Episode 82: HIV Post Exposure Prophylaxis
HIV Background
- Globally, 0.5% of people are infected with HIV and there are 5000 new daily infections.
- The incidence in the U.S. is rising, but global incidence decreasing due to greater availability of ART,prevention programs , use of barrier protection , circumcision for men , and use of pre-exposure prophylaxis (PrEP) .
- The average yearly healthcare cost for one individual with HIV approaches $20,000 (and is closer to $41,000 for those with CD4 counts less than 50).
Risks of HIV seroconversion in the absence of PEP for non-occupational exposures:
- 5-3.38% for receptive anal intercourse
- 06-0.16% for insertive anal intercourse
- 08-0.19% for receptive penile-vaginal intercourse
- 05-0.1% for insertive penile-vaginal intercourse
- Oral intercourse is not typically considered high risk, though genital piercings and the presence of co-existent syphilis increase the risk of transmission.
- The presence of ulcers or trauma to the rectum or vagina or concomitant sexually-transmitted infections (STIs) increases the risk of transmission.
- Injection drug use with a contaminated needle has a seroconversion rate of 7-0.8%.
The risk of HIV seroconversion for occupational exposures is 0.3% for percutaneous injury and 0.09% for mucous membrane exposure.
For percutaneous occupational exposures to HIV-infected blood, the risk of seroconversion is higher if any of the following are present:
- The needle is hollow-bore
- There is visible contamination of blood on the needle
- The needle was inside of the patient’s vasculature
- Untreated HIV in the source patient, especially early- and late-stage
- High HIV viral load in the source patient
- The source patient died of acquired immune deficiency syndrome (AIDS) within two months of the exposure
PEP reduces seroconversion by 89% if given within 72 hours.
Primary indications after a potential exposure
- Divided into occupational versus non-occupational
- PEP unnecessary is someone is compliant with a pre-exposure prophylaxis (PrEP) regimen. You SHOULD give PEP if they haven’t taken their PrEP for 1 week before exposure or they are non-compliant with PrEP on a regular basis.
- PEP should be given within 72 hours of exposure, and ideally within the first 2 hours.
Occupational PEP indications:
- Percutaneous injury (such as a needle or sharp object)
- Exposure of mucous membranes or non-intact skin (cracked, cut, etc.) to one of the following potentially infectious fluids*:
- Amniotic fluid
- Cerebrospinal fluid
- Pericardial fluid
- Peritoneal fluid
- Pleural fluid
- Semen and vaginal secretions
- Synovial fluid
- *note that other body fluids are not considered to carry HIV unless they are visibly contaminated with blood
nPEP indications:

Post-exposure testing
- If you can’t test the source patient, assume they are HIV positive.
- If you can test the source patient, obtain the following:
- HIV; get enough blood for reflex genotype and viral load if the HIV is positive
- Hepatitis B (surface antigen/antibody and core antigen)
- Hepatitis C antibody
- Sexually-transmitted infections (chlamydia, gonorrhea, syphilis) in non-occupational HIV exposure situations
- Obtain testing from your patient for the following (and counsel them that they will need PCP or ID follow up for repeat testing).

Treat nPEP patients for potential concomitant exposures (gonorrhea, chlamydia, hepatitis B, etc.) depending on their situation.
Remember to update tetanus status for oPEP patients with needle stick injuries.
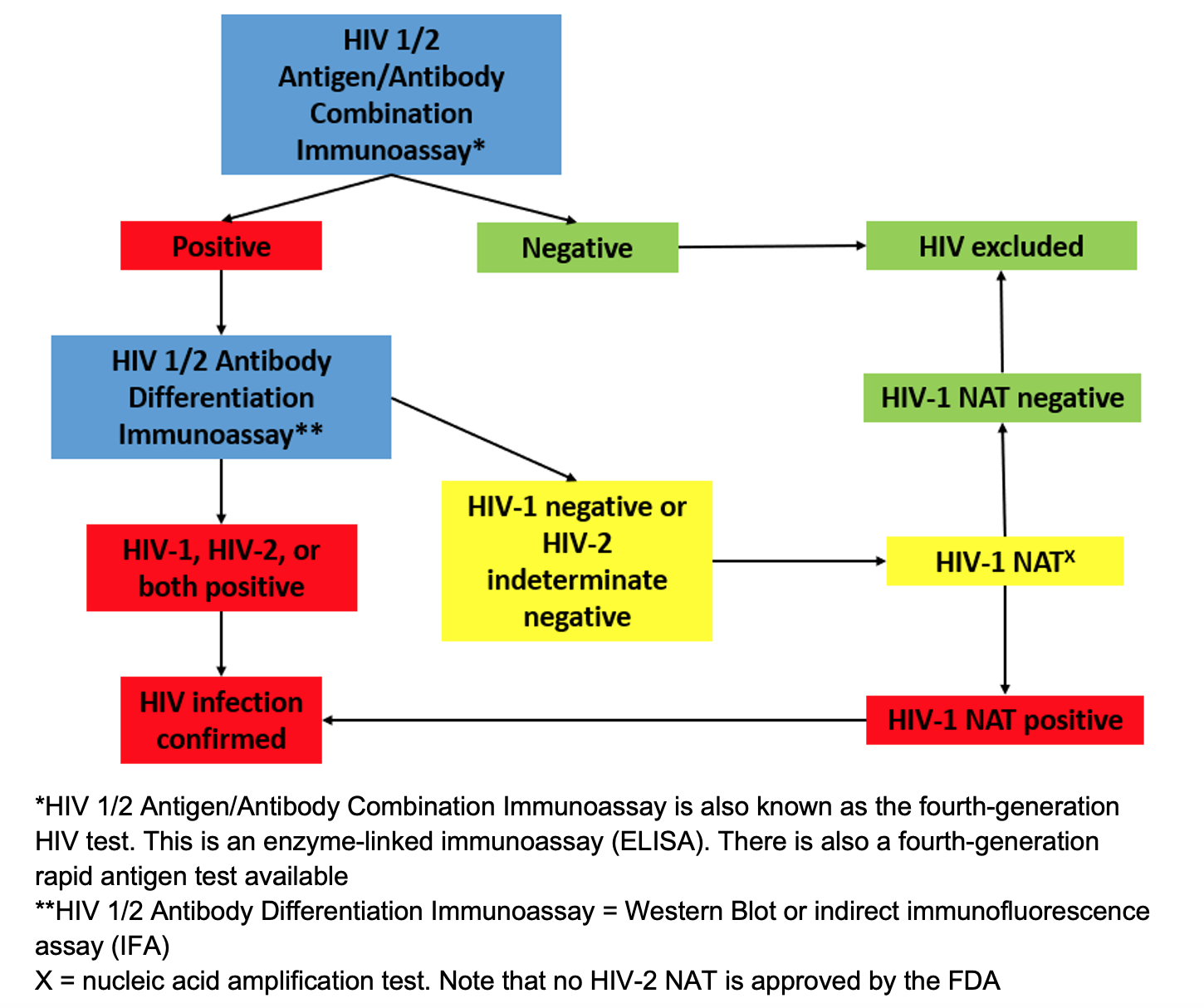
Approach for testing:
Figure 1. Recommended HIV testing algorithm.
Treatment
PEP vs. PrEP
- PrEP usually consists of daily tenofovir/emtricitabine (also known as Truvada).
PEP Dosing and regimens:
- For healthy adults with normal creatinine clearance (CrCl):
- Tenofovir DF 300 mg/emtricitabine 200 mg (Truvada) per os (PO) daily (QD)
- PLUS raltegravir 400 mg PO twice daily (BID) OR dolutegravir 50 mg PO QD
- For CrCl </= 59 ml/min, Truvada should be avoided. The appropriate regimen consists of:
- Zidovudine AND lamivudine with renal dose adjustments
- PLUS raltegravir 400 mg PO BID OR dolutegravir 50 mg PO QD
- For children 2-12 with normal CrCl:
- Tenofovir DF, emtricitabine, and raltegravir, dose-adjusted for age and weight
- For children 28 days-2 years:
- Zidovudine AND lamivudine oral solutions
- PLUS raltegravir OR lopinavir/ritonavir oral solution, dose-adjusted for age and weight
- For children < 28 days: consult infectious disease
- For pregnant women (or women who may become pregnant):
- Tenofovir DF 300 mg/emtricitabine 200 mg (Truvada) PO QD
- PLUS raltegravir 400 mg PO Q12H
- NOTE: Pregnant or breastfeeding women will need to pump and dump until cleared by infectious disease OR should discontinue breastfeeding
There are alternative regimens available for each category of patients, but some of these alternative drugs have more side effects. You can view these alternative regimens in the CDC Clinical Practice Guidelines; we will provide a link in the show notes (https://stacks.cdc.gov/view/cdc/38856, pages 31-32).
Contraindications
- Active HIV infection. PEP administration will breed resistance! Confirm that the patient is negative at baseline before starting PEP
- The following drugs are contraindicated in pregnant women: Didanosine, efavirenz, indinavir (trimesters two and three), nevirapine, and stavudine.

Side Effects
Most side effects are self-limited or mild.
Dolutegravir :
- Abnormal dreams
- Depression
- Diarrhea
- Dizziness
- Fatigue
- Headache
- Nausea
- Insomnia
- Rash
- Vertigo
Emtricitabine :
- Depression
- Diarrhea
- Dizziness
- Elevated creatine kinase and amylase
- Headache
- Hepatic steatosis
- Hypercholesterolemia
- Insomnia
- Nausea
- Rash
Raltegravir :
- Dizziness
- Fatigue
- Headache
- Insomnia
- Nausea
Tenofovir:
- Asthenia/depression
- Headache
- Nausea
- Nephrotoxicity
- Rash
- Transaminitis
Rare and severe side effects include hepatotoxicity from dolutegravir, lactic acidemia from Truvada, and Stevens-Johnson syndrome from raltegravir.
- Patients should be referred to infectious disease and/or the local health department for follow-up testing. Jess:
Addressing barriers to prescribing
Two strategies have been identified to help improve provider comfort with PEP:
- Clear guidelines for first-line PEP drugs, indications, and sexual health support
- Improving providers’ knowledge, practice, and attitudes surrounding PEP
We have addressed both points above in this document and hope this has been helpful! You can call the PEP Hotline anytime for help at 888-448-4911.
ADDITIONAL RESOURCES
http://www.traumayellow.com/uploads/2/0/9/5/20955098/post-exposure_ppx.pdf






