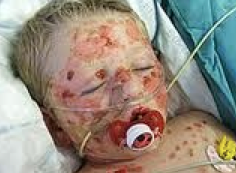
Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis in Pediatric Population
By Stephen Alerhand, MD
EM Resident Physician
Icahn School of Medicine at Mount Sinai
Edited by Alex Koyfman, MD
Definition
- Widespread epidermal necrosis and detachment, with mucocutaneous complications in ~90% 1
| Condition | % Body surface area showing skin detachment |
| SJS | <10% |
| SJS/TEN overlap | 10-30% |
| TEN | >30% |
- Variants of same disease along spectrum
Incidence 2-5
- 2-7 cases per million people per year
- SJS: 1-7 cases
- TEN: 0.4-1.5 cases
- M=F in children 6
Etiology
- TEN triggered by medications or URI in 74%-94% of cases 7-9
- Medications 10-12
- Sulfa Abx, Phenobarbital, Lamotrigine, Carbamazepine, NSAIDS, Allopurinol
- Within first 8 weeks of starting medication
- Greater risk with higher doses 13 and rapid introduction 12, 14
- Symptoms not clearly attributed to a drug by ALDEN (algorithm of drug causality for EN) in 20-25% of cases (higher for children) 15
- Potentially offending drug scored by 6 parameters:
- Time delay from drug administration to reaction onset
- Probability of drug presence in body
- Prior history of exposure to same drug, regardless of reaction
- Presence of drug beyond progression phase
- Drug notoriety as cause of SJS/TEN
- Presence or absence of other etiologies
- Infection 16-21
- Mycoplasma pneumoniae, Cytomegalovirus infections, Herpesvirus, Hep A
- More common for children than medications 22
- Predisposing conditions 23-29
- HIV (100-fold higher risk), malignancy, SLE, radiotherapy, collagen vascular disease, UV light, genetic, underlying immunologic disease
- Potentially offending drug scored by 6 parameters:
Clinical course
- Fever, flu-like symptoms 1-3 weeks after starting drug
- Malaise, myalgia, arthralgia, dysphagia, photophobia, conjunctival itching/burning
- Skin and mucosal involvement 1-3 days later
- Macules with purpuric centers a large blisters, vesicles, bullae a epidermal detachment with Nikolsky’s sign 3-5 days later a large denuded areas
- Start on face and thorax, then spread symmetrically
- Scalp, palms, soles rarely involved
- Tender to touch, skin pain out of proportion to findings
- Start on face and thorax, then spread symmetrically
- Re-epithelization begins 1 week after onset, takes up to 3 weeks; faster in children 30
- Macules with purpuric centers a large blisters, vesicles, bullae a epidermal detachment with Nikolsky’s sign 3-5 days later a large denuded areas
Mucosal and other complications
- Large wound area leads to severe pain, massive fluid and protein loss, electrolyte imbalances, bleeding, evaporative heat loss with subsequent hypothermia, insulin resistance, hypercatabolic state, infection and bacteremia, hypovolemic shock with renal failure, multiple organ dysfunction
- High risk for bacterial infection
- Main cause of death is sepsis, most often by Staph aureus and Pseudomonas aeroginosa
- Greater with BSA >30% and WBC >10 31
- Ophthalmologic
- Up to 30% of surviving children 32
- Acute stage within 2-6 weeks of drug onset
- Most commonly severe conjunctivitis with purulent discharge; swollen and erythematous eyelids, suppurative keratitis, endophthalmitis, pain and photophobia
- Most severe: long-term vision loss from chronic corneal inflammation
- Pulmonary
- PNA, interstitial pneumonitis
- Risk of ARDS
- Mechanical ventilation in 25% 33, associated with higher mortality 34
- GI
- Diarrhea, melena, small bowel ulcerations, colonic perforation, small bowel intussusception, stenosis, strictures, stomatitis
- Skin
- Long-term hyper-/hypopigmentation in virtually all children tends to fade partially fade with time
- Hypertrophic changes, scarring
- Urogenital
- Urethritis
Diagnosis
- Confirmation by skin biopsy 35-36
- Apoptosis, necrosis, and vacuolization of keratinocytes
- Dermo-epidermal detachment
- Lymphocytic infiltration of perivascular regions
- Patch testing, pin prick, intradermal injection lack sensitivity for predicting SJS/TEN 37
- Lab abnormalities
- Anemia, lymphopenia
- Neutropenia in 1/3 of cases, with poor prognosis 7
- Hypoalbuminemia, electrolyte imbalance, increased BUN, increased glucose
Management
- Withdrawal of drug
- Reduces mortality and improves prognosis
- In 10-year observational study of 113 patients, there was better prognosis (outcome measure: no death before hospital discharge) by odds ratio 0.69 (about 30%) for each day before blister development that drug was withdrawn. 38
- In retrospective study of 19 patients, 21% mortality vs. the predicted mortalities by APACHE II (22%) and SCORTEN (30%) 39
- Drugs with long half-lives had increased risk of death (odds ratio 4.9), independent of drug withdrawal 38
- Reduces mortality and improves prognosis
- Supportive care
- Wound care 40
- Debride wounds, moisture-retentive ointments and/or topical Abx to improve barrier function and prevent bacterial infection, non-adherent monocrystalline gauze materials containing silver, whirlpool therapy, anti-shear wound care, fluidized air beds, copper sulfate
- Fluid and electrolyte management
- Increased water loss from denuded dermis
- Volume loss ~1/3 less than that for burn victims 41
- Nutritional support
- Early oral feeding 7
- High-calorie requirements similar to those in burn injury 42-43
- Temperature management
- Temp increased up to 30-32 deg Celsius to prevent excessive caloric expenditures from epidermal loss 1
- Ocular care
- Early baseline Ophtho consult
- Cleaning of eye lids, daily lubrication
- Daily Erythromycin drops to prevent infection, corticosteroid drops to reduce inflammation 44
- Pain control
- Pulmonary toilet
- Monitoring and treatment of super-infections
- Sterile handling, antiseptic solutions
- Repeated cultures q48h of skin, blood, catheters, gastric, urinary tubes
- Signs of infection: visible changes in skin lesions, increase in quantity of cultured bacteria, sudden decrease in temp, general deterioration
- No prophylactic abx; can cause emergence of resistant bacteria and negatively impact survival 45
- Wound care 40
Therapies
- No established therapies
- No randomized prospective trials due to rarity of condition and ethical considerations; few studies and minimal standardization to this point
- Conflicting data in children 46, 48
- IVIg 47, 49-52
- Corticosteroids 53-59
Prognosis/Triage
- SCORTEN (severity-of-illness score) 60
- Validated for adults and children combined on days 1 and 3 of hospitalization 61-62
- Not yet validated for children specifically
- Validated for adults and children combined on days 1 and 3 of hospitalization 61-62
Kaplan-Meier Analysis of survival of patients with TEN based upon SCORTEN score, calculated within 24 hours of hospital admission.
Adapted with permission from: Guegan, S, Bastuji-Garin, S, Poszepczynska-Guigne, E, et al. J Invest Dermatol 2006, 126:272.
- Transfer to non-specialized wards: 63
- Limited skin involvement, SCORTEN score 0-1, non-rapidly progressive disease
- Transfer to ICU or burn unit: 63
- More severe disease, SCORTEN >= 2
- Retrospective review of 199 patients showed mortality of 32% with early ICU/burn unit transfer vs 51% with transfer after 1 week 64
- Longer hospital stay if transferred to burn unit after 1 week 66
- Negative prognostic factors: hypernatremia 67, increased BUN, neutropenia, thrombocytopenia, visceral involvement, delayed presentation 8, 45, 68
Mortality (from large RegiSCAR study) 69
- SJS: 10%
- TEN: >30%
- 23% mortality at 6 weeks, 34% at 1 year
- Slightly lower mortality when a drug was identified as the cause
- Major prognostic factor for death within 90 days only: disease severity
- Beyond 90 days: serious comorbidities and age
- Lower for children
- Major causes of death: sepsis, ARDS, multiple organ failure
Recurrence
- May occur with drug re-exposure
- In a retrospective study of 55 cases, 10 children had recurrence between 2 months and 7 years after first episode, 3 had multiple recurrences, and 1 died 70
- Recurrence of such rare event suggests long-lasting vulnerability and genetic predisposition
ED Pearls
- Be on guard for alerting signs:
- Skin tenderness, blistering, mucositis, and fever >100.4
- If such early signs even minimally present, find out whether “notorious” drugs were initiated within past 2 months:
- Sulfa Abx, Phenobarbital, Lamotrigine, Carbamazepine, NSAIDS, Allopurinol
- Withdraw all non-essential medications immediately
- Strongly consider transfer to burn unit or ICU, or at very least make a consult
- Early Ophtho consult to prevent common and long-lasting sequelae
- Watch for super-infection: visible changes in skin lesions, increase in quantity of bacteria cultured, sudden decrease in temp, general deterioration
- Recurrence of such a rare condition suggests future risk
- Educate about future avoidance, allergy passport
References
- Letko E, Papaliodis DN, Papliodis GN, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: a review of the literature. Ann Allergy Asthma Immunol 2005; 94:419.
- Rzany B, Mockenhaupt M, Baur S, et al. Epidemiology of erythema exsudativum multiforme majus, Stevens-Johnson syndrome, and toxic epidermal necrolysis in Germany (1990-1992): structure and results of a population-based registry. J Clin Epidemiol 1996; 49:769.
- Strom BL, Carson JL, Halpem AC, et al. Using a claims database to investigate drug-induced Stevens-Johnson syndrome. Stat Med 1991; 10:565.
- Chan HL, Stern RS, Amdt KA, et al. The incidence of erythema multiforme, Stevens-Johnson syndrome, and toxic epidermal necrolysis. A population-based study with particular reference to reactions caused by drugs among outpatients. Arch Dermatol 1990; 126:43.
- Schopf E, Stuhmer A, Rzany B, et al. Toxic epidermal necrolysis and Stevens-Johnson syndrome. An epidemiologic study from West Germany. Arch Dermatol 1991; 127:839.
- Prendiville JS, Hebert AA, Greenwald MJ, et al: Management of Stevens-Johnson syndrome and toxic epidermal necrolysis in children. J Pediatr 1989; 115:881-887.
- Roujeau JC, Chosidow O, Saiag P, et al. Toxic epidermal necrolysis (Lyell syndrome). J Am Acad Dermatol 1990; 23: 1039-1058.
- Roujeau JC, Stern RS: Severe adverse cutaneous reactions to drugs. New Eng J Med 1994; 331:1272-1285.
- Spies M, Sanford AP, Aili Low JF, et al. Treatment of extensive toxic epidermal necrolysis in children. Pediatrics 2001; 108:1162-1168.
- Levi N, Bastujo-Garin S, Mockenhaupt M, et al. Medications as risk factors of Stevens-Johnson syndrome and toxic epidermal necrolysis in children: A pooled analysis. Pediatrics 2009; 123:3297-e304.
- Mockenhaupt M, Viboud C, Durant A, et al. Stevens-Johnson syndrome and toxic epidermal necrolysis: assessment of medication risks with emphasis on recently marketed drugs. The Euro-SCAR-study. J Invest Dermatol 2008; 128:35.
- Halevy S, Ghislain PD, Mockenhaupt M, et al. Allopurinol is the most common cause of Stevens-Johnson syndrome and toxic epidermal necrolysis in Europe and Israel. J Am Acad Dermatol 2008; 58:25.
- Famularo G, De Simone C, Minisola G. Stevens-Johnson syndrome associated with single high dose of lamotrigine in a patient taking valproate. Dermatol Online J. 2005; 11:25.
- Rzany B, Correia O, Kelly JP, et al. Risk of Stevens-Johnson syndrome and toxic epidermal necrolysis during first weeks of antiepileptic therapy: a case-control study. Study Group of the International Case Control Study on Severe Cutaneous Adverse Reactions. Lancet 1999; 353:2190.
- Sassolas B, Haddad C, Mockenhaupt M, et al. ALDEN, an algorithm for assessment of drug causality in Stevens-Johnson Syndrome and toxic epidermal necrolysis: comparison with case-control analysis. Clin Pharmacol Ther 2010; 88:60.
- Wetter DA, Camilleri MJ. Clinical, etiologic, and histopathologic features of Stevens-Johnson syndrome during an 8-tear period at Mayo Clinic. Mayo Clinic Proc 2010; 85:131.
- Ferandiz-Pulido C, Garcia-Patos V. A review of causes of Stevens-Johnson syndrome and toxic epidermal necrolysis in children. Arch Dis Child 2013; 98:998.
- Fournier S, Bastuji-Garin S, Mentec H, et al. Toxic epidermal necrolysis associated with Mycoplasma pneumoniae infection. Eur J Clin Microbiol Infect Dis 1995; 14:558-559.
- Meseguer MA, de Rafael L, Vidal ML. Stevens-Johnson syndrome with isolation of Mycoplasma pneumoniae from skin lesions. Eur J Clin Microbiol 1986; 5:167-168.
- Stutman HR. Stevens-Johnson syndrome and Mycoplasma pneumoniae: Evidence for cutaneous infection. J Pediatr 1987; 111:845-847.
- Werblowsky-Constantini N, Livshin R, Burstein M, et al. Toxic epidermal necrolysis associated with acute cholestatic viral hepatitis A. J Clin Gastroenterol 1989; 11:691-693.
- Leaute-Labreze C, Lamireau T, Chawski D, Maleville J, Taieb A. Diagnosis, classification, and management of erythema multiforme and Stevens-Johnson syndrome. Arch Dis Child 2000;83:347-52.
- Mittmann N, Knowles SR, Koo M, et al. Incidence of toxic epidermal necrolysis and Stevens-Johnson syndrome in an HIV cohort: an observational, retrospective case series study. Am J Clin Dermatol 2012; 13:49.
- Gravante G, Delogu D, Marianetti M, et al. Toxic epidermal necrolysis and Stevens-Johnson syndrome in oncologic patients. Eur Rev Med Pharmacol Sci 2007; 11:269.
- Rosen AC, Balagula Y, Raisch DW, et al. Life-threatening dermatologic adverse events in oncology. Anticancer Drugs 2014; 25:225.
- Horne NS, Narayan AR, Young RM, Frieri M. Toxic epidermal necrolysis in systemic lupus erythematosus. Autoimmune Rev 2006; 5:160.
- Ruggiero A, Buonuomo PS, Maurizi P, et al: Stevens-Johnson syndrome in children receiving Phenobarbital therapy and cranial radiotherapy. J Neurooncol 2007; 85:213-215.
- Maiche A, Teerenhovi L. Stevens-Johnson syndrome in patients receiving radiation therapy. Lancet 1985; 2:45.
- Duncan KO, Tigelaar RE, Bolognia JL. Stevens-Johnson syndrome limited to multiple sites of radiation therapy in a patient receiving Phenobarbital. J AM Acad Dermatol 1999; 40:493.
- Knowles SR, Shapiro LE, Shear NH. Drug Eruptions. In: Schachner LA, Hansen RC, editors. Pediatric Dermatology. 3rd Edition ed. London, UK: Mosby; 2003; 1267-76.
- de Prost N, Ingen-Housz-Oro S, Duong Ta, et al. Bacteremia in Stevens-Johnson syndrome and toxic epidermal necrolysis: epidemiology, risk factors, and predictive value of skin cultures. Medicine (Baltimore) 2010; 89:28.
- Power WJ, Ghoraishi M, Merayo-Lloves J, et al. Analysis of the acute ophthalmic manifestations of the erythema multiforme/Stevens-Johnson syndrome/toxic epidermal necrolysis disease spectrum. Ophthalmology 1995; 102:1669-1676.
- de Prost N, Mekontso-Dessap A, Valeyrie-Allanore L, et al. Acute respiratory failure in patients with toxic epidermal necrolysis: clinical features and factors associated with mechanical ventilation. Crit Care Med 2014; 42:118.
- Namdar T, Stang FH, Siemers F, et al. Mechanical ventilation in toxic epidermal necrolysis. Handchir Mikrochir Plast Chir 2010.
- French LE. Toxic epidermal necrolysis and Stevens-Johnson syndrome: our current understanding. Allergol Int 2006; 55:9-16.
- Hockett KC. Stevens-Johnson syndrome and toxic epidermal necrolysis: oncologic considerations. Clin J Oncol Nurs. 2004; 8:27-30, 55.
- Blanca M, Romano A, Torres MJ, et al. Update on the evaluation of hypersensitivity reactions to beta-lactams. Allergy 2009: 64 (2):183-193.
- Garcia-Doval I, LeCleach L, Bocquet H, et al. Toxic epidermal necrolysis and Stevens-Johnson syndrome: Does early withdrawal of causative drugs decrease the risk of death? Arch Dermatol 2000; 136:323-327.
- Gerdts B, Vloemans AF, Kreis RW. Toxic epidermal necrolysis: 15 years’ experience in a Dutch burns centre. J Eur Acad Dermatol Venereol 2007; 21:781-788.
- Pacquet P, Pierard GE. Topical treatment options for drug-induced toxic epidermal necrolysis (TEN). Expert Opin Pharmacother 2010: 11(15): 2447-2458.
- Mayes T, Gottschlich M, Khoury J, et al. Energy requirements of pediatric patients with Stevens-Johnson syndrome and toxic epidermal necrolysis. Nutr Clin Pract 2008; 23:547.
- Coss-Bu JA, Jefferson LS, Levy ML, et al. Nutrition requirements in patients with toxic epidermal necrolysis. Nutr Clin Pract 1997; 12:81-84.
- Hildreth MA. Caloric needs of patients with toxic epidermal necrolysis. J Am Diet Assoc 1990; 90:A99.
- Kumar G, Fadel HJ, Beckman TJ. 36-year-old man with productive cough and diffuse rash. Mayo Clin Proc 2006; 81:945-948.
- Schulz J, Sheridan RL, Ryan CM, et al. A 10-year experience with toxic epidermal necrolysis. J Burn Care Rehabil 2000. 21: 199-204.
- Knowles S, Shear NH. Clinical risk management of Stevens-Johnson syndrome/toxic epidermal necrolysis spectrum. Dermatol Ther 2009. 22(5):441-451.
- Morici MV, Galen WK, Shetty AK, et al. Intravenous immunoglobulin therapy for children with Stevens-Johnson syndrome. J Rheumatol 2000. 27(10):2494-2497.
- Koh MJ, Tay YK. An update on Stevens-Johnson syndrome and toxic epidermal necrolysis in children. Curr Opin Pediatr 2009. 21(4):505-510.
- Metry DW, Jung P, Levy ML. Use of intravenous immunoglobulin in children with Stevens-Johnson syndrome and toxic epidermal necrolysis: seven cases and review of the literature. Pediatrics 2003; 112:1430-1436.
- Prins C, Kerdel FA, Padilla RS, et al. Treatment of toxic epidermal necrolysis with high-dose intravenous immunoglobulins: multicenter retrospective analysis of 48 consecutive cases. Arch Dermatol 2003; 139:26-32.
- Tristani-Firouzi P, Petersen MJ, Saffle JR, et al. Treatment of toxic epidermal necrolysis with intravenous immunoglobulin in children. J Am Acad Dermatol 2002; 47:548-552.
- Brown KM, Silver GM, Halerz M, et al. Toxic epidermal necrolysis: does immunoglobulin make a difference? J Burn Care Rehabil 2004; 25:81-88.
- Kakourou T, Klontza D, Soteropoulou F, et al. Corticosteroid treatment of erythema multiforme major (Stevens-Johnson syndrome) in children. Eur J Pediatr 1997; 95:181-186.
- Schneck J, Fagot JP, Sekula P, et al. Effects of treatments on the mortality of Stevens-Johnson syndrome and toxic epidermal necrolysis: a retrospective study on patients included in the prospective EuroSCAR Study. J Am Acad Dermatol 2008; 58:33-40.
- Hynes AY, Kafkala C, Daoud YJ, Foster CS. Controversey in the use of high-dose systemic steroids in the acute care of patients with Stevens-Johnson syndrome. Int Ophthalmol Clin 2005. 45:25-48.
- Patterson R, Grammer LC, Greenberger PA, Lawrence ID, Zeiss CR, Detjen PF, et al. Stevens-Johnson syndrome (SJS): effectiveness of corticosteroids in management and recurrent SJS. Allergy Proc 1992; 13:89-95.
- Tripathi A, Ditto AM, Grammer LC, Greenberger PA, McGrath KG, Zeiss CR, et al. Corticosteroid therapy in an additional 13 cases of Stevens-Johnson syndrome: a total series of 67 cases. Allergy Asthma Proc 2000; 21:101-5.
- Halebian PH, Corder VJ, Madden MR, Finklestein JL, Shires GT. Improved burn center survival of patients with toxic epidermal necrolysis managed without corticosteroids. Ann Surg 1986; 204:503-12.
- Engelhardt SL, Schurr MJ, Helgerson RB. Toxic epidermal necrolysis: an analysis of referral patterns and steroid usage. J Burn Care Rehabil 1997; 18:520-4.
- Bastuji-Garin S, Fouchard N, Bertocchi M, et al. SCORTEN: A severity-of-illness score for toxic epidermal necrolysis. J Invest Dermatol 2000; 115:149-153.
- Trent JT, Kirsner RS, Romanelli P, Kerdel FA. Use of SCORTEN to accurately predict mortality in patients with toxic epidermal necrolysis in the United States. Arch Dermatol 2004; 140:890.
- Guegan S, Bastuji-Garin S, Poszepczynska-Guigne E, et al. Performance of the SCORTEN during the first five days of hospitalization to predict the prognosis of epidermal necrolysis. J Invest Dermatol 2006; 126:272.
- Valeyrie-Allanore L, Roujeau J-C. Epidermal necrolysis (Stevens-Johnson Syndrome and Toxic Epidermal Necrolysis). In: Fitzpatrick’s Dermatology in General Medicine, 8th Edition, Goldsmith LA, Katz SI, Gilchrest BA, Paller AS. (Eds), Mcgraw-Hill, 2012.
- Palmieri TL, Greenhalgh DG, Saffle JR, et al. A multicenter review of toxic epidermal necrolysis treated in U.S. burn centers at the end of the twentieth century. J Burn Care Rehabil 2002; 23:87.
- Firoz BF, Henning JS, Zarzabal LA, Pollock BH. Toxic epidermal necrolysis: five years of treatment experience from a burn unit. J Am Acad Dermatol 2012; 67:630.
- Kelemen JJ III, Cioffi WG, McManus WF, et al. Burn center care for patients with toxic epidermal necrolysis. J Am Coll Surg 1995; 180:273-278.
- Namdar T, von Wild T, Siemers F, et al. Does hypernatremia impact mortality in toxic epidermal necrolysis? Ger Med Sci 2010; 8:doc30.
- Westly ED, Wechsler HL. Toxic epidermal necrolysis: granulocytic leucopenia as a prognostic indicator. Arch Dermatol 1984; 120:721-726.
- Sekula P, Dunant A, Mockenhaupt M, et al. Comprehensive survival analysis of a cohort of patients with Stevens-Johnson syndrome and toxic epidermal necrolysis. J Invest Dermatol 2013; 133:1197.
- Finkelstein Y, Soon GS, Acuna P, et al. Recurrence and outcomes of Stevens-Johnson syndrome and toxic epidermal necrolysis in children. Pediatrics 2011; 128:723.