Authors: Mike Butterfield, MD, MS, MPH (EM Resident Physician, University of South Florida) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital) and Stephen Alerhand, MD (@SAlerhand)
Introduction
A number of atypical and recently described EKG patterns that may signal risk of transmural myocardial infarction (“STEMI-equivalents”) are must-knows for Emergency Physicians. Below we describe most of these patterns, dividing them between 1) those causing ST depression or T wave changes and 2) those causing ST elevation. For many cases, supporting data is quite limited and frequently recycled in subsequent reports.
After describing each pattern, we offer our recommendations on whether to call a STEMI activation or an EKG alert. At our institution, the latter initiates prompt cardiology consultation when the EKG pattern is concerning but does not meet strict criteria for an activation. At issue, as always, are the needs to balance the potential benefits derived from primary PCI with the risks, early intervention with false activation, sensitivity with specificity.
We agree, as Rokos et al. have argued, that reducing inappropriate cath lab activation should be a vital goal in the emergency management of acute coronary syndrome.1 Their proposed target – reducing the false activation rate to < 5% – may be overly ambitious, however. Standard STEMI criteria do miss a significant number of patients with obstructive lesions who would benefit from primary PCI, making the recognition of high-risk STEMI-equivalent patterns all the more necessary.2–6
Finally, while the discussion below focuses on electrocardiographic patterns, current guidelines identify at least two other clinical findings that should prompt cath lab activation but may be overlooked: intractable ischemic chest pain despite medical management and new regional wall motion abnormalities on bedside echocardiography.7
Part I: ST-Depressions and T-Wave Changes
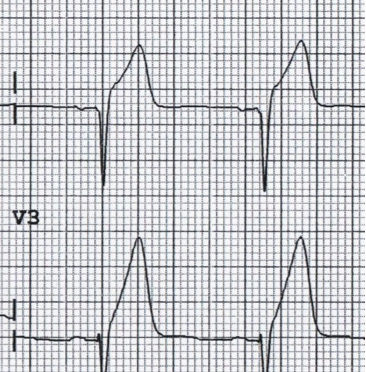
Isolated Posterior Myocardial Infarction.
While the posterior heart is involved in up to 20% of all STEMIs, isolated posterior STEMI is only found 5% of the time. On a typical anterior 12-lead EKG, key features of PMI include ST depressions leads V1-V3, with upright Rs and T-waves, which should trigger a repeat EKG with posterior leads.
Comment: We won’t duplicate the excellent post on posterior STEMIs recently written by Manuel Celedon for this site, found here.
Isolated ST-Depression in AVL
Reciprocal ST-depression (STD) in lead AVL is commonly seen in inferior STEMI, raising the question of whether it can be interpreted as a STEMI equivalent. Because the magnitude of an electrocardiographic vector depends on many factors (lead distance, individual anatomy, etc), STD may be apparent in the absence of clear-cut ST-elevation.8,9 Reciprocal depression in AVL demonstrated a strikingly high sensitivity for inferior STEMI in the two largest series to date by Birnbaum and Smith et al. Birnbaum et al’s (1993) study of 107 patients with “evolving first inferior AMI” found 97% had STD (>0.5 mm) in AVL (but this finding was present in the 93/93 patients with at least one inferior lead with ST-elevation > 1mm—the likely STEMI subgroup). Furthermore, STD was the sole EKG findings in 7.5% of patients who went on to develop biomarker-confirmed inferior MI.
Smith et al’s (2015) more recent study produced similar findings in three separate, retrospective cohorts. In cohort 1, 100% (154/154) of patients diagnosed with inferior STEMI over a 6-year period had some degree (> 0.25mm) of STD in AVL, while this finding was not present in any patients diagnosed with pericarditis (n=54, cohort 2). In a third cohort, 46/54 (85%) patients with “subtle” inferior STEMI” (STE < 1 mm) had STD in AVL.
Comment: The reported sensitivities of STD in AVL in the studies by Birnbaum and Smith are impressive, but their findings require prospective validation in emergency department patients. Nor did these or other studies provide sufficient temporal data to assess whether or not STD in AVL is useful as a sign of early STEMI (or just a concurrent finding). Importantly, both studies excluded patients with confounding patterns such as left ventricular hypertrophy or bundle branch blocks; as Brady (2002) has shown, the specificity of reciprocal changes for STEMI drops considerably when these patients are included.10
Smith’s data on reciprocal changes in AVL to help identify cases of “subtle” STEMI are intriguing and merit further study. Since these patients have outcomes that are similar, not better, than STEMI patients in general, they may warrant early cardiology consultation.11
Hyperacute T-waves (HATWs) are a well-known but variably defined feature of early STEMI (especially anterior STEMI), identifiable anywhere between 5 and 30 minutes after infarction. They tend to be broad based, taller than normal T-waves (> 5mm and 10mm in the limb and precordial leads), and may be symmetric or asymmetric.12 True HATWs may be associated with reciprocal changes in opposing leads as well as increased R wave amplitude (if prior EKG is available).
There are numerous causes of tall T-waves, but perhaps the most important ones to distinguish from HATWs are hyperkalemia, benign early repolarization (BER), and left ventricular hypertrophy (Box 1).13
Box 1
- Hyperkalemia: narrow-based, symmetric, and peaked (sharp); may have a widened QRS
- BER: diffuse, peaked, associated with J-point elevation
- LVH: look for voltage criteria and a strain pattern

[Courtesy of Life in the Fast Lane (LITFL)]
One set of criteria to define HATWs was developed by Collins et al, who compared 21 patients with tall T-waves and AMI with 513 patients with tall T-waves alone (Box 2). While achieving a sensitivity and specificity of 69% and 98%, the criteria are problematic due to their extensive exclusion criteria, relative complexity, and lack of ability to distinguish STEMI from NSTEMI.14 Although not formally evaluated, a simpler approach suggested by Sovari et al.—looking for a T-wave to QRS ratio of > 75%–seems like a reasonable option.15
Box 2
- ST on/T amp of greater than 25%
- T amp/QRS amplitude (QRS amp) of greater than 75%
- ST on of greater than 0.30 mV
- Patients older than 45 years
Comment: Hyperacute T-waves should prompt a STEMI activation, after consideration of BER or LVH. Because morphology alone may not easily distinguish HATWs from hyperkalemic T-waves, it is reasonable to delay activation and obtain a stat blood gas electrolyte panel when the latter remains high in the differential.
De Winter Pattern
In 2008, using data from a large Dutch registry, De Winter and colleagues described a distinct EKG pattern found in approximately 2% of patients with angiographically-proven anterior myocardial infarction: upsloping ST elevation in AVR (≥ 1 mm) with ST depressions and tall T waves in the precordial leads. Unlike the hyperacute T waves they may resemble, de Winter T waves are not transient findings and remain present in subsequent EKGs.16
De Winter and Verouden corroborated these findings in a follow-up study from 2009 (presumably using a different dataset), and more recently Taglieri et al. reported on nine patients with the pattern – all with proximal LAD occlusion – in their large retrospective series.17
(Courtesy of LITFL)
Comment: One major problem with the de Winter pattern is that it has only been described in retrospective series of angiography-proven infarction. Their data tells us little about the specificity and other test characteristics of this pattern in a population of ED patients presenting with chest pain. Nevertheless, we believe that at the very least patients presenting with this concerning pattern deserve STEMI alert and prompt consultation with cardiology.
Part II: ST-Segment Changes
ST Elevation (STE) in AVR
Several small case series from the early 2000s suggested that diffuse ST depression combined with STE in AVR may be indicative of “subendocardial ischemia” from significant left main coronary (LMCA) stenosis, left main equivalent disease (LMEQ, significant disease of the left anterior descending and left circumflex), or three-vessel disease.18–20 Some authors have advocated for these findings to be recognized as a STEMI equivalent, and American Heart Association/American College of Cardiology has recommended that computerized interpretation of this pattern be read as “ischemia due to multivessel or left main coronary artery obstruction.”21,22
However, the subendocardial pattern cannot be applied to patients with other potential causes of diffuse ST depression such as cardiomyopathies, left bundle branch block or left ventricular hypertrophy. Even with these exclusions, the lack of specificity of this pattern is documented in case reports as well as a recent study by Knotts et al (2013).23 In this series, only 23% of 57 patients with the subendocardial pattern who underwent angiography had significant LMCA or LMEQ disease, which increased to 43% if triple vessel disease was included. The subgroup of patients presenting with ACS (n=34) was not more likely to have significant disease in any distribution, but did receive intervention more frequently. Citing the study by Knotts, one pair of prominent cardiologists have argued against interpreting the subendocardial pattern in isolation as a STEMI equivalent, since doing so “could lead to overdiagnosis and utilization of emergent coronary angiography.”24 Nevertheless, current guidelines recommend early reperfusion for patients presenting with this pattern.
Comment: While it is clear that unstable patients with a subendocardial ischemia pattern require STEMI activation, it less clear which patients with stable hemodynamics may actually benefit from emergent angiography. Some of these patients may indeed have high-grade lesions in the left main or its proximal branches that would place a large myocardial territory at risk of infarction. Given the lack of consensus in the literature, we would recommend initiating an EKG alert rather than activation and prompt cardiology consultation (while holding off on clopidogrel or other P2Y12 inhibitors). Performing bedside echocardiography may be particularly helpful in this group of patients.
Presumed New Left Bundle Branch Block
Historically, new or presumed new left bundle branch block identified in the setting of acute coronary syndrome was designated as a STEMI equivalent. While that general sentiment remains prevalent, it is not in keeping with current evidence or guidelines.
(Courtesy of LITFL)
For example, Chang et al (2009) showed that patients with chest pain and (presumed) new LBBB (n= 55) were not more likely to have myocardial infarction (STEMI or non) than those with an old or without LBBB (n = 136 and 7746), nor were they more likely to die within 30 days.25 A review of recent literature by Neeland et al. (2012) found that only about 40% (89/225) of patients with presumed new LBBB had a culprit lesion on angiography.26 STEMI guidelines from 2013 have removed new or presumed new LBBB as diagnostic of myocardial infarction “in isolation.”
Comment: It is problematic to lump together patients with new and “presumed new” LBBB, given the natural history of LBBB—many of the latter likely had chronic but undetected LBBB. But the patchwork state of our medical records systems does not allow us to easily distinguish between these two groups at present.
New or presumed new LBBB is thus not sufficient to prompt STEMI activation in patients presenting with ACS. Clear-cut exceptions include those patients with unstable hemodynamics or meeting Sgarbossa criteria (below). Additionally, bedside echocardiography demonstrating severely reduced ejection fraction or wall motion abnormalities.
STEMI Equivalent in Paced Rhythms
The problem of diagnosing STEMI in patients with ventricular pacemakers has paralleled that of STEMI in patients with LBBB. In the overwhelming majority of paced rhythms, depolarization is initiated from a right ventricular lead, bypassing the Bundle of His and prolonging depolarization of the left ventricle. On EKG, this means that most paced rhythms have a morphology that mimics LBBB, with depolarization characterized by deep S-waves followed by ST-elevation and upright T-waves during repolarization (discordance).27,28
Finding sufficient numbers of patients with both a pacemaker and angiographic evidence of acute occlusion has been difficult. The largest series to date was published by Sgarbossa et al, who applied their criteria for STEMI in patients with LBBB to a subset of 32 patients with pacemaker and coronary occlusion from the GUSTO-I trial. These criteria (Box 3) had high specificity but low sensitivity for detecting STEMI.29
Box 3: Original Sgarbossa Criteria
- Concordant ST elevation > 1mm
- Concordant ST depression > 1mm in leads V1-3
- Discordant ST elevation > 5mm
More recently, Smith et al. proposed replacing Sgarbossa’s criterion of an absolute measure of discordant ST-elevation (≥ 5mm) for a relative one (ST-elevation ≥ 25% of the S-wave), reporting significantly increased sensitivity for detecting STEMI in patients with LBBB, with minimal decrease in specificity.30 These modified Sgarbossa criteria have also been applied to patients with paced rhythms.31,32 Validation studies for either group of patients (LBBB or paced) have yet to be performed, however.
Comment: Though the literature is limited, the modified Sgarbossa criteria by Smith et al. appear to be a useful guide for deciding to call a STEMI activation in patients with paced rhythms.
Conclusion
We have outlined above many EKG patterns that emergency physicians should readily recognize and act upon. Though the data supporting several of these patterns is still preliminary (e.g. STD in AVL, modified Sgarbossa), all should be reason enough to at least broker timely discussion with cardiology. At the same time, we should be adept at recognizing the clinical STEMI equivalents of intractable symptoms and new wall motion abnormalities on bedside echocardiography.
References / Further Reading
Beyond the primary literature, this post has benefitted considerably from related discussions on Life in the Fast Lane, EM Crit, and Steve Smith’s ECG blog. An extended discussion between Smith and Scott Weingart can be found here.
- Rokos IC, French WJ, Mattu A, et al. Appropriate cardiac cath lab activation: optimizing electrocardiogram interpretation and clinical decision-making for acute ST-elevation myocardial infarction. Am Heart J 2010;160(6):995–1003, 1003.e1–8.
- Bachour F, Asinger R. Activating primary percutaneous coronary intervention for STEMI that is not: the collateral damage of improving door-to-balloon time: comment on “Prevalence and factors associated with false-positive ST-segment elevation myocardial infarction diagnoses at primary percutaneous coronary intervention-capable centers.” Arch Intern Med 2012;172(11):871–2.
- Chung S-L, Lei M-H, Chen C-C, Hsu Y-C, Yang C-C. Characteristics and prognosis in patients with false-positive ST-elevation myocardial infarction in the ED. Am J Emerg Med 2013;31(5):825–9.
- McCabe JM, Armstrong EJ, Kulkarni A, et al. Prevalence and factors associated with false-positive ST-segment elevation myocardial infarction diagnoses at primary percutaneous coronary intervention–capable centers: a report from the Activate-SF registry. Arch Intern Med 2012;172(11):864–71.
- Macfarlane PW, Browne D, Devine B, et al. Modification of ACC/ESC criteria for acute myocardial infarction. J Electrocardiol 2004;37 Suppl:98–103.
- Wagner GS, Macfarlane P, Wellens H, et al. AHA/ACCF/HRS recommendations for the standardization and interpretation of the electrocardiogram: part VI: acute ischemia/infarction: a scientific statement from the American Heart Association Electrocardiography and Arrhythmias Committee, Council on Clinical Cardiology; the American College of Cardiology Foundation; and the Heart Rhythm Society. Endorsed by the International Society for Computerized Electrocardiology. J Am Coll Cardiol 2009;53(11):1003–11.
- O’Gara PT, Kushner FG, Ascheim DD, et al. 2013 ACCF/AHA guideline for the management of ST-elevation myocardial infarction: a report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation 2013;127(4):e362–425.
- Hassen GW, Costea A, Smith T, et al. The neglected lead on electrocardiogram: T wave inversion in lead aVL, nonspecific finding or a sign for left anterior descending artery lesion? J Emerg Med 2014;46(2):165–70.
- Goldberger AL, Erickson R. Subtle ECG sign of acute infarction: prominent reciprocal ST depression with minimal primary ST elevation. Pacing Clin Electrophysiol PACE 1981;4(6):709–12.
- Brady WJ, Perron AD, Syverud SA, et al. Reciprocal ST segment depression: impact on the electrocardiographic diagnosis of ST segment elevation acute myocardial infarction. Am J Emerg Med 2002;20(1):35–8.
- Martí D, Mestre JL, Salido L, et al. Incidence, angiographic features and outcomes of patients presenting with subtle ST-elevation myocardial infarction. Am Heart J 2014;168(6):884–90.
- Brady WJ, Connolly M. Chapter 16. What is a Hyperacute T Wave? In: Critical Decisions in Emergency and Acute Care Electrocardiography. Wiley; 2009. p. 115–21.
- Somers MP, Brady WJ, Perron AD, Mattu A. The prominent T wave: electrocardiographic differential diagnosis. Am J Emerg Med 2002;20(3):243–51.
- Collins MS, Carter JE, Dougherty JM, Majercik SM, Hodsden JE, Logue EE. Hyperacute T-wave criteria using computer ECG analysis. Ann Emerg Med 1990;19(2):114–20.
- Sovari AA, Assadi R, Lakshminarayanan B, Kocheril AG. Hyperacute T wave, the early sign of myocardial infarction. Am J Emerg Med 2007;25(7):859.e1–7.
- de Winter RJ, Verouden NJW, Wellens HJJ, Wilde AAM, Interventional Cardiology Group of the Academic Medical Center. A new ECG sign of proximal LAD occlusion. N Engl J Med 2008;359(19):2071–3.
- Verouden NJ, Koch KT, Peters RJ, et al. Persistent precordial “hyperacute” T-waves signify proximal left anterior descending artery occlusion. Heart Br Card Soc 2009;95(20):1701–6.
- Yamaji H, Iwasaki K, Kusachi S, et al. Prediction of acute left main coronary artery obstruction by 12-lead electrocardiography. ST segment elevation in lead aVR with less ST segment elevation in lead V(1). J Am Coll Cardiol 2001;38(5):1348–54.
- Kurisu S, Inoue I, Kawagoe T, et al. Electrocardiographic features in patients with acute myocardial infarction associated with left main coronary artery occlusion. Heart Br Card Soc 2004;90(9):1059–60.
- Hirano T, Tsuchiya K, Nishigaki K, et al. Clinical features of emergency electrocardiography in patients with acute myocardial infarction caused by left main trunk obstruction. Circ J Off J Jpn Circ Soc 2006;70(5):525–9.
- Hennings JR, Fesmire FM. A new electrocardiographic criteria for emergent reperfusion therapy. Am J Emerg Med 2012;30(6):994–1000.
- Lawner BJ, Nable JV, Mattu A. Novel patterns of ischemia and STEMI equivalents. Cardiol Clin 2012;30(4):591–9.
- Knotts RJ, Wilson JM, Kim E, Huang HD, Birnbaum Y. Diffuse ST depression with ST elevation in aVR: Is this pattern specific for global ischemia due to left main coronary artery disease? J Electrocardiol 2013;46(3):240–8.
- Birnbaum I, Birnbaum Y. High-risk ECG patterns in ACS–need for guideline revision. J Electrocardiol 2013;46(6):535–9.
- Chang AM, Shofer FS, Tabas JA, Magid DJ, McCusker CM, Hollander JE. Lack of association between left bundle-branch block and acute myocardial infarction in symptomatic ED patients. Am J Emerg Med 2009;27(8):916–21.
- Neeland IJ, Kontos MC, de Lemos JA. Evolving considerations in the management of patients with left bundle branch block and suspected myocardial infarction. J Am Coll Cardiol 2012;60(2):96–105.
- Rosner MH, Brady WJ. The electrocardiographic diagnosis of acute myocardial infarction in patients with ventricular paced rhythms. Am J Emerg Med 1999;17(2):182–5.
- Chan TC, Cardall TY. Electronic pacemakers. Emerg Med Clin North Am 2006;24(1):179–94, vii.
- Sgarbossa EB, Pinski SL, Gates KB, Wagner GS. Early electrocardiographic diagnosis of acute myocardial infarction in the presence of ventricular paced rhythm. GUSTO-I investigators. Am J Cardiol 1996;77(5):423–4.
- Smith SW, Dodd KW, Henry TD, Dvorak DM, Pearce LA. Diagnosis of ST-elevation myocardial infarction in the presence of left bundle branch block with the ST-elevation to S-wave ratio in a modified Sgarbossa rule. Ann Emerg Med 2012;60(6):766–76.
- Schaaf SG, Tabas JA, Smith SW. A patient with a paced rhythm presenting with chest pain and hypotension. JAMA Intern Med 2013;173(22):2082–5.
- Walsh BM, Karim R, Smith SW. A patient with a biventricular pacemaker presenting with chest pain. JAMA Intern Med 2015;175(6):1053–5.