Authors: Nima Rejali, DO (EM Resident Physician, Hackensack University Medical Center) and Amit Gupta, MD (Assistant Program Director, Hackensack University Medical Center) // Edited by: Alex Koyfman, MD (@EMHighAK) and Brit Long, MD (@long_brit)
Meningitis is often a consideration in patients presenting to the Emergency Department (ED) with headache. Unfortunately, the “classic triad” of fever, neck pain, and headache is not common. While patients can present along a spectrum of disease, unfortunately the disease can result in rapid decompensation. This article will explore some of the serious complications of meningitis through cases and discuss management strategies to improve mortality and morbidity in the crashing meningitis patient.
Case 1
A 20-year-old male presents to the ED with his roommate. The roommate states that this morning before leaving for class the patient was complaining of slight headache and body aches. When he came back to his apartment after classes, his roommate was still in bed, diaphoretic, warm to touch, complaining of neck pain, and saying things that were not making sense. In the ED you recognize this patient may have bacterial meningitis. You place the patient on droplet isolation; prepare for a lumbar puncture; and order blood cultures with 2gm ceftriaxone, and 1gm vancomycin. When you return from getting your supplies, the patient is now hypotensive 80/40 mm/Hg and tachycardic at 122 bpm. You notice petechial lesions to his right foot and lift his gown to find extensive purpura over both thighs and abdomen.
Discussion
While all causes of meningitis can progress to septicemia (bacterial, viral, fungal), Neisseria meningitides is one of the most dreaded and serious etiologies of meningitis and sepsis. Even with early identification and IV antibiotics, there is a 10-15% mortality associated with this infection1. While it is difficult to ascertain the inciting organism in the acute meningitis patient who presents to the ED, history and exam may suggest meningococcal infection. These patients will typically report at least 1 of the classic symptoms of meningitis (fever, neck stiffness, headache) and also have severe myalgias in nearly all reported cases2. These patients will have the classic petechial/purpuric rash on presentation 50% of the time3. On exam, these patients may also present with pallor and mottling of the skin up to 70% of the time, which should be regarded as an indication of severe systemic illness2. While these patients are often young adults who live in communal settings such as college students or military barracks, rates have been reduced in these populations due to increased meningitis vaccination awareness4,5.

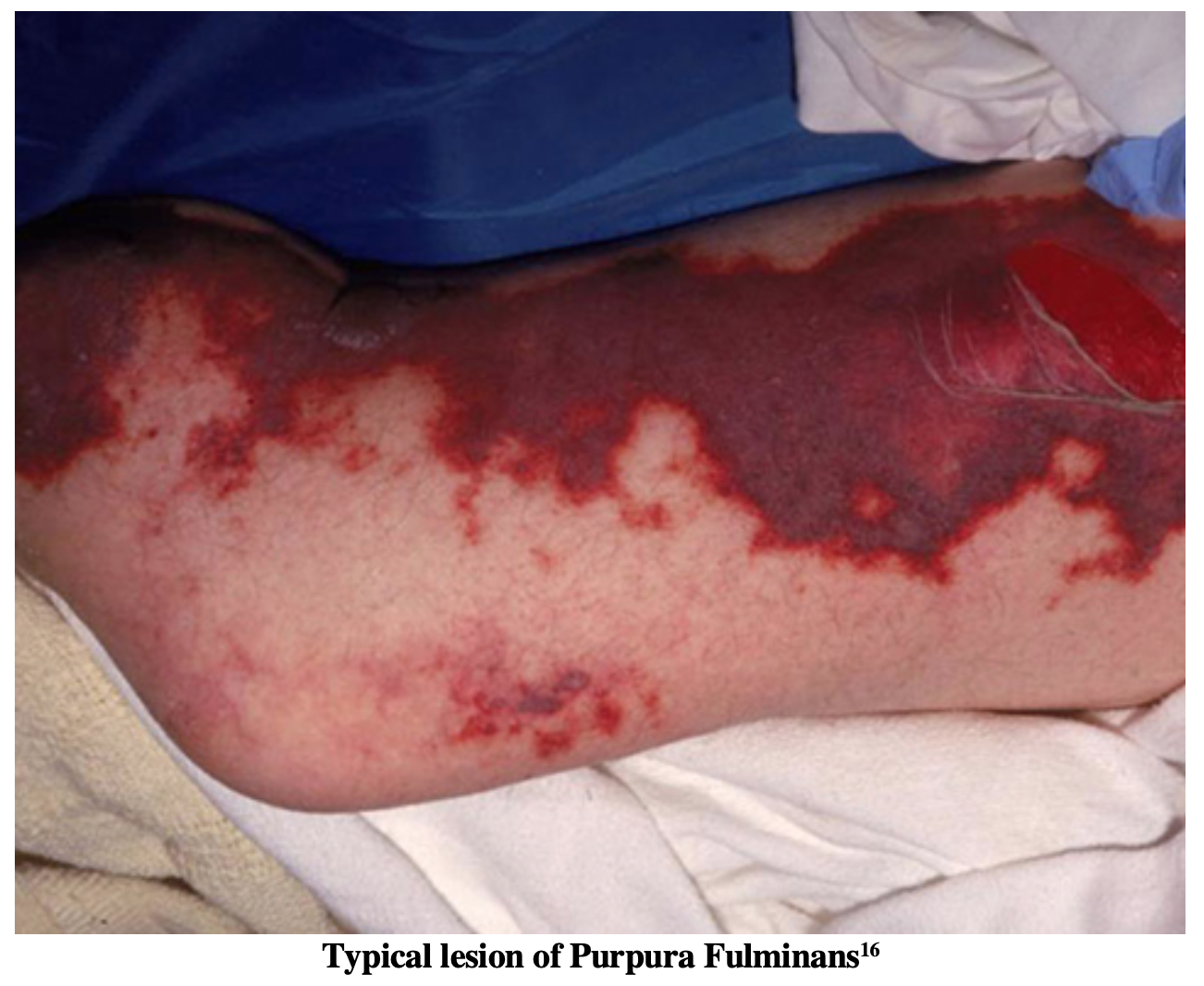
In patients with meningococcal meningitis, rash is a presenting symptom in 50-75% of reported cases3,7. The rash can range anywhere from mild petechial lesions to extensive diffuse purpura which can lead to skin necrosis and amputation of limbs8. Larger and more extensive rashes are an indicator of a greater degree of thrombocytopenia and coagulopathy (DIC) secondary to septicemia9.
The most deadly form of skin involvement secondary to meningococcal infection is purpura fulminans, an acquired, often fatal, coagulopathy characterized by extensive skin involvement which rapidly progresses to skin necrosis with disseminated intravascular coagulation (DIC). These patients will benefit from fresh frozen plasma (FFP) (10-20 ml/kg every 8 hours) and/or protein C concentrate (100-120 units/kg initial bolus)8,10. For reasons not yet thoroughly understood, protein C activation has been found to be remarkably reduced in cases of meningococcal sepsis11. As a result, it is believed that protein C concentrate is beneficial in these patients to help restore the balance of coagulation and anticoagulation factors which have become deranged in DIC. Two clinical trials have been conducted on a small populations of patients with meningococcal meningitis which have been associated with correction of coagulopathy without significant adverse events as well as reducing the rate of amputation in these patients12,13. As the definitive culture may not return until the patient is in the ICU, protein C concentrate will likely only be administered in the ICU.
If soft tissue/skin necrosis is found, consultation with general surgery is recommended as debridement, fasciotomy, and/or amputation may be indicated based on progression of illness14. In addition, the DIC and coagulopathy that occurs secondary to severe septicemia can also lead to thrombosis and hemorrhage of internal organs and exsanguination15. Patients may require aggressive volume expansion with crystalloid and blood products and intervention according to organ involvement such as intubation for respiratory failure from massive hemoptysis, surgical removal of infarcted internal organs, interventional radiology (IR) consultation for embolization, stress dose steroids for adrenal hemorrhage, or emergent hemodialysis for renal failure15.
Another complication of meningococcemia is Waterhouse-Friderichsen Syndrome. Patient with this will develop severe hypotension and shock secondary to adrenal failure from hemorrhage caused by DIC15. These patients can present as obtunded, hypotensive, hypoglycemic, hyponatremia, and hyperkalemic17. Care should be taken to identify and correct any metabolic abnormalities and initiate vasopressors with norepinephrine being the initial vasopressor of choice. This subgroup of patients will also benefit from stress dose steroids. The topic and steroids and sepsis, particularly in the ED, is a topic for another discussion. However, in critically ill patients requiring ICU care, hydrocortisone 100 mg IV should be administered18.
Pearls and Pitfalls
- Early identification and antibiotics save lives. Have a low threshold to start antibiotics, particularly if the patient is unstable.
- Petechial rash can quickly spread to full-blown purpura fulminans. If this is suspected, antibiotics and FFP/Protein C as well as emergent consultation with general surgery is indicated.
Case 2
A36-year-old female presents with her husband for headache and fever. She took acetaminophen prior to arrival with no improvement. She has no significant past medical history, and aside from a “mild cold” a few days ago she has felt otherwise well. Her neurologic exam is remarkable for an isolated cranial nerve VI palsy but is otherwise normal. She states her headaches are getting worse, and you order ketorolac, normal saline, metoclopramide, and a noncontrast CT head. You get a call that your patient is seizing in the CT scanner. You order 2mg of lorazepam and the seizure stops. Repeat exam shows a post-ictal state, but she now also has a new dilated right pupil. Repeat vitals reveal HR 46, BP 170/100, and GCS 4.
Discussion
Neurologic complications from meningitis range from mild such as transient cranial nerve palsy to brain herniation and death. In a review of 493 patients with bacterial meningitis, 28% had neurologic complications, and 9% had permanent deficits at time of hospital discharge19. Two potentially life-threatening complications include seizures and elevated intracranial pressure (ICP). While in our previous discussion shock and purpura were associated with Neisseria meningitides, neurologic complications are commonly associated with Streptococcus pneumoniae, aka pneumococcal meningitis20.
Three separate clinical features are associated with adverse outcomes such as death and permanent neurologic deficits at discharge: hypotension, altered mental status, and seizures20. Seizures in meningitis are thought to occur secondary to inflammatory changes and cytotoxic effects of the disease process in brain parenchyma. Dexamethasone therapy should be administered to all patients early in disease course with or before the initial IV antibiotics, before neurologic symptoms become present21. Although dexamethasone has primarily been shown to be efficacious only in patients with pneumococcal meningitis22, it is still recommended upon clinical suspicion of bacterial meningitis and can either be continued or discontinued as an inpatient. The Infectious Disease Society of America recommends dexamethasone 0.15mg/kg every 6 hours for 4 days23. A randomized placebo-controlled trial including patients with meningitis found significantly less seizure activity in patients receiving dexamethasone vs placebo24. Should seizures occur, standard treatment with parental benzodiazepines, i.e. lorazepam or diazepam, continuous cardiac monitoring, supplemental O2 as necessary, and aspiration prevention by turning the patient on side should be initiated. Once seizures have stopped, the patient should be loaded with an anti-epileptic, such as IV levetiracetam, or other recommend agent as per neurology consultation. Patients should also be evaluated for signs of elevated ICP.
Elevated ICP can present subtly as mild confusion, isolated cranial nerve palsy (most commonly CN VI), and papilledema or with severe symptoms such as obtundation, a nonreactive pupil, and bradycardia with hypertension (Cushing reflex)25. Once elevated ICP is identified or suspected, time is of the essence, as it can progress to herniation and/or death. Forego LP in the critically ill and administer antibiotics and steroids. If the patient is stable, a head CT may identify other causes of AMS such as stroke or mass and provide information regarding herniation or midline shift that may warrant emergent neurosurgical intervention. Magnetic resonance imaging with venography should also be considered to evaluate for cerebral venous sinus thrombosis, which requires anticoagulation. Dexamethasone may reduce ICP through reducing inflammation as well as increase vascular perfusion26,27. By lowering the ICP and increasing the MAP, we are in effect increasing the CPP (cerebral perfusion pressure), calculated by the below equation.

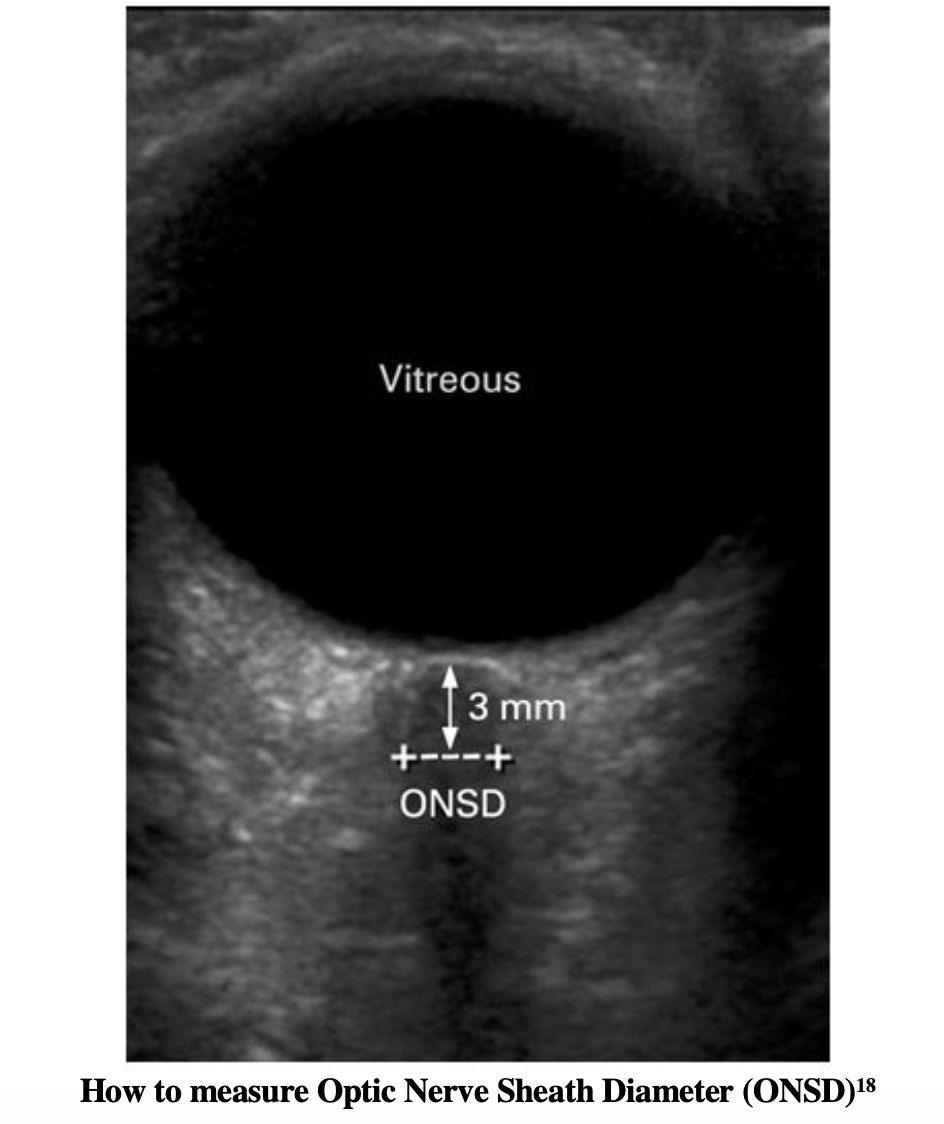
In order to maintain a recommended CPP>60 mm Hg with accurate measurement, invasive ICP monitoring will require neurosurgical consultation. ICP monitors are typically not placed in the ED, but surrogate markers can identify elevated ICP. Aside from the above mentioned physical exam findings, we can utilize bedside ocular US to assess for elevated ICP. Measurements of the optic nerve sheath diameter should be taken 3mm posterior to the globe. The diameter at this position should be measured twice, and if the average is greater than 5mm, elevated ICP should be suspected28. For a video walkthrough on how to perform an ocular US, we recommend the following video from Anna Pickens’ EM in 5.
Should ICP continue to rise, certain interventions may be necessary which should be done in conjunction with the neurosurgery and ICU team such as hypertonic saline infusion, mannitol infusion, and/or craniectomy29. Simple but effective methods to lower ICP are head of bed elevation to 30 degrees, pain control, adequate sedation, and fever control. Traditional teaching included Mannitol 20% solution with an initial bolus of 1g/kg30. As hypertonic saline is being studied more, it has supplanted the use of mannitol in many institutions. However, a meta-analysis suggests no difference in mortality between the two30. Many different concentrations of hypertonic saline exist with varying protocols. A bolus of 250ml 7.5% saline, targeting a goal sodium of 145-155 mEq, has been studied most extensively in the literature31. However, there have been few studies on uses of osmotic agents when elevated ICP is due to infectious processes, and most data and benefit are extrapolated from studies using osmotic agents for elevated ICP from traumatic brain injury31. These patients will require ICU level care and potentially CSF drainage.
Should intubation be required for airway protection, it should be performed by the most experienced physician, as recurrent attempts will potentially increase ICP32. Intravenous lidocaine is no longer recommended as an agent for pre-treatment33. Fentanyl 3 mcg/kg IV may be used, however be cautious with opioids in patients with severe hypotension34. Ketamine can be used for the hypotensive or normotensive patient but should be avoided if possible in hypertensive patients as the reflex hypertension can further increase ICP. Once intubated, the patient should be adequately sedated with a fast acting, easily titratable sedative such as propofol that allows for frequent repeat neuro exams. Tradition teaching has been to hyperventilate patient to pCO2 around 26-30 mmHg with the thought that hypocapnia will induce cerebral vasoconstriction; however, this has been linked to worse outcomes35. Instead, ventilate to a lower limit of 35mmHg with continuous capnography. Hyperventilating to lower CO2 levels should be reserved for when osmotic agents have failed to reduce ICP or for suspected acute herniation35.
Pearls and Pitfalls
- When patients arrive with suspected meningitis, we rarely know the pathogen at time of arrival to the ED. Steroids should be a part of initial management concurrent with the first dose of antibiotics to help minimize elevations in ICP and the complications that come along with it.
- Do not forget the basics when it comes to managing elevated ICP in the ED. Elevate the head of the bed to at least 30 degrees and provide analgesia. Should intubation be necessary, the most experienced physician should intubate, as repeated attempts will increase sympathetic response to laryngoscopy. Do not initially hyperventilate patients, as this is associated with poor outcomes.
References/Further Reading:
- Heckenberg SG, de Gans J, Brouwer MC, et al. Clinical features, outcome, and meningococcal genotype in 258 adults with meningococcal meningitis: a prospective cohort study. Medicine (Baltimore) 2008; 87:185-92.
- Thompson MJ, Ninis N, Perera R, et al. Clinical recognition of meningococcal disease in children and adolescents. Lancet 2006; 367:397-403.
- Carpenter RR, Pertersdorf RS, The clinical spectrum of bacterial meningitis. Am J Med 1962; 33:262-275.
- Brouwer MC, Tunkel AR, van de Beek D. Epidemiology, diagnosis, and antimicrobial treatment of acute bacterial meningitis. Clin Microbiol Rev. 2010;23(3):467–492. doi:10.1128/CMR.00070-09.
- Thigpen MC et al. Bacterial meningitis in the United States, 1998–2007. N Engl J Med2011 May 26; 364:2016-2025DOI: 10.1056/NEJMoa1005384
- “Meningitis.” Nihdirect, 12 Feb. 2019, www.nidirect.gov.uk/conditions/meningitis.
- Tsai J, Nagel MA, Gilden D. Skin rash in meningitis and meningoencephalitis. Neurology. 2013;80(19):1808–1811. doi:10.1212/WNL.0b013e3182918cda.
- Ghosh SK, Bandyopadhyay D, Dutta A (2009). “Purpura fulminans: a cutaneous marker of disseminated intravascular coagulation”. West J Emerg Med. 10(1): 41. PMC 2672288. PMID 19561767.
- Apicella, Michael. Clinical manifestations of meningococcal infection. In: UpToDate, Post, TW (Ed), UpToDate, Waltham, MA, 2018.
- Chalmers E, Cooper P, Forman K, Grimley C, Khair K, Minford A, Morgan M, Mumford AD (2011). “Purpura fulminans: recognition, diagnosis and management”. Arch Dis Child. 96(11): 1066–1071. Doi:10.1136/adc.2010.199919. PMID 21233082.
- Faust SN, Levin M, Harrison OB, et al. Dysfunction of endothelial protein C activation in severe meningococcal sepsis. N Engl J Med 2001; 345:408-16.
- de Kleijn ED, de Groot R, Hack CE, et al. Activation of protein C following infusion of protein C concentrate in children with severe meningococcal sepsis and purpura fulminans: a randomized, double-blinded, placebo-controlled, dose-finding study. Crit Care Med 2003; 31:1839-47.
- White B, Livingstone W, Murphy C, et al. An open-label study of the role of adjuvant hemostatic support with protein C replacement therapy in purpura fulminans-associated meningococcemia. Blood 2000; 96:3719-24.
- Wheeler JS, Anderson BJ, De Chalain TM. Surgical interventions in children with meningococcal purpura fulminans–a review of 117 procedures in 21 children. J Pediatr Surg 2003; 38:597-603.
- Leung L. Clinical features, diagnosis, and treatment of disseminated intravascular coagulation in adults. Post TW, ed. UpToDate. Waltham, MA: UpToDate Inc. https://www.uptodate.com/contents/clinical-features-diagnosis-and-treatment-of-disseminated-intravascular-coagulation-in-adults?topicRef=1274&source=see_link#H4
- Kelly, Robert. Approach to the Patient with Retiform (Angulated) Purpura. Post TW, ed. UpToDate. Waltham, MA: UpToDate Inc .http://www.uptodate.com/contents/approach-to-the-patient-with-retiform-angulated-purpura?search=purpura%2Bfulminans&source=search_result&selectedTitle=1~29&usage_type=default&display_rank=1.
- Kumar V, Abbas A, Fausto N (2005). Robins and Coltran: Pathological Basis of Disease(7th ed.). Elsevier. pp. 1214–15. ISBN 978-0-7216-0187-8.
- Ayyan, S.M. et al. Sonographic Measurement of Optic Nerve Sheath Diameter Compared With CT Scan for Detecting Elevated Intracranial Pressure of Head Injury Patients in Emergency Department Annals of Emergency Medicine, 2017; Volume 70, Issue 4, S155 – S156.
- Durand ML, Calderwood SB, Weber DJ, et al. Acute bacterial meningitis in adults. A review of 493 episodes. N Engl J Med 1993; 328:21-28.
- Aronin SI, Peduzzi P, Quagliarello VJ. Community-acquired bacterial meningitis: risk stratification for adverse clinical outcome and effect of antibiotic timing. Ann Intern Med 1998; 129:862-869.
- van de Beek D, de Gans J. Dexamethasone and pneumococcal meningitis. Ann Intern Med 2004; 141:327.
- Brouwer MC, McIntyre P, Prasad K, van de Beek D. Corticosteroids for acute bacterial meningitis. Cochrane Database Syst Rev 2015; :CD004405.
- Tunkel AR, Hartman BJ, Kaplan SL, et al. Practice guidelines for the management of bacterial meningitis. Clin Infect Dis 2004; 39:1267-84.
- de Gans J, van de Beek D, European Dexamethasone in Adulthood Bacterial Meningitis Study Investigators. Dexamethasone in adults with bacterial meningitis. N Engl J Med 2002; 347:1549-56.
- Tunkel AR, Scheld WM. Pathogenesis and pathophysiology of bacterial meningitis. Clin Microbiol Rev 1993; 6:118-36.
- Lutsar I, Friedland IR, Jafri HS, et al. Factors influencing the anti-inflammatory effect of dexamethasone therapy in experimental pneumococcal meningitis. J Antimicrob Chemother 2003; 52:651-55.
- van Furth AM, Roord JJ, van Furth R. Roles of proinflammatory and anti-inflammatory cytokines in pathophysiology of bacterial meningitis and effect of adjunctive therapy. Infect Immun 1996; 64:4883-90.
- Blaivas M. Elevated intracranial pressure detected by bedside emergency ultrasonography of the optic nerve sheath. Acad Emer Med.2003;10:376-381.
- Smith ER, Madsen JR. Cerebral pathophysiology and critical care neurology: basic hemodynamic principles, cerebral perfusion, and intracranial pressure. Semin Pediatr Neurol 2004; 11:89-104.
- Myles Dustin Boone, Achikam Oren-Grinberg, Timothy Matthew Robinson, Clark C. Chen, Ekkehard M. KasperSurg Neurol Int. 2015; 6: 177. Published online 2015 Nov 23. doi: 10.4103/2152-7806.170248.
- Holmes, JA. Therapeutic uses of hypertonic Saline in the Critically Ill Emergency Department Patient. Emergency Medicine Critical Care 2013. 3:1-16.
- Takahashi S, Mizutani T, Miyabe M, Toyooka H. Hemodynamic responses to tracheal intubation with laryngoscope versus lightwand intubating device (Trachlight) in adults with normal airway. Anesth Analg 2002; 95:480-84.
- Robinson N, Clancy M. In patients with head injury undergoing rapid sequence intubation, does pretreatment with intravenous lignocaine/lidocaine lead to an improved neurological outcome? A review of the literature. Emerg Med J 2001; 18:453-57.
- Splinter WM, Cervenko F. Haemodynamic responses to laryngoscopy and tracheal intubation in geriatric patients: effects of fentanyl, lidocaine and thiopentone. Can J Anaesth 1989; 36:370-76.
- Carney N, Totten AM, O’Reilly C, et al. Guidelines for the Management of Severe Traumatic Brain Injury, Fourth Edition. Neurosurgery 2017; 80:6-15.






