Author: David Cisewski, MD (@PainProfiles – EM Resident Physician, Icahn School of Medicine at Mount Sinai) // Edited by: Manpreet Singh, MD (@MPrizzleER), Alex Koyfman, MD (@EMHighAK), and Brit Long, MD (@long_brit)
ED Migraine Management: The New Kid on the “Block”
“Don’t be good, be GREAT” – Jay Z

Last month, a long-anticipated research trial on ED migraine treatment came out of Bronx, NYC, looking at the use greater occipital nerve blocks (GONB) in the ED setting for the treatment of refractory migraines. That makes this a great opportunity to talk about migraines, what we know migraine treatment, and what nerve blocks bring to the table.
Why was this research so anticipated, you ask?
Over 1 million patients present to the ED each year with migraine-related pain. As Dr. Ben Friedman points out, over 20 different migraine medications are currently being used, and yet less than 25% of patients achieve sustained (>48h) headache relief, indicating that we still fall well short of optimal management [1]. Additionally, many of the analgesic options we currently use contain debilitating side effects such as nausea, dizziness, restlessness, akathisia, and dry mouth. What we need is an analgesic that is quick, efficacious, and with minimal side effects.
Let me just refresh my memory… migraine just means ‘really bad headache’ right?
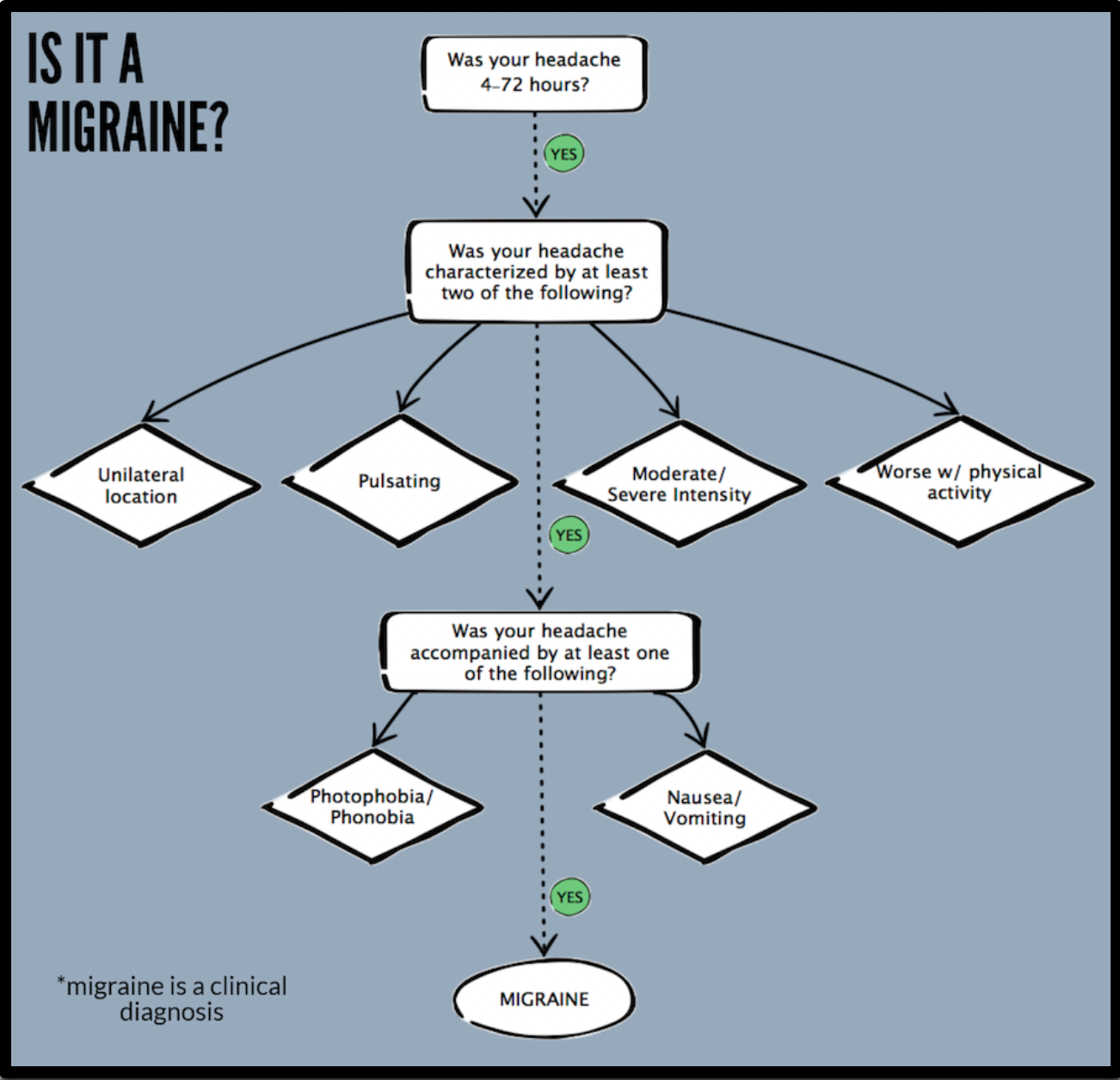
If only it were that simple. ‘Migraine’ is one of the more commonly misused diagnoses used to label patients with headaches in the emergency room. Unfortunately, migraine is a clinical diagnosis without labs or imaging that can confirm the diagnosis. The International Headache Society (IHS) has laid out a very specific diagnostic standard for what defines a migraine:
-
- Last between 4-72 hours
- 2+ of the following:
-
-
- Unilateral location
- Pulsating
- Moderate/severe pain intensity
- Aggravated by physical activity
- 1+ of the following:
- Photophobia/phonophobia
- Nausea/vomiting
-
Seems overkill, why does this matter?
The classification of migraine matters because like all pain presentations, migraines have a unique pathophysiology that may respond differently to certain analgesic regimens. In order to effectively manage pain, we must recognize the individual, subjective nature of each patient’s pain presentation and treat it safely and effectively using the available research data. Additionally, an accurate diagnosis will allow patients to access appropriate care as an outpatient and at future ED visits.
What are the current ED migraine analgesic regimens?
A migraine is a relatively debilitating headache that is typically resistant to oral medications used for mild/moderate pain such as acetaminophen and ibuprofen. Of the 1 million patients presenting to the ED each year for migraine treatment, opioids are still being used in the majority of treatment plans with hydromorphone (Dilaudid) being the analgesic of choice in approximately one-fourth of the cases [2]. However, opioid use for migraines is predominantly based on anecdotal evidence, and recent research by Friedman, et al. has demonstrated non-opioid analgesics (10 mg IV prochlorperazine) have superior analgesic relief [3]. The following is an example of a standardized approach to migraine treatment in the emergency setting (provided by painandpsa.org):
MIGRAINE MANAGEMENT IN THE ED SETTING
Step 1:
- 10 mg IV Metoclopramide ** -or-
- 10mg IV Prochlorperazine ** -or-
- 5 mg IV Haloperidol
- + 10 mg IV Dexamethasone
Step 2:
- Repeat step 1 (wait one hour between successive treatments)
- + 15-30 mg IV ketorolac
Step 3:
- Repeat step 1 (wait one hour between successive treatments)
- + 6 mg SC Sumatriptan
Step 4:
- Bilateral greater occipital nerve block
Step 5:
- Oral opioids (eg, MSIR)
IV fluids have no proven benefit in euvolemic patients and are typically reserved for patients with signs of dehydration [4]. IV dexamethasone has been shown to reduce headache recurrence within 72 hours of ED discharge (NNT = 9) [5]. For more on headache treatment, see this emDocs Cases post.
**Give metoclopramide and prochlorperazine slowly over a 15 min infusion and/or consider 25 mg IV or PO diphenhydramine if using prochlorperazine specifically (we’ve discussed this previously on Pain Profiles).

These are a lot of options… Is there an optimal first step?
While many use metoclopramide as their go-to because of the reduced side effects compared to prochlorperazine, the data are mixed. As pointed out by Michelle Lin, MD at ALiEM, 2 head-to-head comparisons have shown that 10 mg IV prochlorperazine appears to outperform 10 mg IV metoclopramide for acute migraine relief [6, 7].
What’s with the association between migraines and the greater occipital nerve?
Nerve blocks have been used for migraines for years [8, 9]. One of the targeted nerves associated with migraines is the greater occipital nerve (GON). The GON is composed of sensory fibers that derive from the C2 spinal nerve, providing cutaneous innervation to the majority of the posterior scalp [10]. The rationale for the GON block (GONB) for migraines derives from the proximity and convergence of its neurons with the trigeminal nerve fibers which have been implicated in migraine flairs [10, 11]. [Note: This convergence pattern also accounts for the location of pain distribution with migraines, which often includes both anterior and posterior regions of the head/upper neck].
Alright, I’m still with you… so how do you block it?
First you have to locate the nerve. The greater occipital nerve innervates the posterior scalp bilaterally. The following 3-step landmark technique can be used to identify the greater occipital nerve on each side of the scalp [12]:
1 – Place index finger on the occipital protuberance
2 – Place thumb on the mastoid process (either right or left side)
3 – Measure ⅓ the distance between two points extending from the occipital protuberance
This is the approximate location of the greater occipital nerve as it extends up from its exit from the semispinalis capitis muscle at approximately the C1/C2 position (*some indicate you can tell you’re at the GON when you’ve hit the point of maximal tenderness).

Greater Occipital nerve localization. The approximate location of the greater occipital nerve (GON) is one-third the distance between the occipital protuberance and the mastoid process (bilaterally). (*Special thanks to Dr. Ryan O’Halloran(@rto3) for volunteering his professional modeling skills for these visuals).

Okay, I found it…. Now how do I block it?
The procedure involves using 23-gauge needle to infiltrate the GON with a local anesthetic such as 3–5 mL of 1% to 2% lidocaine or 2–4 mL of 0.25% to 0.5% bupivacaine [10]. The optimal technique involves a “fanning technique” in which 1 cc of anesthetic is injected immediately adjacent to the GON, 1 cc medial to the GON, and 1 cc lateral to the GON for maximal infiltration. The “fanning technique” can be repeated bilaterally to cover both GONs.

I like the technique but don’t just conduct pain management based on anecdotal evidence…. what’s the data behind it?
In comes Friedman, et al. with last month’s publication on greater occipital nerve blocks used for migraines refractory to traditional treatment (ie, refractory to metoclopramide).
A Randomized, Sham-Controlled Trial of Bilateral Greater Occipital Nerve Blocks With Bupivacaine for Acute Migraine Patients Refractory to Standard ED Treatment With Metoclopramide
Friedman, 2018 – Headache
This was a randomized, sham-controlled clinical trial of greater occipital nerve blocks (GONB) to determine acute efficacy for migraine following failure of more traditional treatment – in this case, after metoclopramide treatment failure.
Hypothesis: ‘In patients who fail metoclopramide therapy, bilateral GONB would provide greater headache relief than sham intradermal scalp injections’
Who were the patients?
Patients with acute, moderate or severe migraines who failed first-line therapy (metoclopramide) were included in this study. Of the 76 patients screened, only 28 patients met the criteria for enrollment. Exclusion criteria included suspected secondary cause of headache, focal neurologic deficit, head trauma, known bleeding disorder or on anticoagulant, infection over injection site, prior treatment with GONB, pregnant, allergy to treatment medication.
Of the 28 enrollees, patients were predominantly women (80-90%), averaging 35-40 years old, with average headache duration of 2-4 days. The majority had used headache medication prior to arrival, though only 1 of the patients in the study used daily headache or pain medication.
What was the intervention?
Patients in the GONB group received 3 cc bupivacaine 0.5% injected bilaterally (6 cc total) over the greater occipital nerve using the “fanning technique” (see above).
What about the comparison group?
These were the ‘shams’. Patients in the sham group received 0.5 cc bupivacaine 0.5% injected bilaterally (1cc total) intradermally into the posterior scalp overlying the greater occipital nerve.
What is this sham business? Sounds suspicious… don’t you mean placebo?
Placebo does not equal sham. A placebo is similar in every respect to a treatment that has potential or proven efficacy, except that it does not contain the active ingredient or component through which the treatment is assumed to exert its effectiveness (Sedgwick, 2015). In contrast, a sham refers to a faked intervention in which the steps thought to be therapeutically necessary for treatment are omitted. In the case of this study, an injection was made in both study arms and bupivacaine was injected in both groups (ie, not a placebo). The beauty of this study is that the ‘injection effect’ and medication administration were being controlled and what is being studied is whether bathing the greater occipital nerve using the ‘fanning technique’ offered superior analgesic relief vs intradermal injection.
And what did the study find?
In the wise words of Tom Waits, “Sham pain for my real friends, real pain for my sham friends”. The bilateral GONB resulted in significant pain improvements versus the higher pain outcomes seen among the sham arm of the trial.
Primary outcome of complete headache relief at 30 minutes was seen in 0% of the shams and 31% of the GONB grouping (p=0.035). Secondary outcome of sustained headache relief (defined as mild/no headache at 1h and at 48 hour follow up) was 0% in the sham group and only 23% in the GONB group (p=.087). Overall, approximately 4/10 participants in each group said they would not want to receive this treatment again.
How was this primary outcome chosen?
As stated by Friedman, complete headache relief was chosen (versus improvement) to minimize the influence of placebo (‘injection effect’) on the primary outcome. Additionally, 30 minutes was chosen as the timeframe because the researchers felt it unlikely that a patient would progress from moderate or severe migraine to complete headache freedom within 30 minutes spontaneously and without intervention.
Why was the study stopped?
Very slow enrollment. Ultimately the data monitoring committee recommended stopping the study when it was realized the full sample size would not be achieved in a timely enough manner to be relevant.
What’s the verdict?
GONB resulted in greater short-term (@30 min) headache relief compared to sham, and possibly sustained headache relief compared to sham though underpowered to detect this difference. Nevertheless, a significant number of patients continued to have migraine pain at 48 hour follow up despite the best efforts in the ED.
What are the limitations of this study?
You can’t blind an intervention technique. Because the injection technique varied depending on assignment, the clinician performing the procedure could not be blinded. Small sample sizes. The research team wanted 78 patients in total but due to slow enrollment, only 28 patients were enrolled over 3 years – “the data monitoring committee recommended halting the study because the full sample size would not be achieved in a timely enough manner to be relevant”. Uncertain generalizability of the results. This was a study dominated by young and middle-aged women with low-baseline functional impairment. Could a 60-year-old male with a severe migraine benefit from this intervention? This is uncertain.
The Upshot: In the midst of the ‘opioid epidemic’ and the ever-evolving search for non-opioid analgesic alternatives in the ED, nerve blocks are all the rage. Though GONB have been used for years by our neurology colleagues, to our knowledge, this is the first RCT comparing analgesic efficacy of GONB for migraine relief in the ED setting. Further research is needed to support this small study but the results are promising and the technique is simple to learn. Many will fairly argue whether medication is even needed and if saline or dry needling alone would result in migraine improvement. But until research proves otherwise, consider a bilateral greater occipital nerve block as a feasible next step the next time you’ve depleted your analgesic armamentarium on that challenging migraneur that just can’t seem to break.
Summary Infographic:

Further Reading:
- emDocs Cases on ED headache management
- ALiEM w/ Michelle Lin, David Vinson, et al – Prochlorperazine, Metoclopramide, and Diphenhydramine for Acute Migraine Headache
- Ben Friedman, Annals of Emergency Medicine – Managing Migraine
- Ben Friedman, MD on headache management from painandpsa.org Emergency Department Management of Acute Headache
References:
- Friedman, B.W., et al., Recurrence of primary headache disorders after emergency department discharge: frequency and predictors of poor pain and functional outcomes. Ann Emerg Med, 2008. 52(6): p. 696-704.
- Friedman, B.W., et al., Current management of migraine in US emergency departments: an analysis of the National Hospital Ambulatory Medical Care Survey. Cephalalgia, 2015. 35(4): p. 301-9.
- Friedman, B.W., et al., Randomized study of IV prochlorperazine plus diphenhydramine vs IV hydromorphone for migraine. Neurology, 2017. 89(20): p. 2075-2082.
- Friedman, B.W., Managing Migraine. Ann Emerg Med, 2017. 69(2): p. 202-207.
- Colman, I., et al., Parenteral dexamethasone for acute severe migraine headache: meta-analysis of randomised controlled trials for preventing recurrence. BMJ, 2008. 336(7657): p. 1359-61.
- Jones, J., S. Pack, and E. Chun, Intramuscular prochlorperazine versus metoclopramide as single-agent therapy for the treatment of acute migraine headache. Am J Emerg Med, 1996. 14(3): p. 262-4.
- Coppola, M., D.M. Yealy, and R.A. Leibold, Randomized, placebo-controlled evaluation of prochlorperazine versus metoclopramide for emergency department treatment of migraine headache. Ann Emerg Med, 1995. 26(5): p. 541-6.
- Gawel, M.J. and P.J. Rothbart, Occipital nerve block in the management of headache and cervical pain. Cephalalgia, 1992. 12(1): p. 9-13.
- Caputi, C.A. and V. Firetto, Therapeutic blockade of greater occipital and supraorbital nerves in migraine patients. Headache, 1997. 37(3): p. 174-9.
- Ashkenazi, A. and M. Levin, Greater occipital nerve block for migraine and other headaches: is it useful? Curr Pain Headache Rep, 2007. 11(3): p. 231-5.
- Bartsch, T. and P.J. Goadsby, Stimulation of the greater occipital nerve induces increased central excitability of dural afferent input. Brain, 2002. 125(Pt 7): p. 1496-509.
- Friedman, B.W., et al., A Randomized, Sham-Controlled Trial of Bilateral Greater Occipital Nerve Blocks With Bupivacaine for Acute Migraine Patients Refractory to Standard Emergency Department Treatment With Metoclopramide. Headache, 2018.






