We always work hard, but we may not have time to read through a bunch of journals. It’s time to learn smarter.
Originally published at JournalFeed, a site that provides daily or weekly literature updates.
Follow Dr. Clay Smith at @spoonfedEM, and sign up for email updates here.
#1: Simple New Test for Difficult Airway
Spoon Feed
An abnormal upper lip bite test, in which the patient’s lower incisors cannot extend to reach the upper lip, increases the probability of a difficult intubation from 10% to >60%.
Why does this matter?
Any physical examination finding that is easy to identify as a potential predictor of a difficult airway is helpful, so the clinician can prepare appropriately with airway adjuncts and extra backup.
Don’t listen to the orthodontist… underbites are good…at least in this situation…
The authors of this article reviewed 62 studies that involved over 33,000 patients primarily in the OR setting. 10% of these patients were deemed difficult to intubate (primarily by either the Cormack-Lehane grading scale or Intubation Difficulty Scale). They looked at factors that best predicted difficult intubation. Here are some of their findings with associated likelihood ratios of difficult intubation. This list was not surprising to me:
- History of difficult intubation (+LR 16-19)
- Snoring (+LR 3.4)
- Overweight or obese (+LR 2.2)
- Short hyomental distance [range of <3-5.5 cm] (+LR 6.4)
- Retrognathia [mandible measuring <9 cm from angle of jaw to tip of chin] (+LR 6.0)
- Wilson score which includes parameters of weight; neck and jaw mobility; mandible length; and characteristics of dentition/bite [range of >2-3] (+LR 9.1)
- Modified Mallampati score of 3 or more (+LR 4.1)
However, here is a new parameter to add to your armamentarium of difficult airway predicting tools. The upper lip bite test:
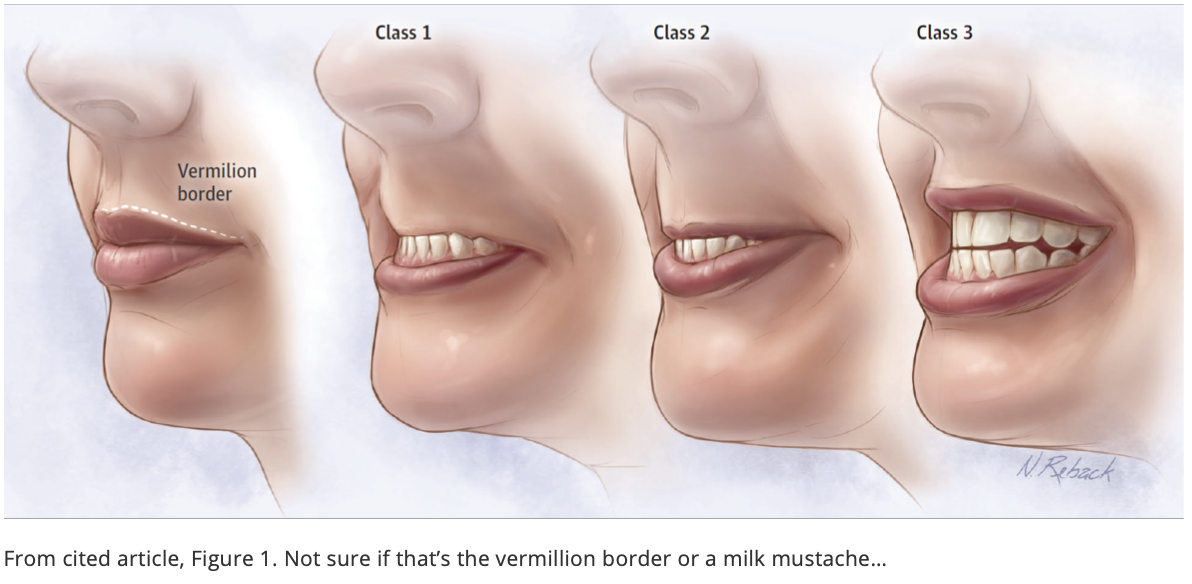
A class 3 upper lip bite test means that the lower incisors cannot extend to reach the upper lip. When that finding is present, the probability of a difficult intubation increases from 10% to >60% (+LR 14). We all know that we often aren’t able to perform long airway assessment scoring systems or measurements in the ED given the acuity of our patients and the emergent nature of our intubations. There will be some acutely decompensating patients or those with altered mental status such that this upper lip bite test may not be feasible. However, it could come in handy for many of our patients, and I will take any new pearl I can find to help me identify a potentially hard airway. So, add this one to your fanny packs of information and combine it with the difficult airway predictors you already knew! An underbite never looked so good!
Source
Will This Patient Be Difficult to Intubate?: The Rational Clinical Examination Systematic Review. JAMA. 2019 Feb 5;321(5):493-503. doi: 10.1001/jama.2018.21413.
Open in Read by QxMD
#2: POCUS Pearls for Tamponade
This study is from our very own Stephen Alerhand!
Spoon Feed
The characteristic findings of pericardial tamponade on bedside ultrasound include:
- Pericardial effusion
- Diastolic right ventricular collapse (high specificity)
- Systolic right atrial collapse (earliest sign)
- Plethoric inferior vena cava with minimal respiratory variation (high sensitivity)
- Exaggerated respiratory cycle changes in mitral and tricuspid valve in-flow velocities as a surrogate for pulsus paradoxus
Why does this matter?
Pericardial tamponade is a potential life-threatening condition that can be difficult to diagnose. Diagnosis is often challenging and is usually based upon history, physical exam, and cardiac imaging. Definitive diagnosis is confirmed with clinical response to pericardial fluid drainage. Point-of-care ultrasound (POCUS) in the Emergency Department (ED) can be an extremely useful adjunct for both diagnosis and treatment of pericardial tamponade.
POCUS Making Waves for Diagnosis of Pericardial Effusion
This was a review that succinctly summarized the echocardiographic findings of pericardial tamponade and potential pearls and pitfalls of POCUS evaluation.
See figure 10 from the article below for a high yield summary and pocket primer for ultrasound findings of pericardial tamponade:

Other key points from the review are listed below:
Pericardial Effusion
- Rate of fluid accumulation is more important than the actual effusion size
- Beware of false positives such as a pleural effusion (posterior to descending aorta on parasternal long axis [PLAX] view) or pericardial fat pad (echoic “stippled appearance” structure in the anterior atrioventricular groove that appears to move in concert with the myocardium)
- Can be seen in PLAX, parasternal short axis (PSAX), apical 4-chamber (A4C), or subxiphoid (SX) views, but best seen in PLAX or SX view
Diastolic Right Ventricular Collapse
- High specificity (75-90%) but low sensitivity (48-60%) for tamponade
- Best seen in PLAX, A4C, or SX views
Systolic Right Atrial Collapse
- Earliest echocardiographic sign of pericardial tamponade because pressure is lower in right atrium than ventricle
- Variable sensitivity and specificity depending on duration of chamber collapse
- The absence of both right atrial systolic and right ventricular diastolic collapse has a 90% negative predictive value for tamponade.
- Best seen in A4C or SX view
Plethoric Inferior Vena Cava
- A dilated, plethoric IVC with minimal respiratory variation (defined as diameter > 2.1 cm and <50% inspiratory reduction) has high sensitivity for tamponade (95-97%) but low specificity (~40%)
Doppler Surrogate of Pulsus Paradoxus
- Tamponade physiology will produce an approximately 25% decrease in mitral valve in-flow velocity and 40% increase in tricuspid valve inflow velocity with inspiration
- Best seen and measured in the A4C view with doppler
Another Spoonful
Do not miss this rap on detecting tamponade with POCUS by The EMC. It is unbelievably good!
This emDocs post from Stephen Alerhand, the author of the study, provides great information on when a pericardial effusion becomes tamponade and POCUS.
Source
What echocardiographic findings suggest a pericardial effusion is causing tamponade? Am J Emerg Med. 2019 Feb;37(2):321-326. doi: 10.1016/j.ajem.2018.11.004. Epub 2018 Nov 17.
Open in Read by QxMD
#3: New Landmark PECARN Rule for Infant Fever
Spoon Feed
Infants ≤60 days are low risk for serious bacterial infection (SBI) if they have a negative urinalysis, ANC ≤4090/µL, and procalcitonin ≤1.71 ng/mL. NPV was 99.6%; negative likelihood ratio (NLR) 0.04. Keep in mind, this hasn’t yet been externally validated.
Why does this matter?
The big issue in febrile infants is LP and hospitalization. Do all these children need a LP? Do they all need antibiotics and hospitalization? Could we define a low risk cohort that could be screened in the ED and discharged with close follow up? More institutions are developing protocols to risk stratify infants with fever, especially those 29-60 days, as we will see tomorrow.
New landmark in risk-stratifying infant fever
The PECARN group derived and internally validated a new infant fever prediction rule in 26 EDs around the US. They included infants ≤60 days who underwent workup for SBI. Overall prevalence of SBI was 9.3% (170/1821); UTI was most common (8.3%), then bacteremia (1.4%), followed by meningitis (0.5%). There were 908 in the derivation set, and it was internally validated in 913. They found that a negative urinalysis, ANC ≤4090/µL, and procalcitonin ≤1.71 ng/mL identified infants as low risk for SBI. Diagnostic accuracy in the validation group was: sensitivity 97.7%, specificity 60%, NPV 99.6%, and NLR 0.04. Clinician pretest suspicion and Yale Observation Score weren’t predictive. The rule missed one patient with bacteremia, two with UTI, and none with meningitis. Only one patient with UTI also had concomitant meningitis.
This study is exciting, but I have some reservations. First, many centers don’t have a rapid procalcitonin assay. Also, keep in mind, sometimes with external validation, the accuracy may change. Finally, we need to be very cautious using risk stratification ≤28 days. For now, I think these children still need a full workup, including LP. My view in infants 29-60 days is evolving, and I now think checking blood, urine, +/- inflammatory markers, +/- multiplex PCR, and not performing LP on every infant is a reasonable approach in well appearing, term infants.
Source
A Clinical Prediction Rule to Identify Febrile Infants 60 Days and Younger at Low Risk for Serious Bacterial Infections. JAMA Pediatr. 2019 Feb 18. doi: 10.1001/jamapediatrics.2018.5501. [Epub ahead of print]
Open in Read by QxMD
#4: Prevalence of UTI With Bronchiolitis
Spoon Feed
When UTI was defined as a positive urine culture plus abnormal UA, the prevalence of concomitant UTI in febrile infants with bronchiolitis was 0.8% – below the usual testing threshold.
Why does this matter?
Studies have shown that febrile infants <3months with bronchiolitis may also have concomitant UTI in 3.3%. But how was “UTI” defined in these studies? Most defined it as having a positive urine culture. However, a positive culture in the absence of an abnormal UA (pyuria or nitrite) may represent asymptomatic bacteriuria or contamination. In 2011, the AAP revised the definition of UTI to, “include both an abnormal UA result (pyuria or bacteriuria) and a positive urine culture (>50000 cfu/mL).” How does this definition affect the prevalence of concomitant UTI with bronchiolitis?
UTI + bronchiolitis = bad luck
This was a systematic review of 18 studies looking at the prevalence of concomitant UTI in infants presenting with bronchiolitis. When looking at all 18, the prevalence was 3.1%. However, when only the seven studies with both positive UA (pyuria or nitrite) and urine culture were included, the prevalence was 0.8%. That’s 125 infants needed to test to find one UTI. The AAP does not recommend testing for UTI unless the pretest probability of disease is 1-3%. UTICalc can help determine pretest probability. When children <90 days were excluded, the prevalence of UA+/culture+ UTI was 0.5%. The study was limited in that the definition of UTI varied across centers, as did the definition of bronchiolitis (some RSV+ only, some clinical). It also included a wide range of ages, from <60 days to 36 months or older. This study changes my practice. Previously, in febrile children with bronchiolitis, I was apt to ignore the bronchiolitis as the source of fever and check a urine anyway. Now, if they clearly have bronchiolitis, I probably won’t check a UA. However, this does not change the fact that children who lack other localizing symptoms for fever still need a urinalysis/urine culture if the pretest probability for UTI exceeds the threshold of ~2%.
Source
Association of Diagnostic Criteria With Urinary Tract Infection Prevalence in Bronchiolitis: A Systematic Review and Meta-analysis. JAMA Pediatr. 2019 Jan 28. doi: 10.1001/jamapediatrics.2018.5091. [Epub ahead of print]
Open in Read by QxMD







2 thoughts on “Journal Feed Weekly Wrap-Up”
Pingback: Förutsäga svår luftväg vid intubation – Mind palace of an ER doc
Pingback: Tamponad – ultraljud – Mind palace of an ER doc