Authors: Kayvan Moussavi, PharmD, BCCCP (Assistant Professor, Marshall B. Ketchum University College of Pharmacy) and Nhu Quyen Dau, PharmD, BCCP (Assistant Professor, Marshall B. Ketchum University College of Pharmacy) // Reviewed by: Skyler Lentz, MD (@skylerlentz); Alex Koyfman, MD (@EMHighAK); and Brit Long, MD (@long_brit)
Case
A 77-year-old, 83 kg male presents to your ED for altered mental status and stroke-like symptoms. GCS is 14, O2 saturation 99%, BP 170/90 mmHg, HR 65. POC glucose is 120 mg/dL. Other labs are drawn but results are pending. After a brief exam, you determine the patient is having stroke-like symptoms but is clinically stable, so your ED team rapidly takes the patient to CT. A few minutes later, the radiologist calls you and states the patient has an intracranial hemorrhage. Lab results are also available and notably include INR 3.9. You quickly review the patient’s chart and find that the patient takes warfarin for atrial fibrillation. Instinctively, you order a nicardipine infusion titrated to SBP 110-139 mmHg, vitamin K 10 mg IV, and place a neurosurgery consult. You would also like to order Kcentra® (the prothrombin complex concentrate [PCC] used at your hospital), but cannot remember the dosing. You call your ED pharmacist who states the dose is 25 units/kg if INR is 2 to <4, 35 units/kg if INR is 4-6, and 50 units/kg for INR >6. You quickly do the math and realize the Kcentra® dose should be 2075 units. You thank her for the info, place the Kcentra® order, and ask her to help retrieve and prepare Kcentra® STAT. “No problem,” she says.
A few hours later, you see the ED pharmacist in the cafeteria. You thank her for helping with the Kcentra® dosing and administration. She asks how that patient is doing. You tell her the patient went to the neuro ICU for monitoring, but appeared to be stable during his time in the ED. She asks you “have you heard of fixed dosing for PCC?” You say, “I heard about it on FOAMed site, but can’t remember much about it.” You ask her to give you a quick summary…
Introduction
Despite the emergence of direct oral anticoagulants (DOAC) in recent years, vitamin K antagonists (VKA) remain popular due to clinician familiarity, superior anticoagulation efficacy for mechanical heart valves, ability to easily monitor efficacy, and availability of reversal agents [1-3]. VKA include warfarin, acenocoumarol, phenprocoumon, dicoumarol, tecarfarin, and fluindione [2]. In the United States, warfarin is the most frequently used VKA and is frequently associated with bleeding complications that require treatment in the emergency department (ED) [3]. Warfarin and other VKA work by inhibiting vitamin K-dependent carboxylation of coagulation factors II, VII, IX, and X [2, 4]. This reduces the activity of these clotting factors and reduces clotting ability [2]. Anticoagulant effects can be measured via the international normalized ratio (INR) and higher INR values reflect reduced ability to form blood clots [2, 3]. Risk of hemorrhage also increases with increasing INR, although patients can develop life-threatening hemorrhages even if INR is within therapeutic range (e.g. 2-3 for atrial fibrillation) [1, 2].
Because warfarin has a half-life that ranges from 20-60 hours, patients with life-threatening hemorrhage require prompt reversal [2]. Vitamin K (phytonadione) provides the substrate required to synthesize coagulation factors inhibited by warfarin and other VKA [1-3]. The main limitation of vitamin K is the delayed onset of action [1-3]. Intravenous doses of 5-10 mg have an onset of 4-6 hours with peak reversal effect at 16-24 hours [1-3]. Due to delayed onset of action, vitamin K must be used in combination with other reversal agents, for example, prothrombin complex concentrates (PCC) [1-3].
PCC refers to medications that contain variable amounts of factors II, VII, IX, X, proteins C, S, and Z, and heparin [1, 2]. Some PCC contain three coagulation factors (II, IX, X) and others contain four factors (II, VII, IX, X) [2]. PCC are dosed based on the factor IX component [2]. PCC have immediate onset, are more concentrated than fresh frozen plasma (FFP), and can be administered rapidly [2]. PCC is preferred to FFP due to more rapid INR correction and lower risk of infection, transfusion-related acute lung injury (TRALI), transfusion-associated circulatory overload (TACO), and pulmonary edema [2]. Although the half-lives of factors in PCC vary from 2 hours to 60 hours, the clinical duration of action ranges from 6-8 hours [2,8]. Because VKA reversal for longer than 6-8 hours is generally preferred, several guidelines recommend that patients with life-threatening bleeding due to VKA receive vitamin K IV plus a PCC [1-3].
Despite the advantages of PCC use for bleeding in patients using VKA, there are several disadvantages. First, dosing can be complicated. For example, Kcentra® (4-factor PCC approved for use in the United States) dosing depends on weight and INR [5]. Specifically, patients with pretreatment INR between 2-<4 should receive 25 units/kg, INR 4-6 should receive 35 units/kg, and INR >6 should receive 50 units/kg; however, doses should be capped at 100 kg, meaning doses should not exceed 2500, 3500, or 5000 units for each INR range, respectively [5]. Second, the dosing recommended on package inserts may be excessive. A recent study found that in patients presenting with INR 2-3.9 that required reversal with PCC, there was no difference in INR reduction for those receiving <30 units/kg and ≥30 units/kg [6]. Theoretically, excessive dosing can increase the risk for thromboembolism. Third, PCC vials contain variable amounts of factor IX. For example, a Kcentra® 500 unit vial can contain 400-620 units and a 1000 unit vial can contain 800-1240 units [5]. Most hospitals are required to bill based on exact units administered, so pharmacies often need to adjust Kcentra® orders to reflect the units that will be administered. This can result in treatment delays, especially if pharmacists are required to contact the ordering provider prior to making these adjustments. Finally, PCC are expensive with prices ranging from $1 to $3 per unit [7, 8]. These prices can result in a significant financial burden for EDs and hospitals. These disadvantages have prompted clinicians to examine alternative dosing strategies that may simplify dosing, limit treatment delays, provide cost savings, and limit the risk of thromboembolism [9].
Fixed PCC Dosing
Fixed PCC dosing has emerged as an alternative to package insert dosing recommendations. Fixed-dosing refers to giving a predetermined PCC dose instead of an individualized dose based on weight and INR [9]. For example, if a patient presented to the ED with an intracranial hemorrhage while on warfarin, a fixed-dose regimen could be Kcentra® 1500 units. This dose would be given regardless of weight or INR. Theoretical advantages of fixed-dosing include decreased treatment delays due to simplified dosing, more appropriate dosing that promotes hemostasis without causing thromboembolism, and cost savings [9]. Fortunately, there have been several recent studies evaluating the efficacy and safety of fixed-dose regimens. We will briefly review these studies to determine if available evidence supports fixed-dosing and if there is an ideal fixed-dose for most clinical scenarios.
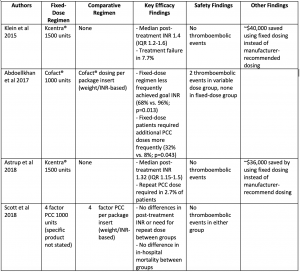
Klein et al 2015 [10]
Study Design/Patients
- Retrospective, single-center review of patients that received fixed-dose PCC
- 39 patients with indications for emergent warfarin reversal
PCC Dosing
- Kcentra® 1500 units
- Comparator: none
Demographics
- Indications (n, %)
- Intracranial hemorrhage: 28 (71.8%)
- Gastrointestinal hemorrhage: 4 (10.3%)
- Ruptured abdominal aortic aneurysm: 1 (2.6%)
- Intrathoracic hemorrhage: 2 (5.1%)
- Spinal cord hemorrhage: 1 (2.6%)
- Neck hematoma: 1 (2.6%)
- Other emergent surgical indication: 2 (5.1%)
- 3% received vitamin K
- Initial INR (median [IQR]): 3.3 (2.5-4)
Efficacy
- Post-treatment INR (median [IQR]): 1.4 (1.2-1.6) (p<0.001)
- Decrease in INR (%, 95% CI): 56.7 (41.7-70)
- Post-treatment INR <2 (n, %): 36 (92.3%)
- Post-treatment INR ≤5 (n, %): 28 (71.8%)
- Mortality (n, %): 9 (23.1%)
- Treatment failures in 3 patients (post-treatment INR ≥2): 1 patient required additional 500 units
Safety
- No thrombotic events within 7 days of treatment
Other Findings
- PCC doses given
- Units/kg (median [IQR]): 20.4 (17.3-22.6)
- Units (median [range]): 1659 (1569-1710)
- Cost savings
- $40,273 saved using fixed dosing over 11 month study period
Abdoellkhan et al 2017 [11]
Study Design/Patients
- Retrospective, single-center, before and after study of fixed vs. variable dosing
- 53 patients with intracranial hemorrhage on VKA (n=28 in fixed dose, n=25 in variable dosing)
PCC Dosing
- Fixed: Cofact® 1000 units
- Note: If INR ≤5 not achieved, another 500 units could be administered
- Variable: Cofact® dosing per package insert (weight and pretreatment INR-based)
Demographics
- No significant differences in baseline characteristics
- Median (IQR) pre-treatment INR: 3.1 (1.8-9) vs. 3.3 (1.7-9) (p=0.175)
- Most frequently used VKA phenprocoumon: 76% vs. 82% (p=0.737)
*Note: above results listed as variable vs. fixed dosing
Efficacy
- Goal INR (≤5) achieved more frequently in variable dosing: 96% vs. 68% (p=0.013)
- Additional PCC doses required less frequently in variable dosing: 8% vs. 32% (p=0.043)
- Post-treatment INR lower in variable dosing (median [min-max]): 1.3 (1-1.9) vs. 1.4 (1.2-2) (p=0.001)
- No rebleeding events in either group
- No differences in mortality, duration of ICU stay, or duration of hospital stay
- Hospital discharge mortality: 12% vs. 22% (p=0.474)
- 30 day after treatment mortality: 16% vs. 25% (p=0.509)
- ICU stay (median [min-max]): 1 (0-17) vs. 2 (0-14) days (p=0.088)
- Hospital stay (median [min-max]): 8 (1-43) vs. 11.5 (1-52) days (p=0.643)
*Note: above results listed as variable vs. fixed dosing
Safety
- No differences in door to PCC administration time (median [min-max]): 81 (33-231) vs. 60 (24-251) minutes (p=0.42)
*Note: above results listed as variable vs. fixed dosing
- 2 patients with thromboembolism in variable dosing (cerebral ischemia)
- No thromboembolic events in fixed dosing
Other Findings
- Total PCC dose higher in variable dosing (median [min-max]): 1750 (1000-2500) vs. 1000 (1000-3000) units (p=0.005)
Astrup et al 2018 [7]
Study Design/Patients
- Retrospective, single-center review of patients that received fixed-dose PCC
- 37 patients with indications for urgent or emergent warfarin reversal
PCC Dosing
- Kcentra® 1500 units
- Note: could repeat dose if clinically indicated
- Comparator: none
Demographics
- Indications (n, %)
- Intracranial hemorrhage: 17 (45.9%)
- Gastrointestinal bleed: 5 (13.5%)
- Other bleed location: 7 (18.9%)
- Urgent surgery: 7 (18.9%)
- Other: 1 (2.7%)
- 1% received vitamin K IV
- Initial INR (median [IQR]): 3.06 (2.17-5.21)
Efficacy
- Post-treatment INR (median [IQR]): 1.32 (1.15-1.5)
- Change in INR post-treatment (median [IQR]): 1.55 (0.91-3.62)
- Second PCC dose require (n, %): 1 (2.7%)
- Mortality (n, %): 9 (24.3%)
Safety
- No thrombotic events ≤72 hours or >72 hours after PCC administration
Other Findings
- PCC doses given
- Units/kg (median [IQR]): 20.1 (18.1-24.4)
- Units (mean [range]): 1601 (1491-1691)
- Cost savings
- $982 saved per patient using fixed vs. package insert dosing (weight and INR-based)
- Total savings $36,365 over 8 month study period
Scott et al 2018 [12]
Study Design/Patients
- Retrospective, single center cohort study
- 61 patients requiring warfarin reversal due to intracranial hemorrhage (n=31 in variable dose, n=30 in fixed dose)
PCC Dosing
- Fixed: 4 factor PCC 1000 units
- Note: product not specified
- Comparator: 4 factor PCC per package insert (weight and pretreatment INR-based)
Demographics
- No significant differences in baseline characteristics
- Aspirin use in 26% of variable dose patients and 17% of fixed dose patients (p=0.38)
- Initial INR (mean [SD]) in variable dose 2.98 (1.25) vs. 2.84 (1.18) in fixed dose (p=0.39)
- 3% received vitamin K in variable dose vs. 93.3% in fixed dose (p=0.67)
Efficacy
- No differences in efficacy-related outcomes
- Reached INR <1.6 (n, %): 25 (81%) vs. 22 (73%) (p=0.49)
- Reached INR <1.5 (n, %): 22 (71%) vs. 16 (53%) (p=0.15)
- Required repeat dose (n, %): 5 (16%) vs. 5 (17%) (p=0.95)
- In-hospital mortality (%): 26% vs. 27% (p=0.93)
*Note: above results listed as variable vs. fixed dosing
Safety
- No thromboembolic events in either group
Other Findings
- PCC dose lower in fixed dose group (mean [SD]): 1045 (750) vs. 2120 (732) units (p<0.001)
Bitonti et al 2019 [13]
Study Design/Patients
- Prospective, multicenter, noninferiority, before and after cohort study
- 54 patients with indications for warfarin reversal (n=30 in variable dose, n=24 in fixed dose)
PCC Dosing
- Fixed: Kcentra®
- Baseline INR ≤7.5 and ≤100 kg: 1500 units
- Baseline INR >7.5 or >100 kg: 2000 units
- Could redose if clinically indicated
- Comparator: Kcentra® dosing per package insert (weight and pretreatment INR-based)
Demographics
- No significant differences in baseline characteristics
- Mean baseline INR 3.83 vs. 4.58 (p=0.34)
- Indications
- Surgery: 3.3% vs. 4.2% (p-value not reported)
- Intracranial hemorrhage: 63.3% vs. 37.5% (p=0.27)
- Gastrointestinal bleed: 26.7% vs. 50% (p=0.27)
- Abdominal bleed: 6.7% vs. 4.2% (p-value not reported)
- Other bleed: 0% vs. 4.2% (p-value not reported)
- Vitamin K given to 100% of variable vs. 95.8% of fixed dosing (p=0.44)
*Note: above results listed as variable vs. fixed dosing
Efficacy
- No differences in post-treatment INR
- Mean INR post-infusion: 1.328 vs. 1.38 (p-value not reported)
- Mean INR 24 hours post-infusion: 1.32 vs. 1.31 (p-value not reported)
- Post-infusion INR <2: 96.7% vs. 95.8% (p=0.0035 for noninferiority)
- Post-infusion INR <1.5: 90% vs. 75% (p>0.45 for noninferiority)
- 7-day mortality: 13.3% vs. 8.3% (p=0.009 for noninferiority)
*Note: above results listed as variable vs. fixed dosing
Safety
- 7-day VTE: 0% vs. 0% (p=0.021 for noninferiority)
- Note: above results listed as variable vs. fixed dosing
Other Findings
- Mean PCC dose: 2591 vs. 1725 units (p=0.001)
- Note: above results listed as variable vs. fixed dosing
Fuh et al 2019 [14]
Study Design/Patients
- Retrospective, single-center cohort study
- 63 patients requiring urgent warfarin reversal
PCC Dosing
- Fixed: Kcentra® 1500 units
- Note: if goal INR not achieved, could give remainder of weight/INR-based dose recommend on package insert
- Comparator: none
Demographics
- Indications (n, %)
- CNS bleed: 48 (76%)
- Other bleed: 9 (14%)
- Urgent procedure: 6 (10%)
- Median initial INR 2.6
- 90% of patients received vitamin K
Efficacy
- Median post-treatment INR: 1.4
- 17/63 (17%) required supplemental PCC dose
- In-hospital mortality (n, %): 19 (30%)
Safety
- Not reported
Other Findings
- $107,729 saved over the study period (~1 year) by using fixed dosing instead of weight/INR-based dosing
Gilbert et al 2019 [15]
Study Design/Patients
- Retrospective, single-center cohort study
- 60 patients requiring urgent warfarin reversal (30 patients per group)
PCC Dosing
- Fixed: Kcentra® 1500 units if intracranial hemorrhage or 1000 units if other bleed type
- Note: patients with pretreatment INR >10 or >100 kg could receive additional 500 units
- Comparator: Kcentra® per package insert (weight and pretreatment INR-based)
Demographics
- No significant differences in baseline characteristics
- Median initial INR (IQR): 3.25 (2.1-30) vs. 2.95 (2.2-30) (p=0.28)
- Indications
- Intracranial hemorrhage: 36.7% vs. 50% (p=0.29)
- Gastrointestinal bleed: 20% vs. 23.3% (p=0.75)
- Emergent surgery: 36.7% vs. 20% (p=0.15)
- Other: 6.7% vs. 6.7% (p=1)
- 90% received vitamin K in variable dose group vs. 96.7% in fixed dose group (p=0.3)
*Note: above results listed as variable vs. fixed dosing
Efficacy
- No differences in post-treatment INR
- INR <1.6: 90% vs. 86.7% (p=0.68)
- INR <1.4: 73.3% vs. 50% (p=0.06)
- Median INR (IQR): 1.3 (1.1-19) vs. 1.35 (0.9-2.1) (p=0.16)
- Hospital length of stay (median [IQR]): 6 (2-41) vs. 6 (3-18) days (p=0.81)
- Mortality: 26.7% vs. 13.3% (p=0.19)
- In patients with intracranial hemorrhage: 45.5% vs. 9.1% (p=0.02)
*Note: above results listed as variable vs. fixed dosing
Safety
- No difference in thrombotic events at 7 days post-infusion: 0% vs. 3.3% (p=1)
- No difference in door to administration time (median [IQR]): 56 (45-135) vs. 48 (24-63) minutes (p=0.11)
*Note: above results listed as variable vs. fixed dosing
Other Findings
- PCC dosing lower in fixed dose group (median [IQR]): 1521 (980-2099) vs. 2551 (1620-5210) units (p=0.004)
Evidence Summary Table

Putting It All Together – What Should We Do With This Data?
Several institutions have transitioned to fixed PCC dosing for VKA reversal, likely for cost savings [7,10,14]. From a clinical perspective, there is evidence to support the use of fixed PCC dosing over manufacturer recommended dosing; however, this data is limited to retrospective studies with heterogeneity in study design [7,9-14]. These studies varied in the indications for reversal, patient population, and PCC dose received [7,9-14]. They also used INR target as a surrogate marker of clinical success [7,9-14] so it remains uncertain if there are differences in patient centered outcomes, either positive or negative, with fixed versus variable dosing. All these studies used 4-factor PCC (e.g. Kcentra®, Cofact®), so it is unclear if 3-factor PCC would provide similar results [7,9-14].
The 2017 ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants recommends that patients requiring immediate VKA reversal receive either manufacturer recommended dosing (i.e. weight and INR-based) or fixed dosing, specifically, 1000 units for major bleeding and 1500 units for intracranial hemorrhage [4]. The authors did not discuss the rationale for this specific recommendation or list the data to support it [4]. We are left to assume that the experts agreed that a fixed dosing regimen of 1000-1500 units may be adequate at the time. Since the publication of this guideline, additional studies have emerged that suggest patients with intracranial hemorrhage and high pretreatment INR may need a higher PCC dose [13]. For an example, Bitonti et al demonstrated that administering an increased dose of 2000 units, instead of 1500 units, if the patient weighed >100 kg or had a baseline INR >7.5 was safe and effective [13]. However, there were also limitations to the study that included small sample size (54 patients) and non-inferiority study design [13]. A randomized control trial comparing various fixed dosing regimens and correlating INR to clinical outcomes would shed light on which fixed dosing regimen is best. Notably, there is currently a randomized, open label trial designed to compare fixed and variable PCC dosing for warfarin reversal in progress (NCT03064035) [16]. The results of this trial are eagerly awaited.
Case Resolution
You thank the ED pharmacist for sharing information about fixed dosing for PCC. Both of you return to the ED to complete your shifts. A few weeks later, you receive an invitation to join a task force at your hospital designed to evaluate available evidence for alternative PCC dosing strategies for VKA reversal. You recall the discussion you had with the ED pharmacist regarding fixed PCC dosing, so you decide to accept the invitation. After a few meetings, your multidisciplinary task force team decides that a fixed dose PCC approach is reasonable for the ED and hospital. Your team decides to recommend the dosing regimen evaluated by Bitonti et al (Kcentra® 1500 units or 2000 units if INR >7.5 or > 100 kg) to your hospital’s P&T committee [13]. After much debate at the P&T meeting, the P&T committee accepts your recommendations and grants approval to update the hospital’s VKA reversal guideline and medication order sets.
Take Home Points
- Patients with urgent or emergent need for VKA reversal require IV vitamin K 5-10 mg and PCC.
- PCC dosing approved by manufacturers for VKA reversal may be complicated, result in excessive administration of clotting factors, and be more expensive than other dosing strategies.
- Fixed-dose PCC refers to giving a predetermined dose (e.g. 1500 units) instead of a variable dose based on weight and pretreatment INR (e.g. 25-50 units/kg for INR 2 to >6).
- There is currently limited evidence to support fixed-dose strategies; however, some studies suggest doses 1500-2000 units can provide similar efficacy and safety when compared to weight/INR-based regimens.
- Fixed doses ≤1000 units may be insufficient for VKA reversal, especially in patients with intracranial hemorrhage.
- If a fixed dose strategy is adopted, consider adding a re-dose option (e.g. if INR goal not achieved, give remainder of weight/INR-based dose per package insert).
References / Further Reading
- January CT et al. 2019 AHA/ACC/HRS Focused Update of the 2014 AHA/ACC/HRS Guideline for the Management of Patients with Atrial Fibrillation. J Am Coll Cardiol 2019;X(XX).
- Frontera JA et al. Guideline for Reversal of Antithrombotics in Intracranial Hemorrhage: A Statement for Healthcare Professionals from the Neurocritical Care Society and Society of Critical Care Medicine. Neurocrit Care 2016;24(1):6-46
- Baugh CW et al. Anticoagulant Reversal Strategies in the Emergency Department Setting: Recommendations of a Multidisciplinary Expert Panel. Ann Emerg Med 2019;X(XX).
- Tomaselli GF et al. 2017 ACC Expert Consensus Decision Pathway on Management of Bleeding in Patients on Oral Anticoagulants: A Report of the American College of Cardiology Task Force on Expert Consensus Decision Pathways. J Am Coll Cardiol 2017;70(24):3042-3067.
- Kcentra (R) [package insert], CSLBehringGmbH, Editor. 2018: Kankakee, IL.
- Yohe AS et al. Four-factor prothrombin complex concentrate dose response relationship with INR for warfarin reversal. Am J Emerg Med 2019;37(8):1534-1538.
- Astrup G et al. Fixed dose 4-factor prothrombin complex concentrate for the emergent reversal of warfarin: a retrospective analysis. J Thromb Thrombolysis 2018;45(2):300-305.
- Lexi-Drugs. Prothrombin Complex Concentrate (Human). 2019, Lexicomp: Hudson, OH.
- Schwebach AA et al. Fixed-Dose Four-Factor Prothrombin Complex Concentrate for Vitamin K Antagonist Reversal: Does One Dose Fit All? Pharmacotherapy 2019;39(5):599-608.
- Klein L et al. Evaluation of fixed dose 4-factor prothrombin complex concentrate for emergent warfarin reversal. Am J Emerg Med 2015;33:1213-1218.
- Abdoellakhan RA et al. Fixed Versus Variable Dosing of Prothrombin Complex Concentrate in Vitamin K Antagonist-Related Intracranial Hemorrhage: A Retrospective Analysis. Neurocrit Care 2017;26(1):64-69.
- Scott R et al. Evaluation of fixed-dose four-factor prothrombin complex concentrate for emergent warfarin reversal in patients with intracranial hemorrhage. J Emerg Med 2018;54(6):861-866.
- Bitonti MT et al. Prospective Evaluation of a Fixed-Dose 4-Factor Prothrombin Complex Concentrate Protocol for Urgent Vitamin K Antagonist Reversal. J Emerg Med 2019;X(XX).
- Fuh L et al. Initiation of a Fixed-Dose Four-Factor Prothrombin Complex Concentrate Protocol. J Thromb Thrombolysis 2019;X(XX).
- Gilbert BW et al. Modified Version of the American College of Cardiology’s Recommendation for Low-Dose Prothrombin Complex Concentrate is Effective for Warfarin Reversal. Am J Emerg Med 2019;X(XX):1-4.
- HealthPartners Institute. Fixed Versus Variable Dosing of 4-factor Prothrombin Complex Concentrate for Emergent Warfarin Reversal (kcentra). 2019[cited January 30, 2020]; Available from: https://clinicaltrials.gov/ct2/show/NCT03064035.







