Author: Nidhi Sahi (Resident, McGill University, Montreal, Canada), Cynthia Santos, MD (Assistant Professor Emergency Medicine, Medical Toxicology, Rutgers New Jersey Medical School, Newark, NJ) // Editors: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital), and Brit Long, MD (@long_brit, EM Attending Physician, San Antonio, TX)

Case Presentation:
You are working as a military physician on the field when a 34-year-old male, military soldier, had an exposure to an unknown gas/liquid compound. He and his regiment were on a mission involving decontamination of an area with possible weapons of opportunity buried. There was an accident during the mission which resulted in a tear of his PPE which went unnoticed by the soldier. During the accident one of the containers opened and he received a splash of liquid onto his torn PPE suit. He is confused, drooling, diaphoretic, short of breath, and his pupils are large.
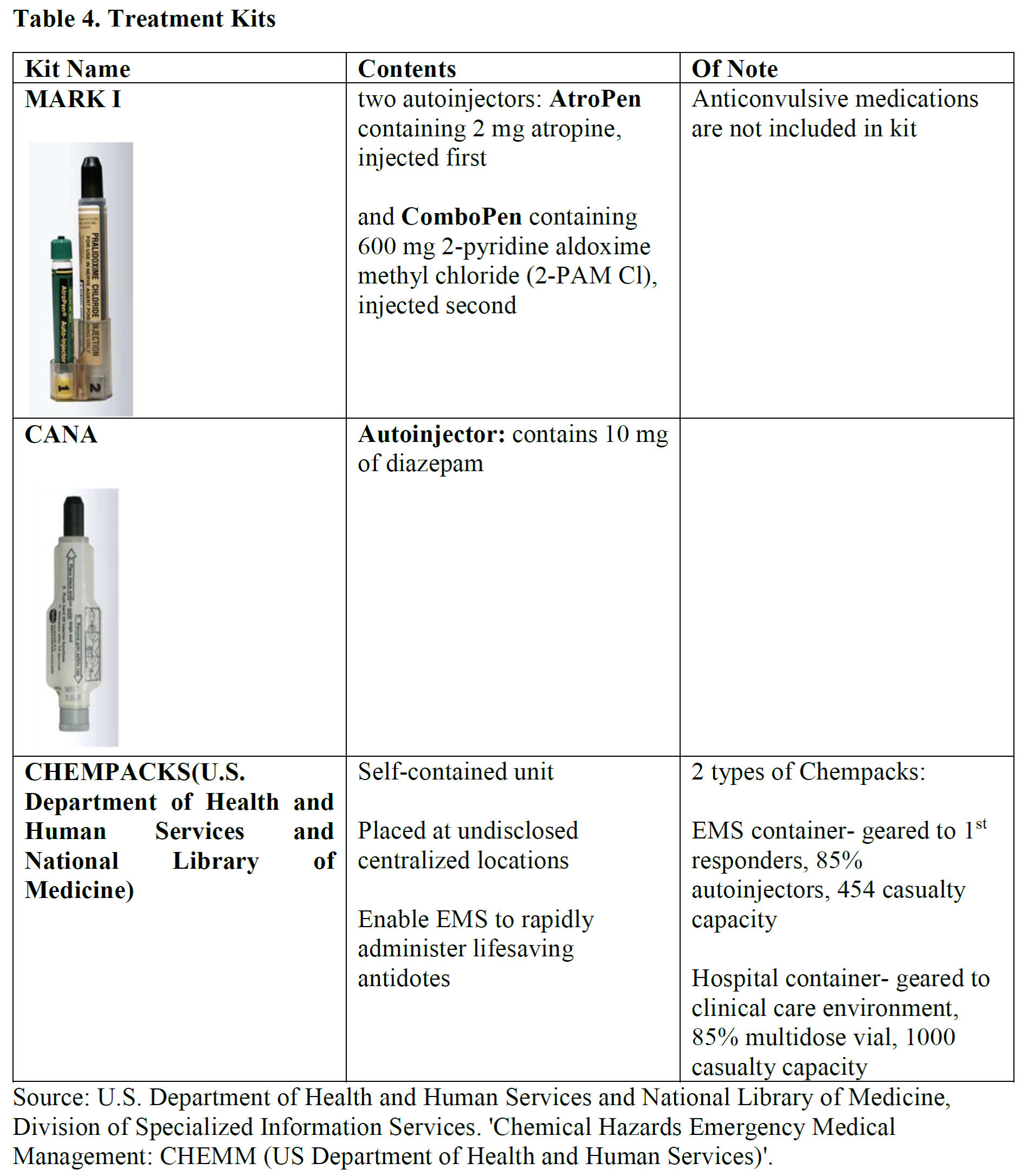
You suspect an organophosphate toxicity and notice the patient is having ventilatory difficulty. You approach the scene with the medics wearing level A PPE. You assist in removing his wet clothing and washing his skin. On primary survey, he is confused and agitated. He has bilateral crackles and wheezing. You give an IM shot using the MARK 1 kit that the soldier had on him. You finish decontaminating the soldier at the scene and you bring him back to the military ED.
Vital signs: HR 50, BP 91/ 56, RR 46, O2 sat 90% on 100% non-rebreather, T 38.1, Glucose 112
Just as you are completing your primary survey in the ED the patient has a seizure. You treat the seizure with lorazepam 2 mg IV boluses. His sergeant arrives to the ED confirming your suspicions for nerve gas exposure. You immediately secure the airway by intubating the patient. Your next step is to begin atropine: you start with 2 mg IV and ask for pralodoxime. In the meantime, you call the poison control center to help guide your management, and you call the military ED pharmacist to enquire about the stock of available antidotes.
Background:
Nerve agents are a class of phosphorous-containing organic chemicals and are considered the most toxic agents of chemical warfare.[1] There are 2 classes of nerve agents: G agents and V agents. G series agents (G= German) were discovered and synthesized by a German team led by Dr. Gerhard Schrader in 1936. G agents are highly volatile and easily aerosolized and thus do not persist in the environment for long. Because of their high volatility, they tend to pose an inhalational and immediate hazard. Health care workers are less at risk from secondary exposure. Examples of G agents include tabun (GA), sarin (GB), soman (GD), and cyclosarin (GF).[2]
An example of a G series agent attack is the 1995 Tokyo subway attack. Five members of Aum Shinrikyo carried liquid sarin in plastic bags wrapped in newspaper. The packets were dropped and punctured, releasing the toxic gas into the subway car. Sarin is a yellow colored, heavy vapor, and due to its high volatility, it quickly evaporates and spreads. Twelve people were killed, 50 others were severely injured, and over 5000 people had temporary vision problems. Sarin gas is also suspected to be the agent used in the Syrian civilian attacks in 2013 which caused the death of nearly 2000 people, and in the April 2017 attack which killed dozens of persons.[3] Sarin is also suspected as the agent responsible for the recent attack in Syria this past April.
In 1952 British chemist Dr. Ranajit Ghosh discovered V series class of nerve agents (V= venomous). V series agents are sticky, oily substances with low volatility. As a result, they can persist on clothing, skin, and in the environment for long periods of time. V agents are significantly more toxic and more lethal than G series agents. Examples of V series agents include VE, VG, VM, VR, and VX (venomous agent X).[2] In 2017, the alleged murder of the half-brother of the North Korean leader is thought to have been due to having VX wiped onto his face.
Mechanism of Action
Nerve agents are similar to organophosphate pesticides in their mechanism of action and symptomology. They phosphorylate and inactivate the enzyme acetylcholinesterase. As a result, with no enzyme to break it down, acetylcholine accumulates in the synapse and continues to exert its downstream effects.[4]
Aging and Aging Time
When the organophosphate initially binds to the acetylcholinesterase, a conformational change occurs in the organophosphate. This change increases the binding and eventually makes it irreversible. This concept is termed aging, and the time required for this change to take place is referred to as aging time. Depending on the type of organophosphate, the time required for the aging process varies between 2 to over 40 hours. [4]

Q1: What are 2 key components of initial management?
Personal Protective Equipment (PPE) – First responders and health care workers must ensure their own protection. Protective suiting and respiratory protection (e.g. SCBA) are key for decontaminating patients with liquid or vapor exposure. Decontamination can only take place inside the hospital if there is a decontamination facility with negative air pressure and floor drains to contain the contamination. Depending on exposure risk further escalation or deescalation in PPE can be considered.[5]
Decontamination – Steps include remove from source, remove contaminated clothing, washing skin with copious amounts of soap and water or dilute bleach, and proper disposal of water runoff and contaminated clothing. Nerve agents are inactivated by alkaline solutions. As an alternative to soap and water, a neutralizing agent such as 0.5% sodium hypochlorite solution (one part household bleach plus nine parts water) can be used.[5] Use of soap and water is often the fastest and a very effective decontamination technique.
Q2: What are common symptoms and signs of nerve agents?
Nerve agent exposure is a generalized whole-body exposure. Similar to organophosphates, symptoms and signs result from a combination of central and peripheral acetylcholine excess. Acetylcholine exerts its effects via nicotinic and muscarinic receptors. In contrast to organophosphates, nerve agents tend to produce more nicotinic than muscarinic effects.
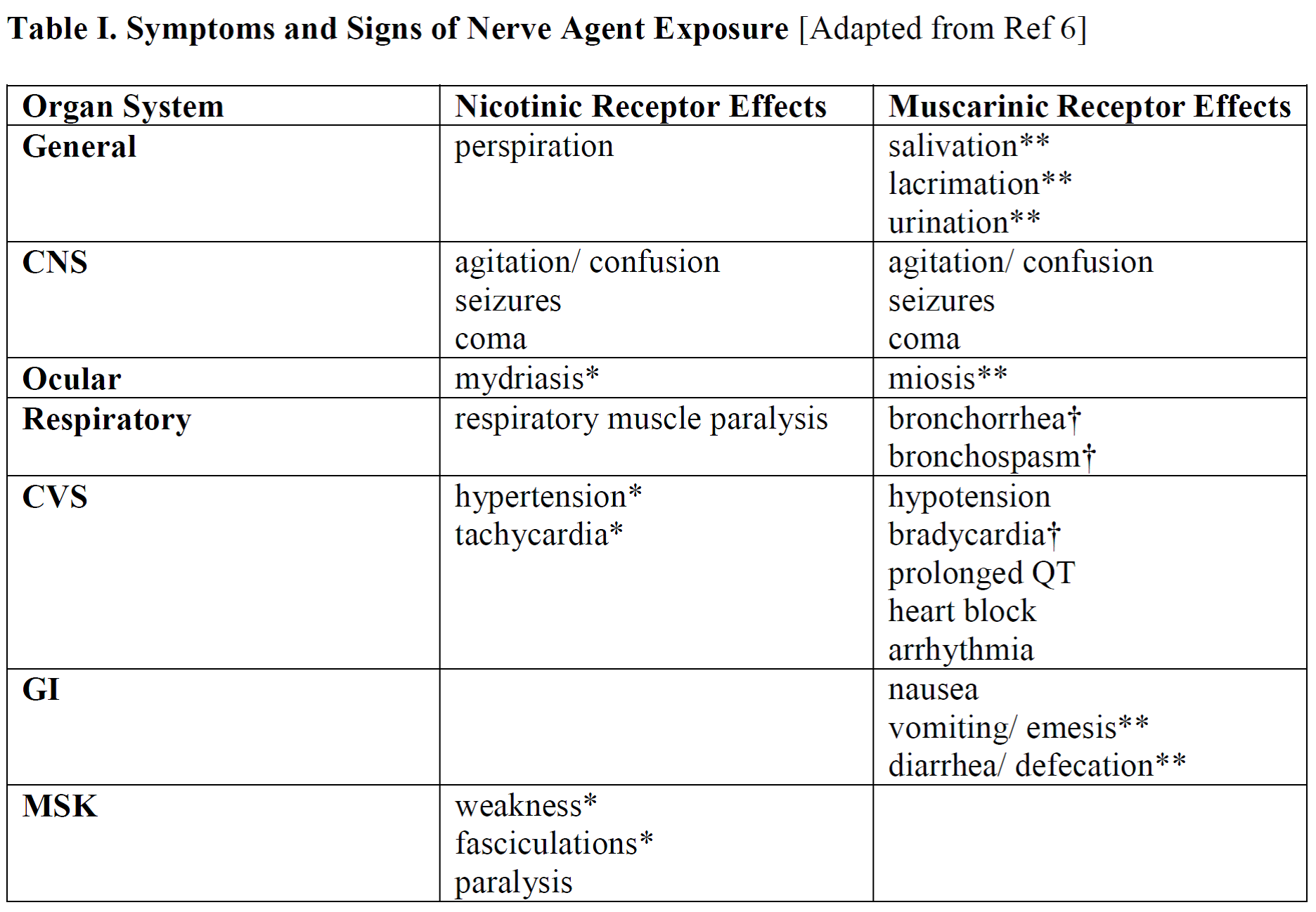
Mnemonics:
*MTWThF – Mydriasis/ Muscle cramps, Tachycardia, Weakness, Twitching, Hypertension/ Hyperglycemia, Fasciculation
**DUMBBELLS – Defecation, Urination, Miosis, Bronchospasm, Bradycardia, Emesis, Lacrimation, Lethargy, Salivation
†3 Killer Bs – Bradycardia, Bronchorrhea, Bronchospasm
Q3: What is the drug management of nerve gases?
In addition to management of ABCs, exposure to nerve gases requires antidotes. There are 3 important drug classes used in acute management of nerve gas exposure.[7] The types and dosing of antidotes used to treat nerve gas exposure is controversial [8]. Of the cited antidotes, atropine is the most important. 2-PAM while cited to reverse the nicotinic effects of the nerve agent is not consistently available in many countries and its clinical significance remains controversial.[9] As per the CDC initial recommended doses for atropine most commonly range from 2-6 mg. Eddleston et al suggest an initial atropine dose of 1-3 mg IV bolus (depending on severity of symptoms) and to reassess the patient every 5 minutes doubling the atropine dose if no improvement in symptoms.[10] An atropine infusion may be required. Continue atropinasation until lung auscultation reveals clear breath sounds. Monitor for signs of excess atropine. Of note, tachycardia itself is not an indication to withhold atropine. [9] The CDC dosing recommendation is outlined in Table 2.


Take Home Points
- Protect yourself and your staff, including PPE for EMS transport of patient.
- Decontaminate.
- Protect the airway and support the circulation.
- Atropine is the mainstay of treatment. Double the dose every 3-5 minutes. Give atropine until secretions are dry.
- Monitor for signs of atropine toxicity
References/Further Reading:
- Eitzen, Edward, J Pavlin, T Cieslak, G Christopher, and R Culpepper. 1998. “Medical management of biological casualties handbook.” In.: Fort Detrick, Frederick, MD: US Army Medical Research Institute of Infectious Diseases.
- Talbot T, Lukey B, Gennady P. Medical Aspects of Chemical Warfare. Borden Institute, United States Army. Available at: http://www.cs.amedd.army.mil/borden/Portlet.aspx?id=d3d11f5a-f2ef-4b4e-b75b-6ba4b64e4fb2
- National Consortium for the Study of Terrorism and Responses to Terrorism (START). (2013). Global Terrorism Database [Data file]. Retrieved from http://www.start.umd.edu/gtd
- Agency for Toxic Substances and Disease Registry. Cholinesterase Inhibitors: Including Insecticides and Chemical Warfare Nerve Agents, Part 4 – Section 11, Management Strategy 3: Medications 2-PAM (2-Pyridine Aldoxime Methylchloride) (Pralidoxime). Available at: https://www.atsdr.cdc.gov/csem/csem.asp?csem=11&po=23
- Agency for Toxic Substances and Disease Registry. Medical Management Guidelines for Nerve Agents: Tabun (GA); Sarin (GB); Soman (GD); and VX. Available at: https://www.atsdr.cdc.gov/toxfaqs/tf.asp?id=523&tid=93
- Candiotti, Keith. 2017. ‘A primer on nerve agents: what the emergency responder, anesthesiologist, and intensivist needs to know’, Canadian Journal of Anesthesia/Journal canadien d’anesthésie: 1-12.
- Abraham, Ron Ben, Valery Rudick, and Avi A Weinbroum. 2002. ‘Practical guidelines for acute care of victims of bioterrorism: conventional injuries and concomitant nerve agent intoxication’, Anesthesiology: The Journal of the American Society of Anesthesiologists, 97: 989-1004.
- U.S. Department of Health and Human Services and National Library of Medicine, Division of Specialized Information Services. ‘Chemical Hazards Emergency Medical Management: CHEMM (US Department of Health and Human Services).







4 thoughts on “TOXCard: Nerve Agents”
Dear EM Docs: I understand that this is a contrived case created for teaching purposes, but I can’t help but to respond to some points you have made.
1. Are we really to believe that a soldier who is out on a military mission where there is likelihood of exposure is: A) not already wearing PPE, B) not decontaminated in the field, and C) not given antidote that he and his team are likely already carrying?
2. Now that we got that out of the way… after you put on your PPE (which should only be level A in this case (note the response photos from the recent UK event) how are you going to get the IV started to give lorazepam? Since you cannot, IM lorazepam or midazolam would be preferred.
3. By the way, can you explain how the sergeant who has transported this patient is not sick and not wearing PPE and yet you have to? The only real world experiences (ie Tokyo and Syria) suggest that exposure risk to responders is very small. That being said I would protect myself from a nerve agent. Experience from places where pesticide poisoning (not military grade) is common also supports very low risk to health care workers and this must be part of your messaging for providers.
4. Let’s move on to decontamination. While you are correct about alkalinization and dilute bleach, time wasted to find and prepare these things make copious water (with or without soap) the fluid of choice (look at real-world Syria experience again)
5. Your atropine dosing regimen in the table is far too inefficient – please see Eddleston et al (PMID: 15533026). More so, given that you already have an IV in place (as per your scenario) it is unclear why you are not recommending IV atropine?? You seem to be in a better place in your take home message but are now internally inconsistent, leaving the readers likely very confused as to what you would like them to do.
5. That same comment can be applied to route for pralidoxime (with appropriate dosing)
I hope you will take time to clarify these points
Thank you Bob for your comments. We made a few changes and clarifications to make the scenario more realistic. The soldier was wearing PPE but because of an accident it got torn so he got exposed. He was decontaminated in the field and given an antidote that he was already carrying. We agree level A PPE should be used in this situation and thus IM meds would have to be used initially. This sergeant was not in the field but met up with you when you arrived in the ED with the patient. We agree that exposure risk to physicians is likely very small but we should still be cautious when approaching patients exposed to military grade nerve agents and don appropriate PPE. As for decontamination copious water would be the practical fluid of choice but we site the CDC reccs that alkalinization and dilute bleach should be used if available. The recommended doses for atropine vary widely. We appreciate the Eddleston dosing method and added it to our post. We still think physicians should be aware of the CDC’s recommended dosing strategy. We agree that IV meds should be used once an IV is able to be placed. Thank you so much for the feedback!
Pingback: Länkar v49-50 | Internmedicin
Pingback: Weekend Knowledge Dump- May 4, 2018 | Active Response Training