Authors: Thomas Yang, MD (EM Resident Physician, Aventura Hospital and Medical Center, Aventura, FL) and Annalee Baker, MD (Assistant Program Director, Emergency Medicine Residency, Aventura Hospital and Medical Center, Aventura, FL) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Clinical Case
A 45-year-old woman with a 20-year history of Crohn’s disease presented with fever, chills, and fatigue for 10 days. Shortly before the onset of symptoms, the patient underwent an uncomplicated colonoscopy during which she had 17 biopsies performed for surveillance or polypectomy. During colonoscopy, her Crohn’s disease was in remission, and there was no mucosal inflammation noted.
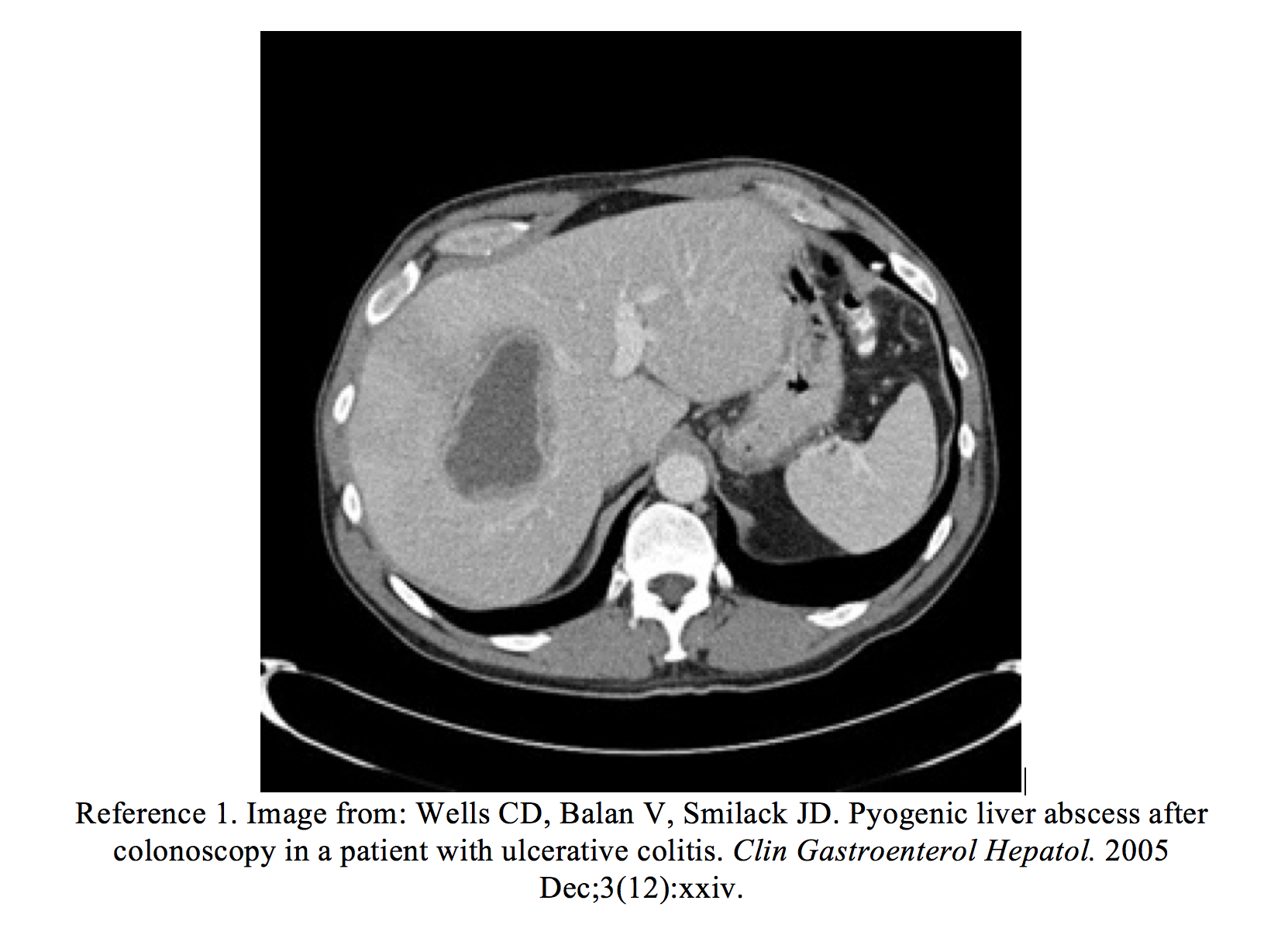
In the ED, the patient’s vital signs were HR 110, BP 120/80, RR 18, T 102.5. She had mild right upper abdominal tenderness. Laboratory studies revealed an elevated white blood cell count (15,000) and elevated AST and ALT (120/137 U/L), with slightly elevated alkaline phosphatase of 200 IU/L. A computed tomography (CT) scan of the abdomen revealed multiple areas of low-attenuation in the right hepatic lobe consistent with liver abscesses. Drainage of the abscesses was cultured and grew Streptococcus anginosus. Resolution of all lesions occurred after they were drained by interventional radiology, and the patient was given a 6-week course of intravenous antibiotics.
Introduction
Colonoscopy is a commonly performed procedure in the diagnosis and treatment of a wide range of conditions including cancer, inflammatory bowel diseases, polyps, bleeding, and strictures. According to the American Cancer Society, screening colonoscopy is recommended every 10 years starting at age 50.1 In high risk patients such as those with inflammatory bowel disease, colonoscopy is performed more often. The procedure is still the most effective way to find and remove small pre-cancerous polyps, and thereby decrease the risk of death from colon and rectal cancer.
In general, colonoscopy is very safe. A 2008 systematic review of 12 studies totaling 57,742 colonoscopies performed for average risk screening showed an overall serious adverse event rate of just 2.8 per 1000 procedures and mortality of 0.007%.2 However, the risk of colonoscopy is not constant across groups, and some complications are more common than others. Older adults are at increased risk for serious complications compared with younger patients. In a study of 53,220 colonoscopies, patients aged 80 to 84 years had a significantly higher rate of serious complications compared with patients aged 66 to 69 years. The risk of serious complications was also increased among patients with comorbid conditions such as a history of stroke, COPD, atrial fibrillation, and heart failure.
Pyogenic liver abscess (PLA) is a rare but serious complication of colonoscopy. Most complications of colonoscopy will present within 7 days, but PLA may present days or even weeks later than that, and the history of recent colonoscopy may not even be elicited unless the clinician is aware of this entity.4-6 Because of the delayed nature of PLA, these patients are likely to be diagnosed in the emergency department.
Other late complications such as post-polypectomy electrocoagulation syndrome, and the more common problems of delayed hemorrhage, nonspecific abdominal pain, reactions to sedation agents, and perforation can also be seen in the ED. By contrast, gas explosion or immediate bleeding will rarely be seen in the ED, as these issues typically present rapidly in the endoscopy suite, where they are addressed emergently. ED physicians must be aware of all possible complications and their time course and have the tools to correctly diagnose and treat the unlucky post-colonoscopy patient who suffers a serious complication.
Complications
Pyogenic Liver Abscess
PLA is a dangerous disease with high morbidity and mortality that occurs at a rate of 10 to 20 cases per 100,000 hospital admissions.4-6 With advances in diagnostic techniques and interventions, mortality is improving.7 However patients are still in grave danger, especially if the diagnosis is delayed, as it often is, because patients may present nonspecifically. A retrospective study of 63 patients with PLA showed that fever was common (59%), but other signs and symptoms such as right upper quadrant pain and peritoneal signs were present in only 39% and 14%, respectively.8 Diagnosis in this study was often delayed an average of one week from symptom onset.
Colonoscopy is a rare but also probably under-recognized precipitant of PLA. It is thought that silent micro-perforations during the procedure lead to infection which can seed the liver through the portal circulation. Similarly, other intra-abdominal infections such as diverticulitis can potentially cause PLA if the infected area is drained by the portal vein.9 Hepatic clearance of bacteria via the portal network is a normal phenomenon in healthy patients, but biliary obstruction, poor perfusion, and high bacterial load (as in the case of infection or multiple biopsies during colonoscopy seeding the bloodstream) can predispose to proliferation and abscess formation. The literature describes approximately 34% of PLA cases as cryptogenic, but a portion of these may well be patients with recent colonoscopy that was not recognized as a risk factor for the disease process.8
Most PLA isolates are polymicrobial and comprised of normal enteric flora. Blood cultures are positive in 33-65% of cases, but abscess cultures are nearly all positive.10 The most commonly isolated microorganisms are Streptococcus species (37%), Eserichia Coli (33%), Bacteroides species (24%), Klebsiella pneumoniae (18%), and microaerophilic Streptococci (12%).10-11 Strep anginosus, found in this patient’s case, is a facultative anaerobic Gram-positive coccus and one of the more common organisms found in PLA.
Prompt diagnosis of PLA is critical, because even as diagnosis and therapy improves, mortality remains at 10%.5,7 Infectious complications of colonoscopy in general are rare, but again the causal relationship is also probably under-recognized. Colonoscopy has been implicated in at least a few cases of PLA.12-14 But transient systemic bacteremia, seen in 4% of colonoscopies, does not seem to increase risk of infection per se.15 Rather, the infection in PLA is thought to spread via actual microperforations.
While it is known that frank perforations are relatively rare (less than 0.1% of colonoscopies), the incidence and pathophysiology of microperforations is not entirely known.16-17 Armed with knowledge of this clinical entity, a clinician who elicits a good history and has a high index of suspicion can more quickly diagnose PLA in a patient with recent colonoscopy.
Infection
It is commonly accepted that transient bacteremia after colonoscopy occurs in approximately 4% of procedures, though the range is reported as 0-25%, and risk factors for bacteremia are not defined.18 Higher rates of bacteremia occur during routine daily activities such as tooth brushing and chewing food. Although signs or symptoms of systemic infection in patients with transient bacteremia are rare, patients may feel fever, chills, and complaints mimicking everything from upper respiratory to abdominal infections.18
Although transient bacteremia is an accepted risk of colonoscopy, there is no proven benefit for routine antibiotic prophylaxis, and most patients will not have taken peri-procedure antibiotics.18 Current guidelines from the American Society for Gastroenterology (ASGE) agree with the American Heart Association (AHA) and recommend against antibiotic prophylaxis for patients undergoing routine colonoscopy.19
It can be difficult to differentiate symptoms of transient bacteremia from true infection.18 In general, patients with infectious symptoms in the days following colonoscopy warrant laboratory studies including blood cultures, and depending on the index of suspicion for perforation, abdominal imaging.20-21 If suspicion for perforation is low or the diagnosis is ruled out, IV hydration and antibiotics can be given if warranted by the patient’s clinical status.
Bleeding
Post-polypectomy bleeding can be either immediate or delayed. Immediate bleeding is associated with polypectomy techniques that do not use cautery or when blended current is used for the procedure. Delayed bleeding is more commonly seen in the ED, as immediate bleeding is often detected at the time of procedure and addressed by the endoscopist. Delayed bleeding can be seen up to 7 days after the procedure. It is thought to occur due to sloughing of an eschar that was covering a blood vessel or due to extension of the zone of thermal necrosis to non-injured tissue, which could result in bleeding if it involves a blood vessel.19,22
Several large studies have reported hemorrhage in 1 to 6 per 1000 colonoscopies (0.1%-0.6%).22 However, a study analyzing over 50,000 colonoscopies found the rate of GI hemorrhage was significantly different depending whether polypectomy was performed. Post-polypectomy bleeding occurs at 1-2%, with higher rates seen with the removal of larger polyps. The risk of post-polypectomy bleeding may be increased in patients with thrombocytopenia or coagulopathies.23
Known risk factors for bleeding post-colonoscopy include polyp size, number of polyps removed, history of recent anticoagulant use, or polyp histology.22 Oddly enough, multiple, large studies did not find aspirin use associated with post-polypectomy bleeding.24-26
Patients with delayed hemorrhage can present with any signs of bleeding or anemia, including fatigue, weakness, pallor, gross bleeding or clots upon wiping, tachycardia, chest pain, and shortness of breath. Patients might describe hematochezia or melena, depending upon the location of the bleeding.
Initial management of a patient with suspected hemorrhage includes addressing basic airway, breathing, and circulation with 2 large-bore IVs, crystalloid fluids, and cardiac monitoring. Patients with gross lower GI bleeding can decompensate quickly and should have an active type and screen in case transfusion is required.
Imaging is not always necessary, and if possible the decision for imaging should be made along with the GI consult, preferably the physician who performed the colonoscopy. Repeat colonoscopy is often the diagnostic and therapeutic procedure of choice. The American College of Radiology gives the highest appropriateness rating to colonoscopy, though it also includes CTA of the abdomen/pelvis and Tc-99m RBC scan abdomen/pelvis as possible appropriate steps in diagnosis.27
If the patient is unstable and the performing endoscopist is unavailable, the ED physician should consult general interventional radiology (IR), GI, and surgery.
Perforation
Colonoscopic perforation (CP) is one of the most serious and feared complications of colonoscopy. Although rare, CP results in high morbidity and mortality.28-32 Perforation may result from mechanical forces against the bowel wall, barotrauma, or electrocautery and can lead to abscess formation, fistula formation, intra-abdominal sepsis, prolonged hospital stay, and even death.
CP occurs in approximately 0.016% of diagnostic colonoscopies but may be seen in up to 5% of therapeutic colonoscopies.33-35 Depending on what was actually done during the colonoscopy, risk for perforation may be higher or lower than the average, so obtaining the operative report or history from the endoscopist can be helpful in determining risk.
Risk of CP for varied colonoscopic procedures:1,36-37
-Screening colonoscopy: 0.01-0.1%
-Anastomotic stricture dilation: 0-6%
-Crohn disease stricture dilation: 0-18%
-Stent placement: 4%
-Colonic decompression tube placement: 2%
-Colonic endoscopic mucosal resection 0-5%
Aside from the events during the procedure, specific risk factors for CP include advanced age, multiple comorbidities, diverticulosis, obstruction, resection of polyps, polyps that are larger than 1cm in size or those found in the right colon, reduced mobility of the colon, existing weakness in the colonic wall, previous incomplete attempt at colonoscopy, or endoscopist inexperience.20
Patients with CP can present with sudden, worsening abdominal pain, followed by the falsely reassuring complete dissipation of pain as the perforation decompresses the inflamed organ. As GI contents continue to spill into the peritoneum, worsening pain and peritoneal symptoms develop. However, symptoms will vary based on the location and size of perforation, the degree of fecal seepage into the peritoneum, and the patient’s age and comorbidities.
Notably, perforations can also be retro-peritoneal, corresponding to the location of the ascending and descending colon and the hepatic and splenic flexures. Although the most common symptom of a colonic perforation is abdominal pain, elderly patients may have a blunted response. Other symptoms include fever, nausea, vomiting, dyspnea, chest pain, scapular pain, and neck pain. Retroperitoneal perforations in particular may have minimal or atypical symptoms, and vital signs may show tachycardia and fever, but could also be normal.20
Initial management of a suspected perforation includes attention to airway, breathing, circulation with 2 large-bore IVs, crystalloid fluids, and cardiac monitor. Labs including CBC, CMP, lipase, and type and screen obtain should be obtained. Patient should be strictly NPO with empiric IV antibiotics. Stat imaging with CT abdomen/pelvis or abdominal x-ray if unstable should be ordered alongside a stat surgical consult.21
Post-polypectomy syndrome
Post-polypectomy syndrome, also known as post-polypectomy electrocoagulation syndrome or transmural burn syndrome, results from electrocoagulation injury to the bowel wall, creating a transmural burn and focal peritonitis without frank perforation.38 The reported incidence varies from 3 per 100,000 (0.003%) to 1 in 1000 (0.1%). Post-polypectomy syndrome was reported in 6 patients out of 16,318 colonoscopies performed between 1994 and 2002 in a large integrated health system.39 Risk factors for this uncommon complication are unknown.
Patients with post-polypectomy syndrome usually present within 12 hours of colonoscopy with fever, tachycardia, and abdominal pain. However, the onset of symptoms may be delayed by up to 5–7 days after the procedure.40 Patients often have leukocytosis and may exhibit rigidity and tachycardia, all of which mimic colonic perforation.41
However, unlike perforation, which may require emergent surgery, patients with post-polypectomy syndrome are generally managed conservatively with intravenous fluids, pain control, and gradual advancement of diet, with or without antibiotics targeted toward gram-negative and anaerobic pathogens.42 Less severe cases can even be managed in an outpatient setting with oral antibiotics and clear liquids for 1–2 days.42-43
As these patients may be indistinguishable from patients with perforation, any patient suspected of post-polypectomy syndrome warrants labs and imaging. In post-polypectomy syndrome, CT scan will usually exhibit focal thickening of the colonic wall with surrounding fat stranding without any extramural air, though in mild cases CT may be negative.42 Regardless of imaging result, as with perforation, if post-polypectomy syndrome is suspected, the patient should have a surgical consult.
Pearls
- Colonoscopy is a widely-used screening test in patients over 50. Complications can be early or delayed. EM Physicians should be aware of the possible complications and their expected timeline, in order to accurately diagnose and manage any possible presenting complication.
- Complications include hemorrhage, perforation, post-polypectomy syndrome, infection, gas explosion, and pyogenic liver abscess.
- History about patient factors and the procedure itself (underlying conditions, medications, whether polyps were removed, if cautery was used, whether there was immediate bleeding) may guide diagnosis and management in the ED.
- Pyogenic liver abscess may present 1-2 weeks following colonoscopy with right upper quadrant pain and fever.
- Much of the colon is retroperitoneal. Retroperitoneal perforations may cause vague and atypical symptoms, so index of suspicion must be high.
- Post-polypectomy syndrome may mimic perforation and can usually be differentiated by imaging.
References / Further Reading:
- Wells CD, Balan V, Smilack JD. Pyogenic liver abscess after colonoscopy in a patient with ulcerative colitis. Clin Gastroenterol Hepatol. 2005 Dec;3(12):xxiv.
- Whitlock, E.P., Lin, J.S., Liles, E. et al. Screening for colorectal cancer: a targeted, updated systematic review for the U.S. Preventive Services Task Force. Ann Intern Med. 2008; 149: 638–658.
- Warren JL, Klabunde CN, Mariotto AB, Meekins A, Topor M, Brown ML, Ransohoff. Adverse events after outpatient colonoscopy in the Medicare population. Ann Intern Med. 2009;150(12):849.
- Johannsen EC, Sifri CD, Madoff LC. Pyogenic liver abscesses.Infect Dis Clin North Am. 2000;14:547–63.
- Meddings L, Myers RP, Hubbard J, et al. A population-based study of pyogenic liver abscesses in the United States: Incidence, mortality, and temporal trends. Am J Gastroenterol. 2010;105:117–24.
- Kaplan GG, Gregson DB, Laupland KB. Population-based study of the epidemiology of and the risk factors for pyogenic liver abscess. Clin Gastroenterol Hepatol. 2004;2:1032-8.
- Petri A, Höhn J, Hódi Z, Wolfárd A, Balogh A. Pyogenic liver abscess – 20 years’ experience. Comparison of results of treatment in two periods. Langenbecks Arch Surg. 2002;387:27–31.
- Pang TC, Fung T, Samra J, Hugh TJ, Smith RC. Pyogenic liver abscess: An audit of 10 years’ experience. World J Gastroenterol. 2011;17:1622–30.
- Murarka S, Pranav F, Dandavate V. DisseminatedStreptococcus anginosus from sigmoid diverticulitis. J Glob Infect Dis. 2011;3:79–81.
- Branum GD, Tyson GS, Branum MA, Meyers WC. Hepatic abscess. Changes in etiology, diagnosis, and management. Ann Surg. 1990 Dec. 212(6):655-62.
- Gyorffy EJ, Frey CF, Silva J Jr, McGahan J. Pyogenic liver abscess. Diagnostic and therapeutic strategies. Ann Surg. 1987 Dec. 206(6):699-705.
- Bonenfant F, Rousseau É, Farand P. Streptococcus anginosuspyogenic liver abscess following a screening colonoscopy. The Canadian Journal of Infectious Diseases & Medical Microbiology. 2013;24(2):e45-e46.
- Ian G. Harnik. Pyogenic liver abscess presenting after malignant polypectomy.Dig Dis Sci. 2007;52:3524–5.
- Paraskeva KD, Bury RW, Isaacs P.Streptococcus milleri liver abscesses: An unusual complication after colonoscopic removal of an impacted fish bone. Gastrointest Endosc. 2000;51:357–8.
- Low DE, Shoenut JP, Kennedy JK, et al. Prospective assessment of risk of bacteremia with colonoscopy and polypectomy. Dig Dis Sci. 1987;32:1239–43.
- Becker F, Nusko G, Welke J, Hahn EG, Mansmann U. Follow-up after colorectal polypectomy: A benefit-risk analysis of German surveillance recommendations. Int J Colorectal Dis. 2007;22:929–39.
- Macrae FA, Tan KG, Williams CB. Towards safer colonoscopy: A report on the complications of 5000 diagnostic or therapeutic colonoscopies. Gut. 1983;24:376–83.
- Nelson, D.B. Infectious disease complications of GI endoscopy: part II, exogenous infections. Gastrointest Endosc. 2003; 57: 695–711.
- Khashab, M.A., Chithadi, K.V., Acosta, R.D. et al. Antibiotic prophylaxis for GI endoscopy. ASGE Standards of Practice Committee. Gastrointest Endosc. 2015; 81: 81-89.
- Lohsiriwat V. Colonoscopic perforation: Incidence, risk factors, management and outcome. World Journal of Gastroenterology : WJG. 2010;16(4):425-430.
- American College of Radiology ACR Appropriateness Criteria : Acute (Nonlocalized)
- Ko, C.W. and Dominitz, J.A. Complications of colonoscopy: magnitude andmanagement. Gastrointest Endosc Clin N Am. 2010; 20: 659–671.
- Warren JL, Klabunde CN, Mariotto AB, et al. Adverse events after outpatient colonoscopy in the Medicare population. Ann Intern Med 2009;150:849-57.
- Hui, A.J., Wong, R.M., Ching, J.Y. et al. Risk of colonoscopic polypectomy bleeding with anticoagulants and antiplatelet agents: analysis of 1657 cases. Gastrointest Endosc. 2004; 59: 44–48.
- Sawhney, M.S., Salfiti, N., Nelson, D.B. et al. Risk factors for severe delayed postpolypectomy bleeding. Endoscopy. 2008; 40: 115–119
- Yousfi, M., Gostout, C.J., Baron, T.H. et al. Postpolypectomy lower gastrointestinal bleeding: potential role of aspirin. Am J Gastroenterol. 2004; 99: 1785–1789.
- American College of Radiology ACR Appropriateness Criteria : Radiologic Management of Lower Gastrointestinal Tract Bleeding. https://acsearch.acr.org/docs/69457/Narrative/ (Accessed July 16, 2017).
- Lohsiriwat V, Sujarittanakarn S, Akaraviputh T, Lertakyamanee N, Lohsiriwat D, Kachinthorn U. Colonoscopic perforation: A report from World Gastroenterology Organization endoscopy training center in Thailand. World J Gastroenterol. 2008;14:6722–6725.
- Iqbal CW, Cullinane DC, Schiller HJ, Sawyer MD, Zietlow SP, Farley DR. Surgical management and outcomes of 165 colonoscopic perforations from a single institution. Arch Surg. 2008;143:701–706; discussion 706-707.
- Teoh AY, Poon CM, Lee JF, Leong HT, Ng SS, Sung JJ, Lau JY. Outcomes and predictors of mortality and stoma formation in surgical management of colonoscopic perforations: a multicenter review. Arch Surg. 2009;144:9–13.
- Lüning TH, Keemers-Gels ME, Barendregt WB, Tan AC, Rosman C. Colonoscopic perforations: a review of 30,366 patients. Surg Endosc. 2007;21:994–997.
- Mai CM, Wen CC, Wen SH, Hsu KF, Wu CC, Jao SW, Hsiao CW. Iatrogenic colonic perforation by colonoscopy: a fatal complication for patients with a high anesthetic risk. Int J Colorectal Dis. 2010 Apr;25(4):449-54.
- Rathgaber SW, Wick TM. Colonoscopy completion and complication rates in a community gastroenterology practice. Gastrointest Endosc. 2006;64:556–562.
- Damore LJ, Rantis PC, Vernava AM, Longo WE. Colonoscopic perforations. Etiology, diagnosis, and management. Dis Colon Rectum. 1996;39:1308–1314.
- Repici A, Pellicano R, Strangio G, Danese S, Fagoonee S, Malesci A. Endoscopic mucosal resection for early colorectal neoplasia: pathologic basis, procedures, and outcomes. Dis Colon Rectum. 2009;52:1502–1515.
- Chukmaitov A, Bradley CJ, Dahman B, et al. Association of polypectomy techniques, endoscopist volume, and facility type with colonoscopy complications. Gastrointest Endosc. 2013; 77:436.
- Fisher DA, Maple DT, et al. Complications of Colonoscopy. ASGE Standards of Practice Committee Gastrointest Endosc. 2011;74:745. Abdominal Pain and Fever or Suspected Abdominal Abscess; Variant 1: Postoperative patient with fever. https://acsearch.acr.org/docs/69356/Narrative/ (Accessed July 17,2017).
- Hirasawa K, Sato C, Makazu M, et al. Coagulation syndrome: Delayed perforation after colorectal endoscopic treatments. World Journal of Gastrointestinal Endoscopy. 2015;7(12):1055-1061.
- Levin TR, Zhao W, Conell C, Seeff LC, Manninen DL, Shapiro JA, Schulman J. Complications of colonoscopy in an integrated health care delivery system. Ann Intern Med. 2006;145(12):880.
- Kim HW. What is different between postpolypectomy fever and postpolypectomy coagulation syndrome? Clin Endosc. 2014;47(3):205–6.
- Stock C, Ihle P, Sieg A, Schubert I, Hoffmeister M, Brenner H Adverse events requiring hospitalization within 30 days after outpatient screening and nonscreening colonoscopies. Gastrointest Endosc. 2013 Mar;77(3):419-29.
- Sethi A, Song LMWK. Adverse events related to colonic endoscopic mucosal resection and polypectomy. Gastrointest Endosc Clin N Am. 2015;25(1):55–69.
- Benson BC, Myers JJ, Laczek JT. Postpolypectomy electrocoagulation syndrome: A mimicker of colonic perforation. Case Rep Emerg Med. 2013;2013 687931.