Authors: Taylor Franz, DO (EM Resident Physician, Virginia Tech Carilion), Janet S. Young, MD (Associate Professor of Emergency Medicine, Virginia Tech Carilion School of Medicine, Roanoke, Virginia) // Reviewed by: Stephen Alerhand, MD; Alex Koyfman, MD (@EMHighAK); Brit Long, MD (@long_brit)
Case:
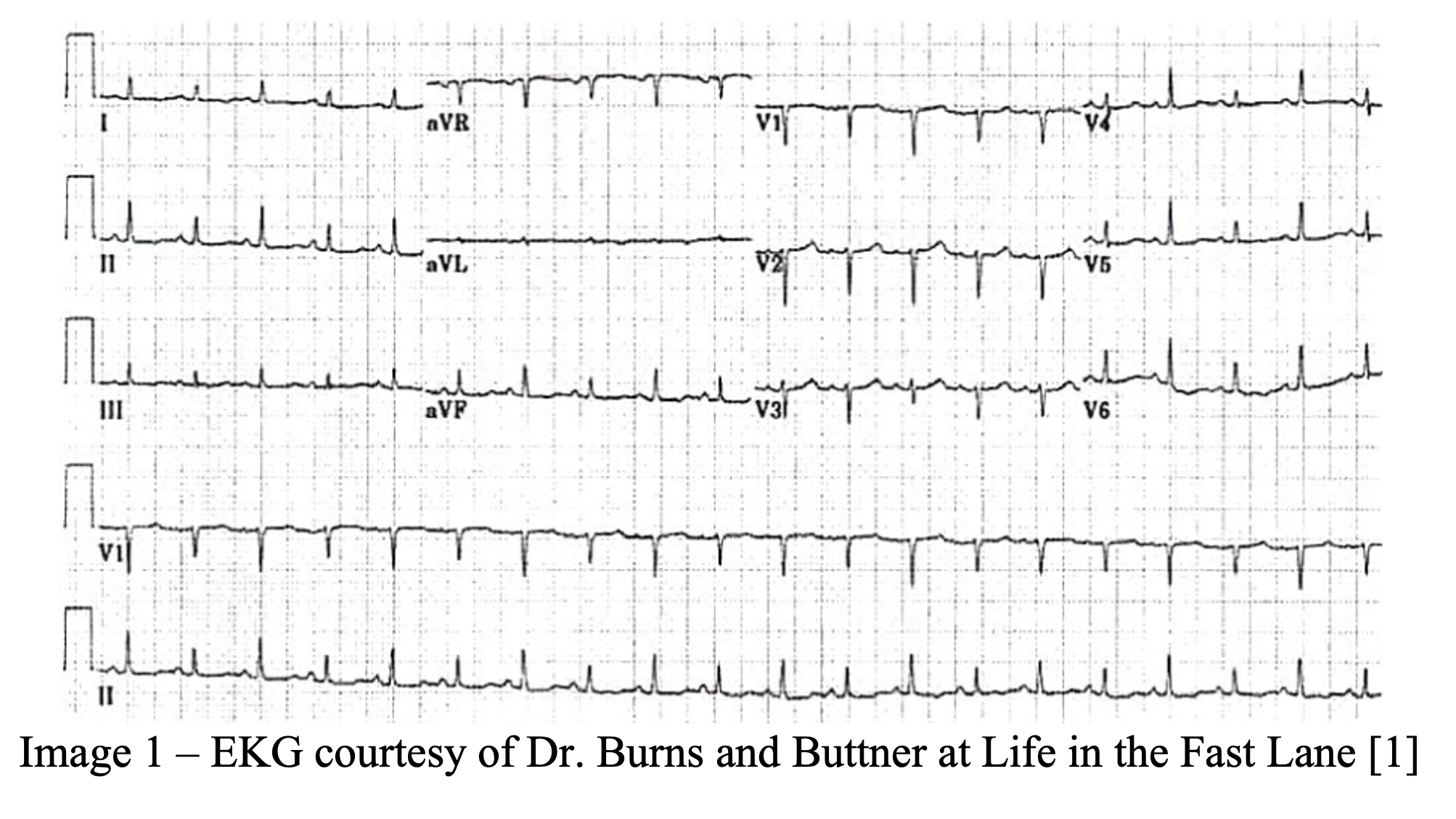
A 75-year-old male with a past medical history of hypertension, hyperlipidemia, and tobacco use presents to the emergency department with chest pain and shortness of breath. The patient describes his chest pain as a dull, pressure-like sensation that worsens with lying flat. The patient describes worsening dyspnea on exertion over the past 24 hours. The patient is placed on telemetry monitoring with pulse oximetry as intravenous lines are established. Vital signs include BP 92/50, HR 107, oxygen saturation 95% on 4L via nasal cannula. An electrocardiogram demonstrates sinus tachycardia with global low voltage and QRS complexes of varying amplitudes from beat-to-beat.
What is the presumed diagnosis? What can you do at the bedside to confirm your suspected diagnosis? What are the next steps in your treatment of this patient?
Answer: Pericardial effusion with possible cardiac tamponade.
Etiology:
While the most common causes of acute pericarditis are viral or idiopathic, pericardial effusions can develop from several other etiologies as seen in Table 1. Pathophysiologic etiologies can be predictive of disease progression from effusion to tamponade. Cardiac tamponade may occur in up to 50% of patients with malignant, tuberculous, or bacterial disease while only 10-15% of patients with idiopathic etiologies progress to tamponade physiology [2-4].

Pathophysiology:
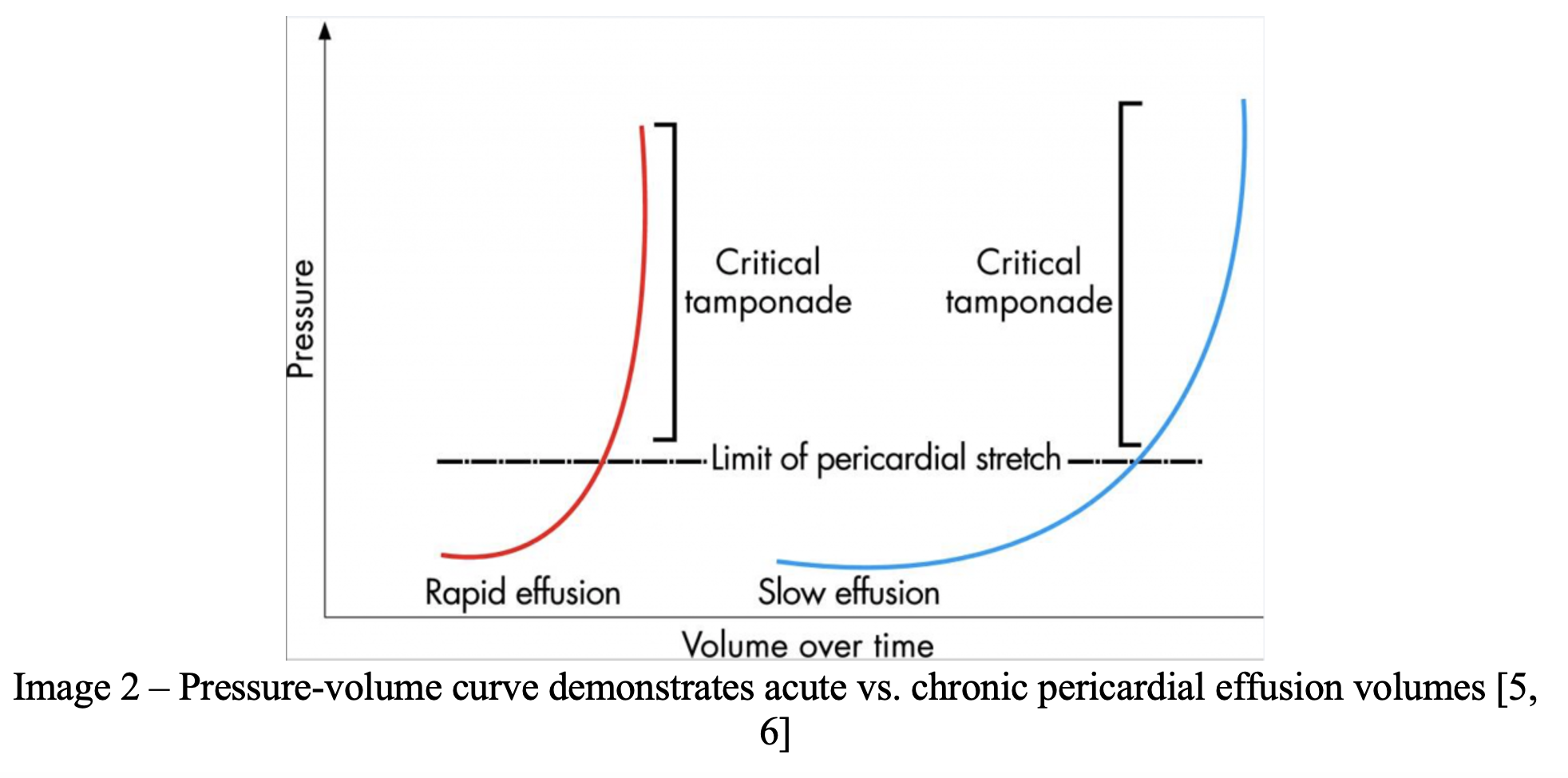
The signs and symptoms of pericardial effusion depend on the rapidity of accumulation and the compliance of the pericardial sac. The pericardial space holds an average of 15-50 mL of pericardial fluid. As little as 100 mL of pericardial fluid can increase intrapericardial pressure enough to develop cardiac tamponade in the acute setting. By comparison, chronic pericardial effusions can grow much larger and have been documented to have volumes in excess of 2 L [2, 5, 6].
Ventricular interdependence
The phenomenon of ventricular interdependence explains the clinical findings of cardiac tamponade. Under normal physiologic circumstances, the right and left ventricles compete for space within the pericardial sac. When present, pericardial effusions also compete for space within the pericardial sac and can interfere with normal ventricular function. Cardiac tamponade occurs when the intrapericardial pressures have increased to the point where they are equal to or greater than the intracardiac pressures. With normal inspiration, right ventricular filling is enhanced, and the interventricular septum is deviated to the left. This deviation of the interventricular septum causes decreased left ventricular filling and, therefore, decreased stroke volume. In tamponade physiology, this decrease in stroke volume is exaggerated and is responsible for the clinical finding of pulsus paradoxus (discussed later) [2, 5, 7].
Equalization of right atrial and right ventricular pressures
Systemic blood pressure in cardiac tamponade is temporarily maintained by the sympathetic nervous system via tachycardia and peripheral vasoconstriction. Cardiac output can be maintained, provided the right ventricular pressure exceeds the right atrial pressure. Peripheral vasoconstriction and pooling of blood in the venous system causes an increase in central venous pressure that is routinely noted with pericardial effusions. An increase in central venous pressure translates to increased right atrial pressures and ultimately equalization of pressures between the right atrium and right ventricle. At this point, diastolic filling of the right ventricle is impaired, resulting in decreased cardiac output and hypotension [5].
Presentation:
Unless a hemodynamically significant effusion is present, patient’s presenting symptoms are usually nonspecific and insensitive. In the case of hemodynamically insignificant effusions, vague symptoms may be related to the underlying cause of the pericardial effusion. In patients with a hemodynamically significant effusion, patient present with signs and symptoms of impaired cardiac function [3]. Stolz and colleagues looked at patient presenting complaints and past medical history in cases of pericardial effusion. Table 2 below demonstrates the top presenting symptoms of patients who were found to have ultrasound confirmed pericardial effusions [9].

Evaluation:
Physical exam
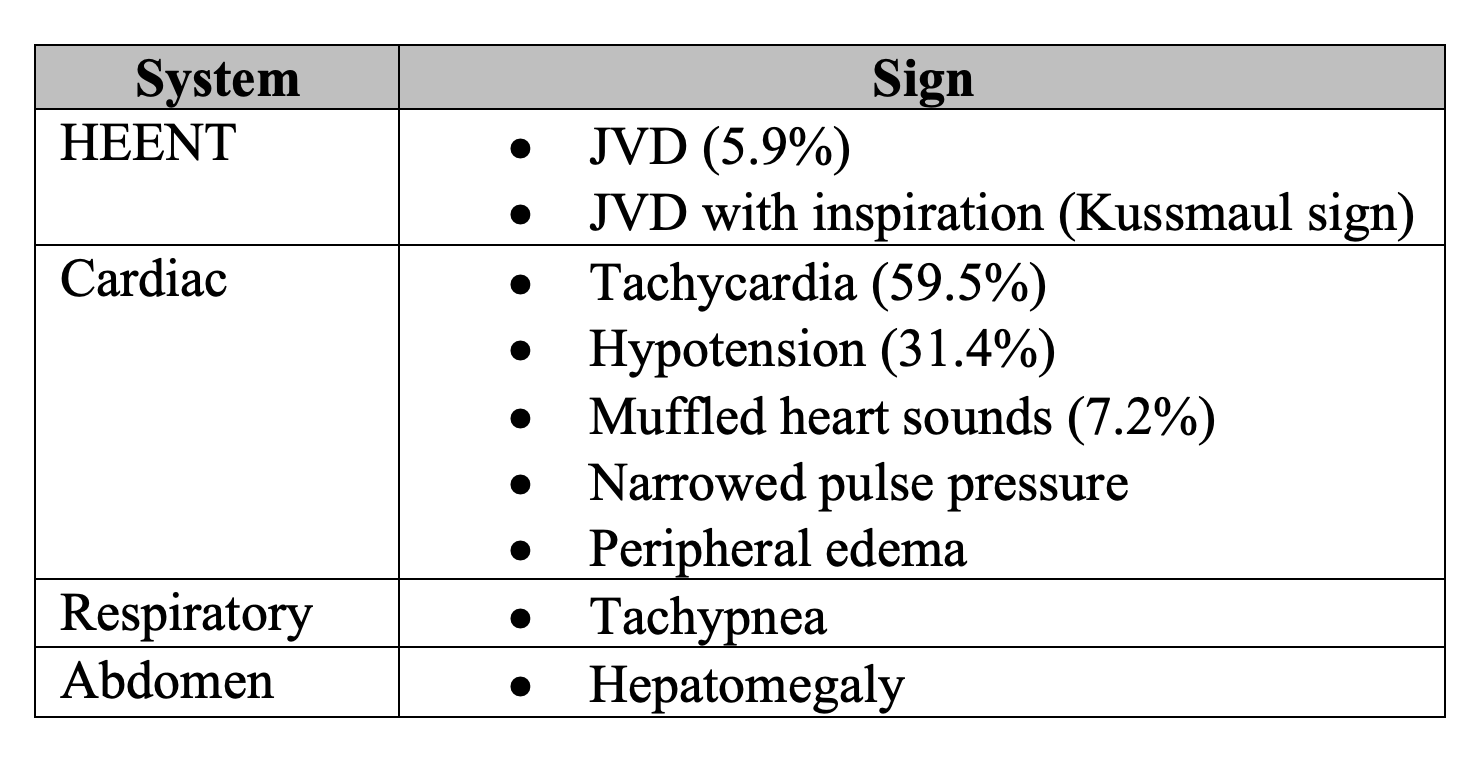
Table 3, adapted from Stolz retrospective study, demonstrates the most common physical exam findings in patients with pericardial effusion [9]. Studies have been unable to consistently demonstrate the percentage frequency of physical exam findings. Tachycardia and tachypnea have routinely been the most common findings of pericardial effusion and have been reported to be present in up to 70% of cases [8-10].
Pulsus Paradoxus
- A small decrease in systolic blood pressure (< 10 mmHg) can be seen as a normal physiologic variant. Pulsus paradoxus is defined as greater than 10 mmHg decrease in systolic blood pressure with inspiration. Although it has been a hallmark of the disease process, pulsus paradoxus may present as a late finding in cardiac tamponade and significant hemodynamic instability can be seen earlier with ultrasound [7]. Pulsus paradoxus can also be seen in massive pulmonary embolism, hemorrhagic shock, or obstructive lung disease [11].
Beck’s triad
- The classic board question of Beck’s triad is made up of jugular venous distention, muffled heart sounds, and hypotension. While all three findings may be present with pericardial effusions, the presence or absence does not make the final diagnosis. In a retrospective study of 150 patients with identified pericardial effusion, zero had all elements of Beck’s triad [9].
- The most common electrocardiographic finding in pericardial effusion and cardiac tamponade is tachycardia. Table 4 compares presence of EKG findings in pericardial effusion versus cardiac tamponade. Arugula and colleagues found the presence of tachycardia, low voltage, and electrical alternans had a positive predictive value of 100% for cardiac tamponade in patients with known malignant pericardial effusions [13].

CXR
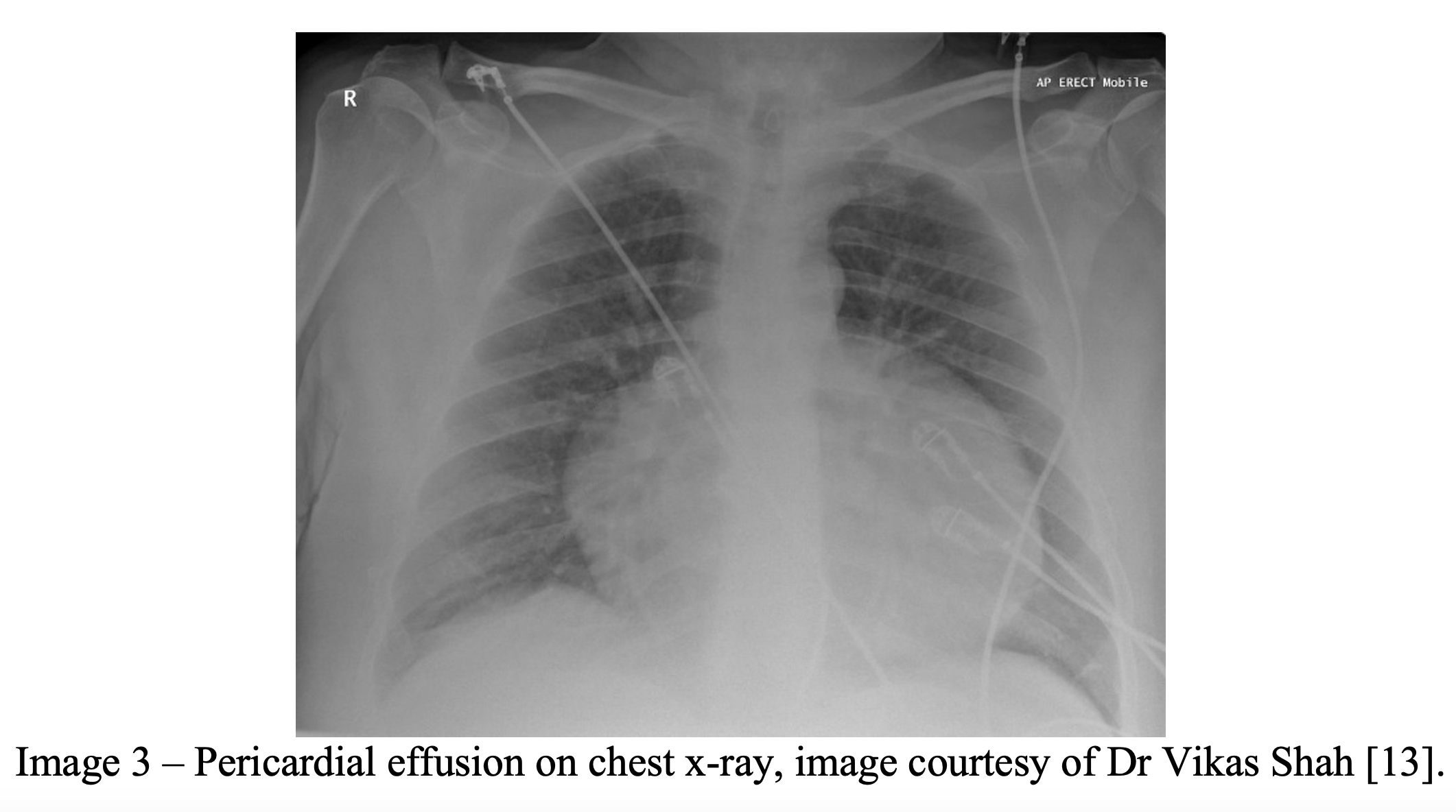
- Radiographic findings of pericardial effusions are nonspecific and mainly used to rule out other etiologies of presenting complaints and physical exam findings.
- Radiographs can show a large, globular heart that has been likened to an old-fashioned water bottle (Image 3) [8, 13].
- Pleural effusions without pulmonary congestion can also be seen on plain films [2, 10, 13]
- CT imaging of the chest can be performed in a stable patient and can make the diagnosis of pericardial effusion; however, you cannot determine if tamponade physiology is present [10].
Bedside Echocardiography
- When performed by emergency physicians, sensitivity and specificity in identification of pericardial effusion has been shown to be as high as 96% and 99%, respectively [14]. Echocardiography can detect as little as 20 mL of pericardial fluid [5].
- Pericardial effusions can be visualized in all four traditional cardiac views (parasternal long axis [PLAX], parasternal short axis [PSAX], apical 4-chamber [A4C], and subxiphoid [SX]). Pericardial effusions should be confirmed in multiple views to increase specificity and rule in/out possible mimics (i.e. pericardial fat pad, pleural effusion) [15, 16]. Estimated volumes of pericardial effusion determined by myocardium-pericardium distance is demonstrated below in Table 5[18].

- Right ventricular collapse in diastole – As described above, cardiac tamponade occurs when the pericardial pressure > intracardiac pressures. The right ventricle (RV) is a thin walled structure compared to the left ventricle (LV), therefore, will collapse under pressure much easier. RV collapse in diastole is the most specific finding of cardiac tamponade [15, 17]. RV collapse can be seen in any of the cardiac views and is demonstrated PLAX view below (Image 4) [15].
Image 4 – Ultrasound clip courtesy of Dr. Jon Nogueira.
- Right atrial collapse in early diastole/systole – Right atrial (RA) pressure < RV pressure and it is lowest during early diastole/systole. During atrial relaxation the RA can collapse under much lower pericardial pressures making RA collapse the first sign of cardiac tamponade. Although it can be the first sonographic finding of tamponade, it lacks the specificity of RV collapse and is much more variable [15, 17]. Below is a clip of RA collapse during systole in the SX view (Image 5).
Image 5 – Ultrasound clip courtesy of Dr. Jon Nogueira.
- Plethoric IVC with lack of inspiratory variation – The decreased cardiac output seen in cardiac tamponade causes back up of blood into the RV and RA leading to decreased inflow from venous circulation. This can be seen on ultrasound as a plethoric IVC with lack of respiratory variation. While a plethoric IVC can be very sensitive for cardiac tamponade it lacks specificity due to other conditions that can cause this finding [15, 17]. Image 6 demonstrates a plethoric IVC with lack of respiratory variation.
Image 6 – Ultrasound clip courtesy of Dr. Jon Nogueira.
- Increased Mitral/Tricuspid valve in-flow velocities – As described above, the phenomenon of ventricular interdependence is vital to understanding tamponade physiology and the finding of pulsus paradoxus. Thoracic pressure decreases with inspiration leading to increased RV filling. MV and TV in-flow velocities can be measured by using pulse wave Doppler and placing the gate between the MV or TV leaflets. Measurements are taken from the first heartbeat in the respiratory cycle (highest waveform) to the last (lowest waveform). In-flow velocity decrease > 25% is a surrogate marker for decreased LV filling and correlates with the > 10 mmHg decrease in SBP seen in pulsus paradoxus (Image 7) [15].

For an in-depth review of ultrasound findings in cardiac tamponade, check out Dr. Alerhand’s post from June 2018.
Treatment:
Initial treatment of pericardial effusions and cardiac tamponade should be focused on the ABCs. Intubation and mechanical ventilation should be avoided if possible, as any form of positive pressure ventilation increases intrathoracic pressure leading to decreased venous return and decreased cardiac output. IVF resuscitation should be used to expand intravascular volume in attempts to maintain a perfusing blood pressure. Dobutamine, dopamine and norepinephrine have all been cited as appropriate vasopressor doses in treating hypotension secondary to cardiac tamponade although the literature is out of date and lacks specific dosing [10]. In hemodynamically stable patients, the preferred method for treatment of pericardial effusion is pericardiocentesis performed under fluoroscopy by an interventional cardiologist. Hemodynamically stable patients may also go to the operating room with cardiothoracic surgeons for a pericardial window. For those patients who are not stable enough to leave the emergency department, a bedside pericardiocentesis must be performed. This procedure should be done in consultation with cardiology unless the patient is actively decompensating [17, 20].
Ultrasound-guided pericardiocentesis:
- Elevate the head of the bed 30-45° which will bring the heart closer to the anterior chest wall [10]. Placing the patient in the left lateral decubitus position will also bring the heart closer to the anterior chest wall [17].
- Obtain your cardiac windows and identify the largest fluid collection closest to the needle pathway [17]. Ultrasound-guided pericardiocentesis can be performed with any of the 4 main cardiac views, but is most commonly performed in the apical and subxiphoid views, although parasternal approach may also be used [20].
- Clean anterior and left chest wall and upper abdomen with betadine. Prepare a large field for the possibility that a second approach may be required. Create your sterile field including ultrasound cover and sterile gel [10, 17].
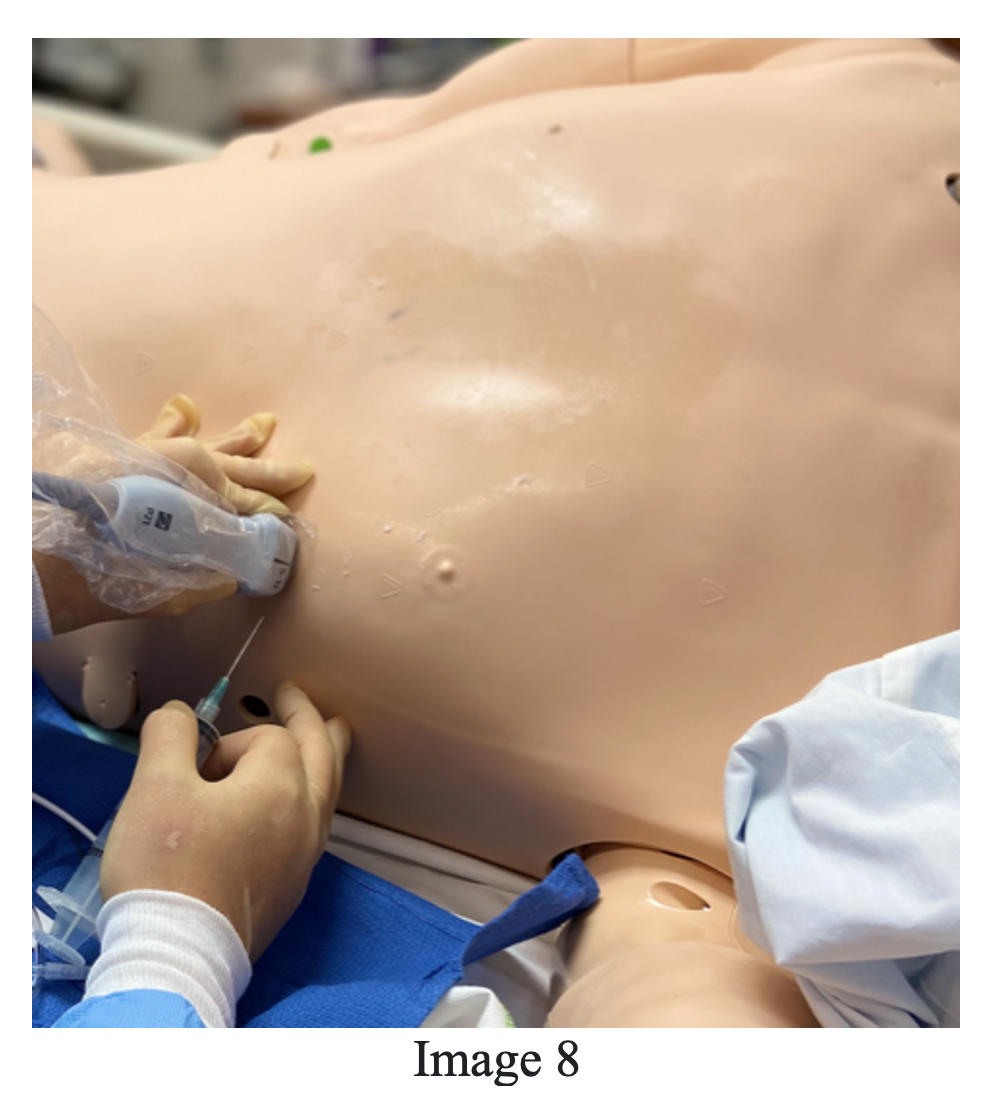
- [If time allows] infiltrate your entry point with local anesthetic. An 18-gauge or larger needle attached to a 10 mL syringe should be entered above the nearest rib space (avoiding neurovascular bundle on lower rib) and visualized with ultrasound at all times. Image 8 demonstrates apical approach with probe and needle placement, courtesy of Dr. Roberta Pritchard [17].
- Aspirate during insertion and stop once your needle tip is visualized in the pericardial space and fluid is returned. If the location of the needle tip is uncertain, 3-5 mL of fluid can be re-injected and visualized in the pericardial space confirming location [17].
- Once confirmed that you are in the pericardial space a guidewire and pigtail catheter can be inserted using Seldinger Technique. Aspiration of as little as 50 mL of fluid can result in significant hemodynamic change and resolution of tamponade physiology [10].
The SX approach the patient will be in the supine position and the needle should be inserted approximately 1 cm at a 30° angle to the skin aiming at the left shoulder. In-plane ultrasound visualization of the needle is most important in the subxiphoid approach to make sure you are avoiding the liver [17].
Pearls:
- Predictive morbidity of developing tamponade may be dependent upon causative etiology. Malignant and tuberculous/bacterial effusions are more likely to develop cardiac tamponade when compared to viral or iatrogenic causes.
- Tachycardia is the most common presenting sign for pericardial effusion. EKG findings are tachycardia, low voltage, and electrical alternans. Bedside ultrasound is diagnostic.
- Hemodynamically stable cardiac tamponade can be managed with pericardiocentesis or pericardial window.
- Hemodynamically unstable patients need emergent ultrasound-guided pericardiocentesis. Prep a large area of the chest in case you need to change your approach. Keep your needle in-plane at all times to avoid damage to surrounding structures.
References/Further Reading:
- Burns MD, Ed. ECG Findings in Massive Pericardial Effusion. Life in the Fast Lane. https://litfl.com/ecg-findings-in-massive-pericardial-effusion/. Published February 3, 2021. Accessed April 26, 2021.
- Hoit, Brian D; Oh, Jae K. Goldman-Cecil Medicine. 20th ed. Chapter 68: Pericardial Disease. Pp 428-436. https://www.clinicalkey.com/#!/content/book/3-s2.0-B9780323532662000680. Accessed April 17, 2021.
- Hoit MD, Brian D. Diagnosis and Treatment of Pericardial Effusion. UpToDate. UpToDate, 2021. Accessed April 17, 2021. uptodate.com
- Hoit MD, Brian D. Etiology of Pericardial Disease. UpToDate. UpToDate; 2021. Accessed April 17, 2021. uptodate.com
- Modak, Raj K; Maracaja, Luiz. Stoeltin’s Anesthesia and Co-Existing Disease. Pericardial Disease and Cardiac Trauma. Pp 225-236. https://www.clinicalkey.com/#!/content/book/3-s2.0-B9780323401371000119?scrollTo=%23hl0000299. Accessed April 17, 2021.
- Ivens, E.L., B.I. Munt, and R.R. Moss. Pericardial disease: what the general cardiologist needs to know. Heart, 2007. 93(8): p. 993-1000.
- Shabetai MD, Ralph; Oh MD, Jae. Cardiology Clinics: Pericardial Diseases. Volume 35, Issue 4. Pericardial Effusion and Compressive Disorders of the Heart. Pp 467-479. https://www.clinicalkey.com/#!/content/journal/1-s2.0-S0733865117300619. Accessed April 17, 2021.
- Simon DO, MHA, Erica. EM@3AM – Cardiac Tamponade. emDOCS. http://www.emdocs.net/em3am-cardiac-tamponade/. Published May 28, 2017. Accessed April 17, 2021.
- Stolz L, Valenzuela J, Situ-LaCasse E, et al. Clinical and historical features of emergency department patients with pericardial effusions. World J Emerg Med. 2017;8(1):29-33. doi:10.5847/wjem.j.1920-8642.2017.01.005
- Honasoge MD, Akilesh P; Dubbs MD, Sarah B. Emergency Medicine Clinics of North American: Hematologic and Oncologic Emergencies. Volume 36, Issue 3. Rapid Fire: Pericardial Effusion and Tamponade. Pp 557-565. https://www.clinicalkey.com/#!/content/journal/1-s2.0-S0733862718300270. Accessed April 17, 2021.
- Mattu A, Martinez J. Pericarditis, Pericardial Tamponade, and Myocarditis. In Emergency Medicine Clinical Essentials, 2nd ed. Philadelphia, Saunders. 2013.
- Argula RG, Negi, Blanchs J, et al. Role of a 12-lead electrocardiogram in the diagnosis of cardiac tamponade as diagnosed by transthoracic echocardiography in patients with malignant pericardial effusion. Clinical Cardiology, 2015; 38 (3), 139-144.
- Feger MD, Joachim. Pericardial effusion. Radiopaedia. https://radiopaedia.org/articles/pericardial-effusion?lang=us. Accessed April 26, 2021.
- American College of Emergency Physicians: Emergency ultrasound guidelines-2008. Ann Emerg Med 53:550, 2009.
- Alerhand MD, Stephen. US Probe: When Does an Effusion Become Pericardial Tamponade? emDOCs. http://www.emdocs.net/us-probe-when-does-an-effusion-become-pericardial-tamponade/. Published June 27, 2018. Accessed April 17, 2021.
- Mallin, Michael P; Butts, Christine. Emergency Medicine: Clinical Essentials. 1st ed. Emergency Cardiac Ultrasound: Evaluation for Pericardial Effusion and Cardiac Activity. Pp 43-49. https://www.clinicalkey.com/#!/content/book/3-s2.0-B9781437735482000057. Accessed April 17, 2021.
- Pritchard MD, Roberta; Clinton MD, Michelle; and Nogueira DO, Jonathan. The POCUS Report – Ultrasound-guided pericardiocentesis. American Academy of Emergency Medicine. https://www.aaem.org/get-involved/sections/eus/newsletter/fall-2020. Published Fall 2020. Accessed April 17, 2021.
- Alerhand, MD, Stephen. American Journal of Emergency Medicine. Volume 37, Issue 2. What echocardiographic findings suggest a pericardial effusion is causing tamponade? Pp 321-326. https://www.clinicalkey.com/#!/content/journal/1-s2.0-S0735675718309124. Accessed May 5, 2021.
- Zgheib H, Wakil C, Shayya S, Bachir R, El Sayed M. Effectiveness and outcomes of 2 therapeutic interventions for cardiac tamponade: A retrospective observational study. Medicine (Baltimore). 2020;99(29):e21290. doi:10.1097/MD.0000000000021290
- Osman A, Wan Chuan T, Ab Rahman J, Via G, Tavazzi G. Ultrasound-guided pericardiocentesis: a novel parasternal approach. Eur J Emerg Med. 2018;25(5):322-327. doi:10.1097/MEJ.0000000000000471