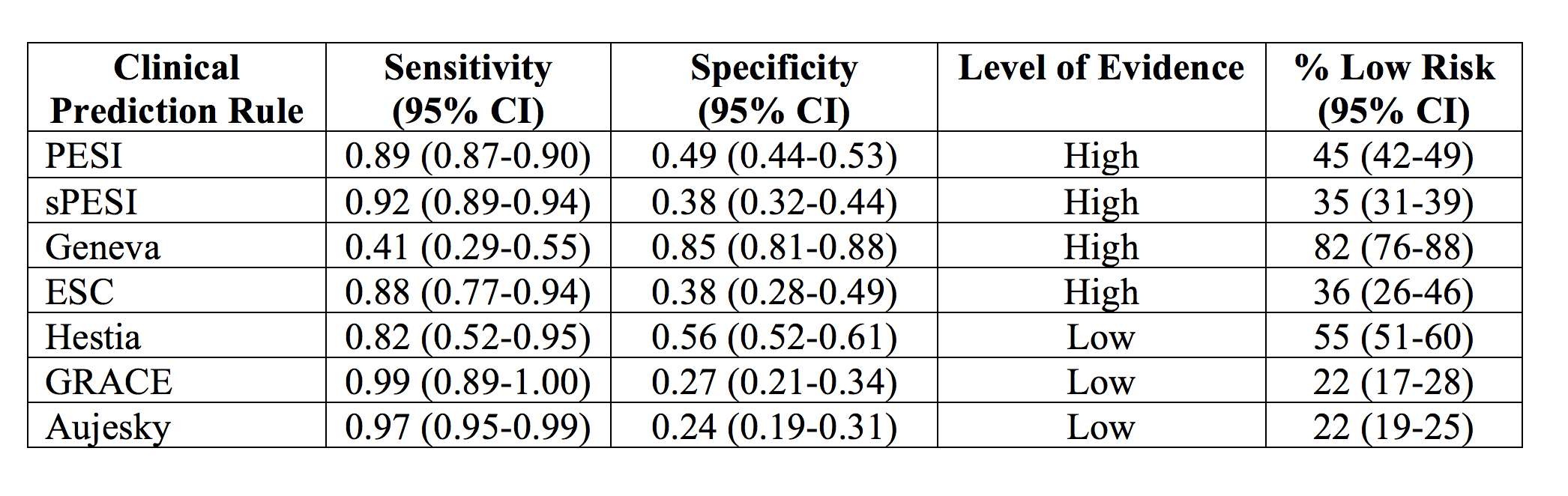
A 2015 meta-analysis identifies several clinical prediction rules with sensitivities near 90% including sPESI, PESI, and European Society of Cardiology (ESC).54 PESI, sPESI, ESC, and Geneva rules demonstrate high quality of evidence, but the scores with lowest study bias include PESI, sPESI, and Geneva. This meta-analysis does not recommend use of the Geneva score due to its low sensitivity of approximately 40%, and the PESI tool is difficult to score due to use of 11 variables. However, sPESI and ESC rules display potential qualities for use in PE risk stratification. These rules also identify approximately 45% as low risk of early all-cause mortality, but specificities only approach 50%.54
Combining Prediction Rules with Biomarkers and Imaging
Patients with acute PE are often assessed with multiple biomarkers including BNP and troponin, and elevations in these markers are associated with increased risk of adverse event. NT-proBNP elevation above 600 pg/mL and right ventricular dysfunction on echocardiogram assist risk stratification with PESI.57 Jimenez et al. finds combining sPESI with negative BNP possesses a NPV for adverse event of over 99%.58 A study by Singanayagam et al. suggests the combination of troponin and PESI to improve the predictive value of 30 day mortality.59 Sanchez et al. demonstrates the addition of RV dysfunction on US in association with PESI can predict adverse outcome.60 Per the ESC, any biomarker elevation or findings of RV dysfunction on imaging places the patient in the intermediate risk category, unsuitable for outpatient therapy per the ESC.34 However, other studies demonstrate the addition of biomarkers to clinical scores may not improve risk stratification. Moores et al. in 2013 finds that negative troponin I with low-risk PESI does not improve NPV ability.61 Zondag et al. in evaluation of the Hestia criteria, which does not take into account biomarkers or imaging, finds it to have greater predictive value of adverse clinical outcome when compared to ESC criteria (the ESC criteria does use biomarkers and echocardiogram).43 Literature may conflict, but risk stratification in association with negative biomarkers may place the patient at low risk for adverse event.
Novel Oral Anticoagulants (NOAC)
The novel, target-specific non-vitamin K antagonists (VKA) oral anticoagulants have improved outpatient therapy.10,62 ACCP guidelines support the use of these agents for three months.10 The EINSTEIN PE Trial randomized patients to rivaroxaban 15 mg two times daily for three weeks followed by 20 mg once per day, versus standard therapy with enoxaparin followed by adjusted dose VKA therapy.63 The rivaroxaban group demonstrates a 2.1% rate of recurrent VTE, versus standard therapy 1.8%. Per study results, major bleeding occurs in 1.1% of the rivaroxaban group and 2.2% of the standard therapy group. The results suggest fixed-dose rivaroxaban is non-inferior to standard therapy with enoxaparin and oral VKA.63 The AMPLIFY study finds apixaban to be noninferior to standard therapy, with lower rates of bleeding in the apixaban group, 4.7% versus 9.7%.64 All-cause mortality and recurrent VTE are similar based on this study.64 A systematic review and meta-analysis finds similar rates of recurrent VTE, death, and major bleeding between warfarin and NOAC treatment.65
The Factor Xa antagonists, rivaroxaban and apixaban, do not require initial parenteral anticoagulation, but edoxaban does.62,66-69 The antithrombin inhibitor dabigatran also requires initial parenteral anticoagulation.67 These medications do not require monitoring, but they have specific dosing routines. Dosing for dabigatran is 150 mg twice daily by mouth, rivaroxaban 15 mg two times daily for 21 days then 20 mg once per day with food, and apixaban 10 mg twice daily for 7 days followed by 5 mg twice daily.66-69 These NOACs do not require routine monitoring. Caution is warranted in patients older than 70 years and those with renal or hepatic disease. These agents are not approved for patients with massive PE or DVT, pregnancy, morbid obesity, active cancer, and serious thrombophilic defect.62,66-69
Bleeding Risk
Balancing risk of recurrent VTE and hemorrhage is necessary for outpatient therapy.62,65,70 Recurrent thrombus rate at one year can reach 27%, but the rate of fatal, major, and minor bleeding during therapy with warfarin is 0.6%, 3.0%, and 9.6%, respectively.71 One of the most feared complications is intracerebral hemorrhage, which is increased 7 to 10 fold with anticoagulation.72
The HAS-BLED score (Hypertension, Abnormal renal/liver function, Stroke, Bleeding history or predisposition, Labile INR, Elderly (> 65 years), Drugs/alcohol concomitantly) was developed in 2010 to assess one year bleeding risk in patients with atrial fibrillation.70 This score has not evaluated risk of bleeding in patients with PE. Patients with < 1 point display a 3.4% risk of major bleeding in one validation study, while patients with scores > 3 points demonstrate a 5.8% risk of bleeding.65,73 This population warrants consideration of other therapies due to bleeding risk. This score possesses greater abilities to predict risk of major hemorrhage when compared to other scores.73-75 This score has been evaluated in patients with atrial fibrillation and not PE, but it does provide an easy means of evaluating bleeding risk.
Another tool for assessing hemorrhage risk for VTE outpatients with warfarin is the Outpatient Bleeding Risk Index (patient > 65 years, history of CVA, history of GI bleeding, recent MI, anemia, Cr > 1.5, or Diabetes).76-78 Major bleeding rates in the low, moderate, and high risk groups are 3%, 12%, and 48%, respectively, in the derivation set and 3%, 8%, and 30%, respectively, in the validation set.77,78
What should the emergency provider do?
Home treatment is feasible with VTE therapies that do not require hospitalization and scoring systems for risk stratification. Risk factors for adverse outcomes, patient ability to comply with treatment and successfully complete therapy, risk of major hemorrhage, and inpatient preferences with shared decision making should be considered.10-12,34,54
Patients with acute PE should undergo risk stratification first, and patients with any hemodynamic instability should be admitted.10,34,54 If the patient is hemodynamically stable, clinical scores can be used, with negative biomarkers. Elevation of these markers places the patient at intermediate-risk for adverse event. Multiple scores are present, and sPESI and ESC are easy to use with high sensitivity. PESI, GRS, and GRACE are difficult to use in the ED. The Hestia Criteria and Aujesky 2006 rule can be used, but they do not possess as much high quality evidence as other criteria. Class 1 and 2 PESI, negative Hestia, negative ESC, or negative sPESI patients are at low risk for PE-related all-cause mortality.10,34,54 If the patient has an alternative reason for admission, patient compliance is questionable, or psychosocial barriers are present, the patient warrants admission for treatment. Assessment for bleeding risk with HAS-BLED or Outpatient Bleeding Risk Index is recommended if considering discharge.70,76 Patients with HAS-BLED score < 1 or Outpatient Bleeding Risk Index score 0 have low risk of bleeding.70,76 If the patient desires outpatient therapy and is stable with adequate social situation, outpatient therapy is a feasible, safe option.
Case Conclusion:
The patient is low risk for adverse events based on sPESI, with negative biomarkers. The patient desires discharge home with treatment, and she appears to have an adequate social situation. You speak with her primary care physician over the phone, who is comfortable with the plan and will see her in several days. You provide a prescription for rivaroxaban and discharge the patient home.
Summary
-Patients with confirmed PE are classically admitted for treatment and monitoring of anticoagulation, with over 90% managed in-hospital.
-Literature suggests outpatient treatment is non-inferior, particularly with novel oral anticoagulants. Recurrent thromboembolism, risk of bleeding, and incidence of major adverse outcome are similar in patients treated as outpatient versus inpatient.
–sPESI, PESI, and ESC possess strong literature support for outpatient PE stratification, with adequate sensitivity for predicting low risk adverse event.
-The HAS-BLED score and Outpatient Bleeding Risk Index allow assessment of bleeding risk.
–Patient compliance, presence of psychosocial barriers, or alternative need for admission should be considered in patient disposition. Consideration of these important aspects with risk stratification and use of NOAC therapy can allow for safe, efficacious treatment as outpatient.
References/Further Reading
- Heit JA. The epidemiology of venous thromboembolism in the community: implications for prevention and management. J Thromb Thrombolysis 2006 Feb;21(1):23-9.
- Horlander KT, Mannino DM, Leeper KV. Pulmonary embolism mortality in the United States, 1979-1998: an analysis using multiple-cause mortality data. Arch Intern Med 2003; 163:1711.
- Silverstein MD, Heit JA, Mohr DN, et al. Trends in the incidence of deep vein thrombosis and pulmonary embolism: a 25-year population-based study. Arch Intern Med 1998; 158:585.
- Naess IA, Christiansen SC, Romundstad P, et al. Incidence and mortality of venous thrombosis: a population-based study. J Thromb Haemost 2007; 5:692.
- Tagalakis V, Patenaude V, Kahn SR, Suissa S. Incidence of and mortality from venous thromboembolism in a real-world population: the Q-VTE Study Cohort. Am J Med 2013; 126:832.e13.
- Martinez C, Cohen AT, Bamber L, Rietbrock S. Epidemiology of first and recurrent venous thromboembolism: a population-based cohort study in patients without active cancer. Thromb Haemost 2014; 112:255.
- Hoffman JR, Cooper RJ. Overdiagnosis of disease: a modern epidemic. Arch Intern Med 2012 Aug 13;172(15):1123-4.
- Moynihan R, Doust J, Henry D. Preventing overdiagnosis: how to stop harming the healthy. BMJ 2012 May 28;344:e3502.
- Burge AJ, Freeman KD, Klapper PJ, Haramati LB. Increased diagnosis of pulmonary embolism without a corresponding decline in mortality during the CT era. Clin Radiol 2008 Apr;63(4):381-6.
- Keaton C, Akl EA, Ornelas J, Balizas A, et alt. Antithrombotic Therapy for VTE disease: CHEST Guideline. Chest 2016. DOI: 10.1016/j.chest.2015.11.026.
- Kline JA and Kabrhel C. Emergency Evaluation for Pulmonary Embolism, Part 1: Clinical Factors that Increase Risk. JEM 2015;48(6):771 – 780.
- Kline JA and Kabrhel C. Emergency Evaluation for Pulmonary Embolism, Part 2: Diagnostic Approach. JEM 2015;49(1):104-117.
- Newman DH, Schriger DL. Rethinking testing for pulmonary embolism: less is more. Ann Emerg Med 2011 Jun;57(6):622-627.
- Park B, Messina L, Dargon P, Huang W, Ciocca R, Anderson FA. Recent trends in clinical outcomes and resource utilization for pulmonary embolism in the United States: findings from the nationwide inpatient sample. Chest 2009 Oct;136(4):983-90.
- Bullano MF, Willey V, Hauch O, Wygant G, Spyropoulos AC, Hoffman L. Longitudinal evaluation of health plan cost per venous thromboembolism or bleed event in patients with a prior venous thromboembolism event during hospitalization. Manag Care Pharm 2005 Oct;11(8):663-73.
- Barritt DW, Jordan SC. Anticoagulant drugs in the treatment of pulmonary embolism: a controlled trial. Lancet 1960;1:1309-12.
- Simonneau G, Sors H, Charbonnier B, et al. A comparison of low-molecular- weight heparin with unfractionated heparin for acute pulmonary embolism. N Engl J Med 1997;337:663-9.
- The Matisse Investigators. Subcutaneous fondaparinux versus intravenous un- fractionated heparin in the initial treatment of pulmonary embolism. N Engl J Med 2003;349:1695-702.
- The van Gogh Investigators. Idraparinux versus standard therapy for venous thromboembolic disease. N Engl J Med 2007;357:1094-104.
- Koopman MM, Prandoni P, Piovella F, Ockelford PA, Brandjes DP, van der MJ, Gallus AS, Simonneau G, Chesterman CH, Prins MH. Treatment of venous thrombosis with intravenous unfractionated heparin administered in the hospital as compared with subcutaneous low-molecular-weight heparin administered at home. The Tasman Study Group. N Engl J Med 1996; 334: 682–7.
- Levine M, Gent M, Hirsch J, Leclerc J, Anderson D, Weitz J, Ginsberg J, Turpie AG, Demers C, Kovacs M. A comparison of low-molecular- weight heparin administered primarily at home with unfractionated heparin administered in the hospital for proximal deep-vein thrombosis. N Engl J Med 1996; 334: 667–81.
- The Columbus Investigators. Low-molecular-weight heparin in the treatment of patients with venous thromboembolism. N Engl J Med 1997; 337: 657–62.
- Dorfman GS, Cronan JJ, Tupper TB, Messersmith RN, Denny DF, Lee CH. Occult pulmonary embolism: a common occurrence in deep venous thrombosis. AJR Am J Roentgenol 1987; 148: 263–6.
- Kovacs MJ, Anderson D, Morrow B, Gray L, Touchie D, Wells PS. Outpatient treatment of pulmonary embolism with dalteparin. Thromb Haemost 2000; 83: 209–11.
- Kovacs MJ, Hawel JD, Rekman JF, Lazo-Langner A. Ambulatory management of pulmonary embolism: a pragmatic evaluation. J Thromb Haemost 2010; 8: 2406–11.
- Ong BS, Karr MA, Chan DK, Frankel A, Shen Q. Management of pulmonary embolism in the home. Med J Aust 2005;183(5):239–42.
- Olsson CG, Bitzen U, Olsson B, Magnusson P, Carlsson MS, Jonson B, Bajc M. Outpatient tinzaparin therapy in pulmonary embolism quantified with ventilation/perfusion scintigraphy. Med Sci Monit 2006;12(2):PI9–13.
- Rhodes S, Bond S. Shifting pulmonary embolism management to primary care. Nurs Times 2006;102(6):23–4.
- Stein PD, Fatta F, Hughes PG, Hourmouzis ZN, Hourmouzis NP, White RM, et al. Home Treatment of Pulmonary Embolism in the Era of Novel Oral Anticoagulants. Am J Med 2016 Sep;129(9):974-977.
- Baglin T. Fifty per cent of patients with pulmonary embolism can be treated as outpatients. J Thromb Haemost 2010; 8: 2404–5.
- Aujesky D, Mazzolai L, Hugli O, Perrier A. Outpatient treatment of PE. Swiss Med Wkly 2009;139(47–48):685–690.
- British Thoracic Society guidelines for the management of suspected acute pulmonary embolism. Thorax 2003;58(6): 470–83.
- Snow V, Qaseem A, Barry P, Hornbake ER, Rodnick JE, et al. Management of venous thromboembolism: a clinical practice guideline from the American College of Physicians and the American Academy of Family Physicians. Ann Intern Med 2007;146(3):204–10.
- Torbicki A, Perrier A, Konstantinides S, Agnelli G, Galie N, Pruszczyk P, et al. Guidelines on the diagnosis and management of acute pulmonary embolism: the Task Force for the Diagnosis and Management of Acute Pulmonary Embolism of the European Society of Cardiology (ESC). Eur Heart J 2008;29(18):2276–315.
- Aujesky D, Stone RA, Kim S, Crick EJ, Fine MJ. Length of hospital stay and post discharge mortality in patients with pulmonary embolism: a statewide perspective. Arch Intern Med 2008;168(7):706–12.
- Harrison L, McGinnis J, Crowther M, Ginsberg J, Hirsh J. Assessment of outpatient treatment of deep-vein thrombosis with low-molecular-weight heparin. Arch Intern Med 1998;158(18):2001–3.
- Aujesky D, Smith KJ, Cornuz J, Roberts MS. Cost-effectiveness of low-molecular-weight heparin for treatment of pulmonary embolism. Chest 2005;128(3):1601–10.
- From the Swiss Federal Statistical Office website. Available at: http://www.bfs.admin.ch/bfs/portal/de/index/themen/14/04/0 1/data/01.html. Accessed April 15, 2015.
- Aujesky D, Roy PM, Verschuren F, Righini M, Osterwalder J, Egloff M, Renaud B, Verhamme P, Stone RA, Legall C, Sanchez O, Pugh NA, N’Gako A, Cornuz J, Hugli O, Beer HJ, Perrier A, Fine MJ, Yealy DM. Outpatient versus inpatient treatment for patients with acute pulmonary embolism: an international, open-label, randomised, non-inferiority trial. Lancet 2011; 378: 41–8.
- Fang MC, Fan D, Sung S, et al. Outcomes in Adults With Acute Pulmonary Embolism Who Are Discharged From Emergency Departments: The Cardiovascular Research Network Venous Thromboembolism Study. JAMA Intern Med 2015;175(6):1060-1062.
- Yoo HH, Queluz TH, El Dib R. Outpatient versus inpatient treatment for acute pulmonary embolism. Cochrane Database Syst Rev 2014; 11: CD010019.
- Erkens PMG, Gandara E, Wells P, Shen AYH, Bose G, Le Gal G, Rodger M, Prins MH, Carrier M. Safety of outpatient treatment in acute pulmonary embolism. J Thromb Haemost 2010; 8: 2412–7.
- Zondag W, Vingerhoets LM, Durian MF, et al; Hestia Study Investigators. Hestia criteria can safely select patients with pulmonary embolism for outpatient treatment irrespective of right ventricular function. J Thromb Haemost 2013; 11(4):686-692.
- Vinson DR, Zehtabchi S, Yealy DM. Can selected patients with newly diagnosed pulmonary embolism be safely treated without hospitalization? A systematic review. Ann Emerg Med 2012 Nov;60(5):651-662.
- Beam DM, Kahler ZP, Kline JA. Immediate discharge and home treatment with rivaroxaban of low risk venous thromboembolism diagnosed in two U.S. emergency departments: a one-year preplanned analysis. Acad Emerg Med 2015;22:789–795
- Aujesky D, Roy PM, Le Manach CP, et al. Validation of a model to predict adverse outcomes in patients with pulmonary embolism. Eur Heart J 2006;27 (4) 476- 481.
- Aujesky D, Perrier A, Roy PM, et al. Validation of a clinical prognostic model to identify low-risk patients with pulmonary embolism. J Intern Med 2007;261(6): 597- 604.
- Jiménez D, Yusen RD, Otero R, et al. Prognostic models for selecting patients with acute pulmonary embolism for initial outpatient therapy. Chest 2007;132 (1) 24- 30.
- Donzé J, Le Gal G, Fine MJ, et al. Prospective validation of the Pulmonary Embolism Severity Index: a clinical prognostic model for pulmonary embolism. Thromb Haemost 2008;100 (5) 943- 948.
- Jiménez D, Aujesky D, Moores L, et al. Simplification of the Pulmonary Embolism Severity Index for Prognostication in Patients With Acute Symptomatic Pulmonary Embolism. Arch Intern Med 2010;170(15):1383-1389.
- Righini M, Roy PM, Meyer G, Verschuren F, Aujesky D, and Le Gal G. The Simplified Pulmonary Embolism Severity Index (SPESI): validation of a clinical prognostic model for pulmonary embolism. J Thromb Haemost 2011; 9: 2115-2117.
- Fermann GJ, Erkens PMG, Prins MH, et al. Treatment of Pulmonary Embolism With Rivaroxaban: Outcomes by Simplified Pulmonary Embolism Severity Index Score from a Post Hoc Analysis of the EINSTEIN PE Study. Acad Emerg Med 2015;22(3):299-307.
- Wicki J, Perrier A, Perneger TV, Bounameaux H, and Junod AF. Predicting adverse outcome in patients with acute pulmonary embolism: a risk score. Thromb Haemost 2000; 84:548-552.
- Kohn CG, Mearns ES, Parker MW, et al. Prognostic Accuracy of Clinical Prediction Rules for Early Post-Pulmonary Embolism All-Cause Mortality: A Bivariate Meta-analysis. Chest 2015 Apr 1;147(4):1043-62.
- Paiva LV, Providencia RC, Barra SN, Faustino AC, Botelho AM, Marques AL. Cardiovascular risk assessment of pulmonary embolism with the GRACE risk score. Am J Cardiol 2013;111(3):425-431.
- Maestre A, Trujillo-Santos J, Riera-Mestre A, Jiménez D, Di Micco P, RIETE Investigators. Identification of Low-Risk Patients with Acute Symptomatic Pulmonary Embolism for Outpatient Therapy. Ann Am Thorac Soc 2015 Aug;12(8):1122-9.
- Lankeit M, Jimenez D, Kostrubiec M, Dellas C, Kuhnert K, Hasenfuss G, Pruszczyk P, Konstantinides S. Validation of N-terminal pro-brain natriuretic peptide cut-off values for risk stratification of pulmonary embolism. Eur Respir J 2014; 43: 1669–77.
- Jimenez D, Kopecna D, Tapson V, Briese B, Schreiber D, Lobo JL, Monreal M, Aujesky D, Sanchez O, Meyer G, Konstantinides S, Yusen RD, On Behalf of the Protect I. Derivation and validation of multimarker prognostication for normotensive patients with acute symptomatic pulmonary embolism. Am J Respir Crit Care Med 2014; 189: 718–26.
- Singanayagam A, Scally C, Al-Khairalla MZ, Leitch L, Hill LE, Chalmers JD, Hill AT. Are biomarkers additive to pulmonary embolism severity index for severity assessment in normotensive patients with acute pulmonary embolism? QJM 2011; 104: 125–31.
- Sanchez O, Trinquart L, Planquette B, Couturaud F, Verschuren F, Caille V, Meneveau N, Pacouret G, Roy PM, Righini M, Perrier A, Bertoletti L, Parent F, Lorut C, Meyer G. Echocardiography and pulmonary embolism severity index have independent prognostic roles in pulmonary embolism. Eur Respir J 2013; 42: 681–8.
- Moores L, Aujesky D, Jimenez D, Diaz G, Gomez V, Marti D, Briongos S, Yusen R. Pulmonary embolism severity index and troponin testing for the selection of low-risk patients with acute symptomatic pulmonary embolism. J Thromb Haemost 2010; 8: 517–22.
- Pollack C. New oral anticoagulants in the ED setting: a review. Am J Emerg Med 2012;30:2046-2054.
- The EINSTEIN–PE Investigators. Oral Rivaroxaban for the Treatment of Symptomatic Pulmonary Embolism. N Engl J Med 2012; 366:1287-1297.
- Agnelli G, Buller HR, Cohen A, Curto M, Gallus AS; AMPLIFY Investigators. Oral apixaban for the treatment of acute venous thromboembolism. N Engl J Med 369(9):799-808.
- Loffredo L, Perri L, Del Ben M, et al. New oral anticoagulants for the treatment of acute venous thromboembolism: are they safer than vitamin K antagonists? A meta-analysis of the interventional trials. Intern Emerg Med 2015;10:499-506.
- Lovenox (package insert). Sanofi-Aventis, Bridgewater, NJ; 2011.
- Pradaxa [package insert]. Boehringer Ingelheim Pharmaceuticals, Inc., Ridgefield, CT; 2011.
- Xarelto [package insert]. Janssen Pharmaceuticals, Inc., Titusville, NJ; 2011.
- Eliquis [summary of product characteristics]. Bristol-Myers Squibb/Pfizer EEIG, Middlesex, UK; 2011.
- Pisters R, Lane DA, Nieuwlaat R, de Vos CB, Crijns HJ, Lip GY. A novel user-friendly score (HAS-BLED) to assess 1-year risk of major bleeding in patients with atrial fibrillation: the Euro Heart Survey. Chest 2010 Nov;138(5):1093-100.
- Kearon C, Gent M, Hirsh J, et al. Extended anticoagulation compared to placebo after three months of therapy for a first episode of idiopathic venous thromboembolism. N Engl J Med 1999;34:901-907.
- Hart RG, Boop BS, Anderson DC. Oral anticoagulants and intracranial hemorrhage. Facts and hypotheses. Stroke 1995; 26:1471-1477.
- Lip GH, Frison L, Halperin JL, Lane DA. Comparative Validation of a Novel Risk Score for Predicting Bleeding Risk in Anticoagulated Patients With Atrial Fibrillation: The HAS-BLED (Hypertension, Abnormal Renal/Liver Function, Stroke, Bleeding History or Predisposition, Labile INR, Elderly, Drugs/Alcohol Concomitantly) Score. J Am Coll Cardiol 2011;57(2):173-180.
- Apostolakis S, Lane DA, Guo Y, Buller H, Lip GY. Performance of the HEMORR(2)HAGES, ATRIA, and HAS-BLED bleeding risk-prediction scores in patients with atrial fibrillation undergoing anticoagulation: the AMADEUS (evaluating the use of SR34006 compared to warfarin or acenocoumarol in patients with atrial fibrillation) study. J Am Coll Cardiol 2012 Aug 28;60(9):861-7.
- Roldán V, Marín F, Fernández H, et al. Predictive Value Of The Has-Bled And Atria Bleeding Scores For The Risk Of Serious Bleeding In A “Real-World” Population With Atrial Fibrillation Receiving Anticoagulant Therapy. Chest 2013;143(1):179-184.
- Wells PS, Forgie MA, Simms M, et al. The Outpatient Bleeding Risk Index: Validation of a Tool for Predicting Bleeding Rates in Patients Treated for Deep Venous Thrombosis and Pulmonary Embolism. Arch Intern Med 2003;163(8):917-920.
- Landefeld CS, Rosenblatt MW, Goldman L. Bleeding in outpatients treated with warfarin: relation to the prothrombin time and important remediable lesions. Am J Med 1989;87:153- 159.
- Beyth RJ, Quinn LM, Landefeld CS. Prospective evaluation of an index for predicting the risk of major bleeding in outpatients treated with warfarin. Am J Med 1998;10:591-99.






2 thoughts on “Outpatient PE Management: Controversies, Pearls, and Pitfalls”
Pingback: emDOCs.net – Emergency Medicine EducationJournalFeed Weekly Wrap-Up - emDOCs.net - Emergency Medicine Education
Pingback: CanadiEM Frontline Primer - Pulmonary Embolism - CanadiEM