Author: Drew A. Long (@drewlong2232, Emergency Medicine, San Antonio, TX) // Reviewed by: Alex Koyfman, MD (@EMHighAK); Manpreet Singh, MD (@MPrizzleER); and Brit Long, MD (@long_brit)
Case 1
A 62-year-old male presents to the Emergency Department (ED) with fatigue, cough, and shortness of breath that has been worsening over the past week. He has a history of COPD, heart failure, and hypertension. His vital signs include HR 95, BP 155/95, RR 35, SpO2 88%, and temperature 99°F. He is in moderate respiratory distress with mild wheezing along with diffuse crackles on exam. The patient is worried he contracted COVID-19 from a coworker who was recently sick. What other life-threatening conditions must be on your differential diagnosis for this patient with cough and shortness of breath?
Case 2
A 36-year-old female presents to the ED with fever, cough, shortness of breath, and malaise over the past several days. She has no medical problems but does occasionally vape and smoke marijuana. She denies any current use of other illicit drugs, though she admits to previous cocaine and heroin abuse. Vital signs are notable for a HR 125, BP 130/80, RR 25, SpO2 90%, and temperature 103°F. She has diffuse rhonchi on pulmonary exam and is in moderate respiratory distress. The patient appears anxious and is asking to be tested for COVID-19, as she has been anxiously following the news. What other life-threatening conditions should be on your differential diagnosis?
Pearls
- Do not anchor on the diagnosis of COVID-19 and ignore other life-threatening conditions in the patient presenting with fever, cough, and/or shortness of breath.
- As COVID-19 becomes more prevalent, the patient may have COVID-19 along with exacerbations of pre-existing medical conditions such as acute heart failure, COPD, etc.
- Ultrasound is an essential tool in the patient with undifferentiated shortness of breath as it can evaluate for multiple pathologic conditions.
- Multiple conditions other than COVID-19 can result in ground-glass opacities on CXR or chest CT.
- Have a low threshold for gathering blood cultures and initiating broad spectrum antibiotics if you are concerned for sepsis, even if the patient likely has COVID-19.
- Be cautious ordering a 30 cc/kg fluid bolus for septic patients if you have a strong suspicion the patient’s presentation is due to acute COVID-19, as this high volume of fluid may contribute to pulmonary edema and worsen respiratory failure.
- Severe COVID-19 infection leads to a hypercoagulable state that may cause DVT and/or PE.
- Order a CT Pulmonary Angiogram if the patient has any concerning features or risk factors for PE.
- If there is high suspicion for COPD or acute heart failure causing respiratory distress, start non-invasive positive pressure ventilation early as this decreases mortality and the need for intubation in COPD and heart failure exacerbations.
- COVID-19 patients with acute myocardial infarction should be treated with PCI if available.
- If giving thrombolytics for acute MI or massive PE in a patient with possible acute COVID-19 infection, do a bedside cardiac US to evaluate for a pericardial effusion prior to administration of thrombolytics.
Introduction
In December 2019, a new virus was discovered that was infecting people in Wuhan, China.1 This virus was found to be a novel RNA betacoronavirus, from the same family as the Severe Acute Respiratory Syndrome (SARS) virus and Middle Eastern Respiratory Syndrome (MERS) virus.2 This virus was named Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2), with the resultant disease process named coronavirus disease of 2019, or COVID-19.3 For simplicity, this manuscript will refer to both the virus and disease process as COVID-19. Over the next several months, this virus spread rapidly across the globe. The number of worldwide cases as of 28 April is over 3 million, with over 1 million cases and 50,000 deaths in the United States.4,5
COVID-19 is an enveloped positive sense RNA betacoronavirus that binds to angiotensin-converting enzyme-2 receptors, which are found in type II alveolar cells of the lungs, as well as cells in blood vessels, the heart, and intestines.2 It is a respiratory virus that is primarily transmitted from person to person by respiratory droplets.3 COVID-19 can also be spread through fomites, which can occur when a person touches a surface contaminated with COVID-19 through respiratory droplets and then touches a mucous membrane (eyes, mouth, or nose).6 The incubation period of the virus is generally 4 to 5 days, with a range of 1 to 14 days.7 COVID-19 is estimated to be slightly more infective than influenza, with a R0 (number of resultant infections from a single individual) of 2 to 3,8 though a more recent study estimated a higher R0 of 4.7-6.6.9 Risk factors for severe disease include advanced age, chronic medical conditions (most notable cardiovascular disease, pulmonary disease, and diabetes), immunocompromised status, and cancer.10,11
Clinical Manifestations
COVID-19 presents similarly to other viral upper respiratory illnesses. The most common symptoms are cough (68-76%), fever (44%), fatigue (38%), sputum production (34%), shortness of breath (19-56%), sore throat (14%), and headache (14%).12,13 Some patients may present with hypoxemia without significant shortness of breath or cough, which occurs more commonly in elderly patients.14 Dyspnea on presentation may indicate a more severe course.13 Patients presenting at a later stage of illness may present with acute respiratory distress syndrome (ARDS), disseminated intravascular coagulation (DIC), and myocarditis.15
Multiple other symptoms have been reported with COVID-19. There are case reports of patients presenting acutely with chest tightness and palpitations secondary to acute COVID-19 infection.16 Gastrointestinal (GI) symptoms have been reported in 10-50% of patients and may precede respiratory symptoms.2,17 The most commonly reported GI symptom is diarrhea.2,17 Anosmia and dysgeusia have also been reported to occur prior to other symptoms, and may be an early clue of COVID-19 infection.18
The patient’s severity of presentation depends on multiple factors, including the stage of disease. A clinical staging of COVID-19 was proposed by Siddiqi and Mehra (Figure 1), in which they divide the COVID-19 infection into three stages: mild (early infection), pulmonary involvement, and systemic hyperinflammation.15 Patients presenting early in the disease course with mild infection will likely have normal vital signs and symptoms of an upper respiratory infection. With stage II, pulmonary involvement, patients will complain of symptoms concerning for pneumonia (cough, shortness of breath, fatigue) and may display significant hypoxemia.15 A minority of patients will transition to the hyperinflammatory stage, in which they display a sepsis-like host response and cytokine storm.19 Patients in this stage have a very high mortality, and may have ARDS, DIC, heart failure, myocarditis, and circulatory failure.

Diagnostic Evaluation
Evaluation for COVID-19 often begins with a chest X-ray (CXR) for patients with respiratory complaints. A study of admitted COVID-19 patients found that only 69% had abnormal CXR on presentation.20 A variety of findings has been reported on CXR, including bilateral reticular nodular opacities, focal consolidations, ground-glass opacities, pulmonary edema, and interstitial opacities.20,21 Findings on CXR that may support an alternative diagnosis include a pleural effusion, lobar pneumonia, or focal mass.20,21 In cases in which the diagnosis is unclear, chest computed tomography (CT) may be helpful for further evaluation of pulmonary involvement or to evaluate for alternative diagnoses.
Chest CT is thought to be much more sensitive than CXR, though indications for obtaining a chest CT are not well defined. One study of 51 patients with diagnoses confirmed through nucleic acid testing found a low rate of missed diagnosis for chest CT, with only 2 cases (3.9%) missed by CT.22 CT findings are thought to occur more commonly in patients with severe disease. Findings include multilobar involvement (70-75%), ground-glass opacities (45-57%), and consolidations (31-33%).22 Feng et al proposed 4 stages of lung involvement in COVID-19 infection, shown in Table 1.23 Lung disease was found to be most extensive on CT scan 10 days after onset of initial symptoms.

Ground-glass opacities (GGO), while common in COVID-19, is a nonspecific finding on imaging. GGO on chest CT refer to areas in the lungs of increased attenuation, with preserved bronchial and vascular markings.24 When present in patients with COVID-19, GGO are most commonly bilateral, peripheral, and basal in distribution.2,10,12,13,16,17,22 While a hallmark of severe COVID-19 infection, GGO can occur in multiple other disease processes (Table 2).25
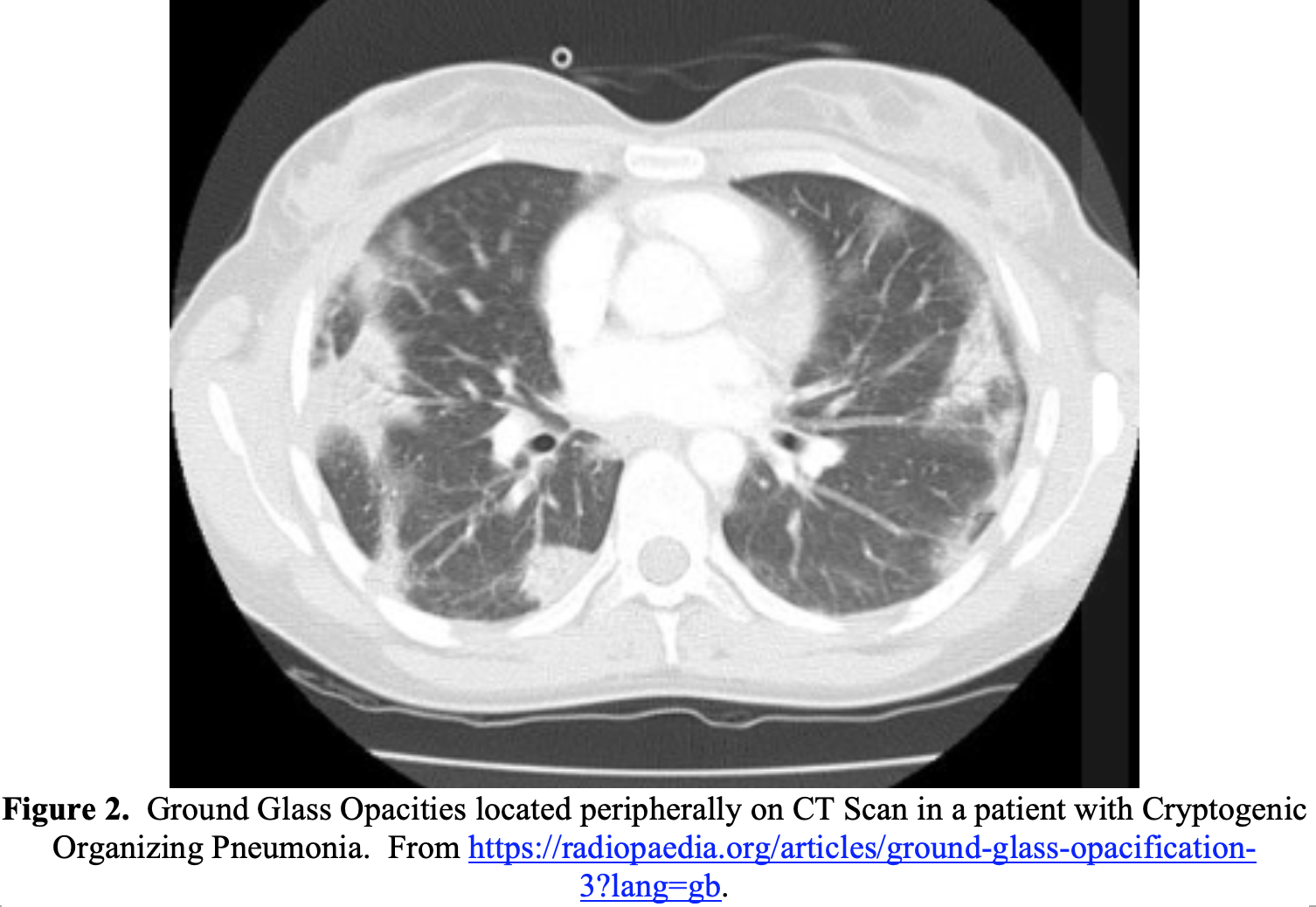

Ultrasound (US) has an unclear role in the evaluation of COVID-19. US findings in COVID-19 include B-lines, pleural irregularities, and subpleural consolidations.26 US may be helpful, especially if CXR shows nonspecific findings and CT is not immediately available. Additionally, US can evaluate for other alternative diagnoses, such as acute heart failure exacerbation or signs of right heart strain. However, US puts the clinician in close proximity to the potentially infected patient. A sterile probe cover should be used when ultrasounding patients with possible COVID-19, and the US must be thoroughly sterilized after use. See this prior emDOCs post on ultrasound in COVID-19 for more details.
Laboratory Evaluation
Laboratory evaluation is often helpful in both making the diagnosis of COVID-19 and risk stratification. Lymphopenia is present in about 70-80% of patients.2 Patients may also have a prolonged prothrombin time, elevated lactate dehydrogenase, thrombocytopenia, leukopenia, and elevated C-reactive protein.2 C-reactive protein elevation seems to coincide with more severe presentations.19 If a patient appears to be in severe respiratory distress and has a normal CRP, this may point away from a diagnosis of COVID-19 to a different etiology (such as CHF, COPD, etc.). D-dimer may also be elevated, and when elevated portends a worse prognosis.27 Troponin elevation may also occur with severe COVID-19 infection, and elevated troponin portends a worse prognosis.27,28 A final laboratory study that may be useful is procalcitonin. The role of procalcitonin is not currently well defined in diagnosis of COVID-19; however, an elevated procalcitonin in the setting of acute COVID-19 infection is considered a poor prognostic sign.29 Elevated procalcitonin is associated with a 5-fold increase in mortality in acute COVID-19 disease.29
If available, a reverse transcriptase-polymerase chain reaction (RT-PCR) test for COVID-19 should be used on patients that are sick or will require hospital admission. Unfortunately, the sensitivity of RT-PCR is thought to be around 75%.30 A Chinese study of 205 patients found that the RT-PCR test was 93% sensitive with bronchoalveolar samples (intubated patients), 72% sensitive with sputum samples, and 63% with nasopharyngeal samples.31 Multiple factors likely contribute to the sensitivity of the test, including the severity of illness, the patient’s viral load, and whether the patient is presenting early or late in his/her disease course. Ultimately, if a clinician is suspicious for COVID-19, a negative RT-PCR test does not rule out COVID-19.
Management
As there are no approved medications for the treatment of acute COVID-19 infection, management consists mainly of supportive care. Specific therapeutics for COVID-19 will not be included in this review. See these emDocs posts for more: Antivirals, Chloroquine/Hydroxychloroquine, and Therapy Update.
COVID-19 Mimics
Multiple conditions can present similarly to COVID-19, some of which carry a significant morbidity and mortality. Table 3 lists conditions that can mimic the presentation of COVID-19, along with red flags and treatment considerations for each disease process. While this is not an exhaustive list of COVID-19 mimics, this list consists of conditions that may present similarly to acute COVID-19 infection that if missed may have dire consequences. Importantly, as COVID-19 infection becomes more prevalent among the general population, COVID-19 may cause exacerbations of underlying medication conditions, such as COPD. Alternatively, the patient’s presentation may be due to another disease process, such as a pulmonary embolism, and the patient may have a concomitant COVID-19 infection that is a red herring to the patient’s acute presentation in the ED. While it may be difficult in the ED to distinguish whether COVID-19 or another disease process is causing the patient’s acute illness, the Emergency Clinician must not anchor on a diagnosis of COVID-19 and continue to consider other potentially life-threatening conditions.
Pulmonary Embolism
Pulmonary embolism (PE) most commonly occurs when a thrombus embolizes to the pulmonary vasculature, most commonly originating from the deep venous circulation. PE can be difficult to diagnose, as clinical presentation can vary greatly. The Emergency Physician must consider certain risk factors when entertaining the diagnosis of PE. Risk factors with the highest odds ratio (OR > 10) for acute PE include spinal cord injury, recent major surgery, major trauma, and hip or knee replacement.32 The highest risk for PE occurs around 10 days after major surgery and is thought to continue for at least 4 weeks.32 Risk factors with an OR of 2-9 for PE include active malignancy, hormone replacement therapy, postpartum state, history of previous deep venous thrombosis or PE, thrombophilia, congestive heart failure, and central venous access.32 For patients with a history of cancer, adenocarcinomas and metastatic cancer lead to the highest risk of PE in patients with solid malignancy, while acute leukemias and myeloma confers the greatest risk among patients with hematologic malignancies.32 Risk factors with an OR < 2 include prolonged sitting (in car or plane flight), increased age over 50, and laparoscopic surgery.32
The clinical presentation of PE can vary greatly. PE may easily be mistaken as acute COVID-19 infection, as a patient with acute PE commonly presents with varying degrees of shortness of breath. The most common complaints from acute PE are dyspnea and tachypnea (81%), pleuritic chest pain (39%), syncope (22%), hemoptysis (5%) and unilateral leg complaints such as pain or swelling (38%).33,34 Exam findings depend on the severity of PE and include tachycardia (24%), crackles (18%), unilateral leg swelling (17%), fever (7%), and cyanosis (6%).33,34
Laboratory markers commonly utilized in PE include biochemical markers for right heart strain, including troponin and brain-natriuretic peptide (BNP). Troponin and BNP help evaluate for right-heart strain, due to a massive or submassive PE. D-dimer may be utilized to help rule-out PE in low risk patients, and D-dimer has a sensitivity of 97% for acute PE and a specificity of 39%.35 Caution must be utilized when interpreting D-dimer in the setting of PE versus possible COVID-19 infection, as COVID-19 may lead to a prothrombotic and hyperinflammatory state, leading to an increased D-dimer.
Imaging studies utilized in the evaluation of PE include CXR, US, and CT pulmonary angiogram. CXR findings are nonspecific and variable. Bedside US can be utilized to evaluate for signs of right heart strain (Figure 3). The gold standard for diagnosis is CT pulmonary angiography, which will also help further evaluate for pulmonary findings of acute COVID-19 infection.
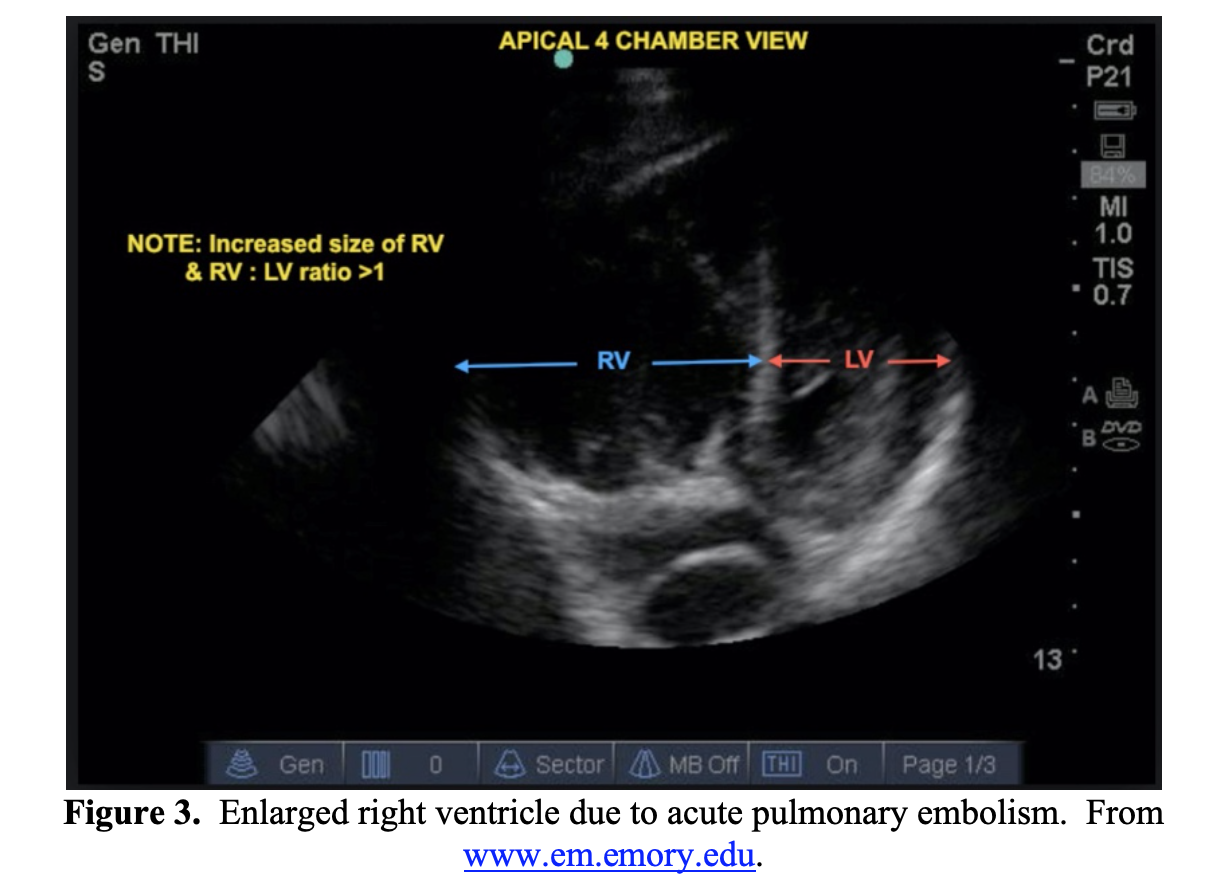
When evaluating the patient with possible COVID-19 with shortness of breath, consider the diagnosis of PE, especially if the patient has risk factors. Patients with severe COVID-19 infection may have a concurrent PE, further confounding the clinical differentiation between the two. Severe COVID-19 infection is thought to lead to both a hyperinflammatory and prothrombotic state, with an increase in DVT and PE in patients with COVID-19. Patients may present with concomitant PE with a severe COVID-19 infection, which would require a CTPA for evaluation.
Bacterial pneumonia
Pneumonia is an infection of the lung that involves the alveoli and may be caused by a bacterium, virus (including COVID-19), or fungus. Pneumonia is common and is the leading cause of infectious death for adults worldwide,36 and is the leading cause of sepsis and septic shock.37
Bacterial pneumonia may present identically to acute COVID-19 infection. Pneumonia may present with acute onset of cough (79-91%), fatigue (90%), fever (71-75%), dyspnea (67-75%), sputum production (60-65%), tachypnea (45-70%), and pleuritic chest pain (39-49%).38,39 Contrary to the classic manifestation of pneumonia, elderly patients more commonly present with atypical signs and symptoms.38,39 Over half of elderly patients present with altered mental status and one third present without fever.38 Other complaints associated with pneumonia include lightheadedness, malaise, weakness, headache, nausea and vomiting, and myalgias.38,39
Labs have limited utility in the diagnosis of pneumonia. Acute phase reactants, such as procalcitonin (PCT) and C-reactive protein (CRP), have not been shown to reliably distinguish between bacterial and non-bacterial pneumonia.38,40,41 A PCT of > 0.25 ng/mL has a sensitivity of 77-88% and specificity of 79-81% for sepsis, while a CRP > 40 mg/L has a sensitivity of 65-70% and specificity of 90% for community-acquired pneumonia.38,40,41 Unfortunately, both CRP and PCT may be elevated with COVID-19, especially in patients with severe illness burden, and likely do not have a role in distinguishing acute bacterial pneumonia from COVID-19 pneumonia.
CXR findings can vary greatly in patients with bacterial pneumonia. Streptococcus pneumoniae pneumonia classically presents with a lobar infiltrate.38,42 Other findings with various organisms include pleural effusions, basilar infiltrates, interstitial infiltrates, or abscesses.38,42 A chest radiograph depicting a lobar pneumonia is shown in Figure 4.
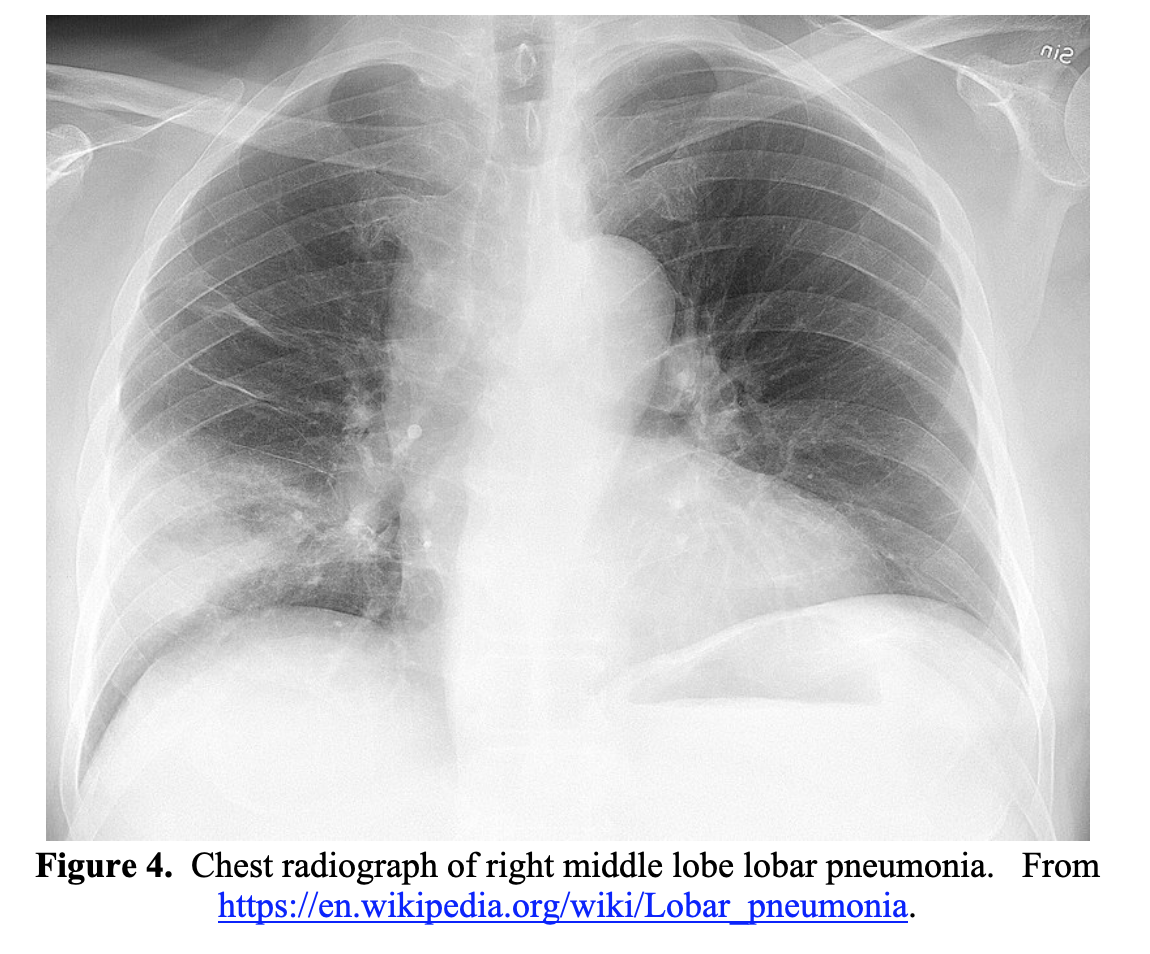
While evaluating a patient that may have COVID-19 or bacterial pneumonia, sepsis secondary to bacterial pneumonia must be considered. If the patient is ill appearing or has clinical or vital sign abnormalities consistent with sepsis, blood cultures should be drawn and broad-spectrum antimicrobial therapy rapidly initiated. If the patient appears clinically dehydrated and does not have significant CHF, liver or kidney disease, a bolus of intravenous crystalloid may be appropriate. However, if concerned for acute COVID-19 infection and the patient is ill appearing or has a chest X-ray with bilateral infiltrates, a 30cc/kg fluid bolus may exacerbate pulmonary injury and lead to clinical deterioration.
For more information on community-acquired pneumonia, see this previous emDOCs post on pneumonia.
Endocarditis
Endocarditis is an infection of the endocardial surface of the heart and can be incredibly challenging to diagnose, as it may present insidiously with vague complaints. The Emergency Physician must consider risk factors for infective endocarditis (IE) in order to consider and appropriately make the diagnosis. Risk factors for IE are shown in Table 4.43
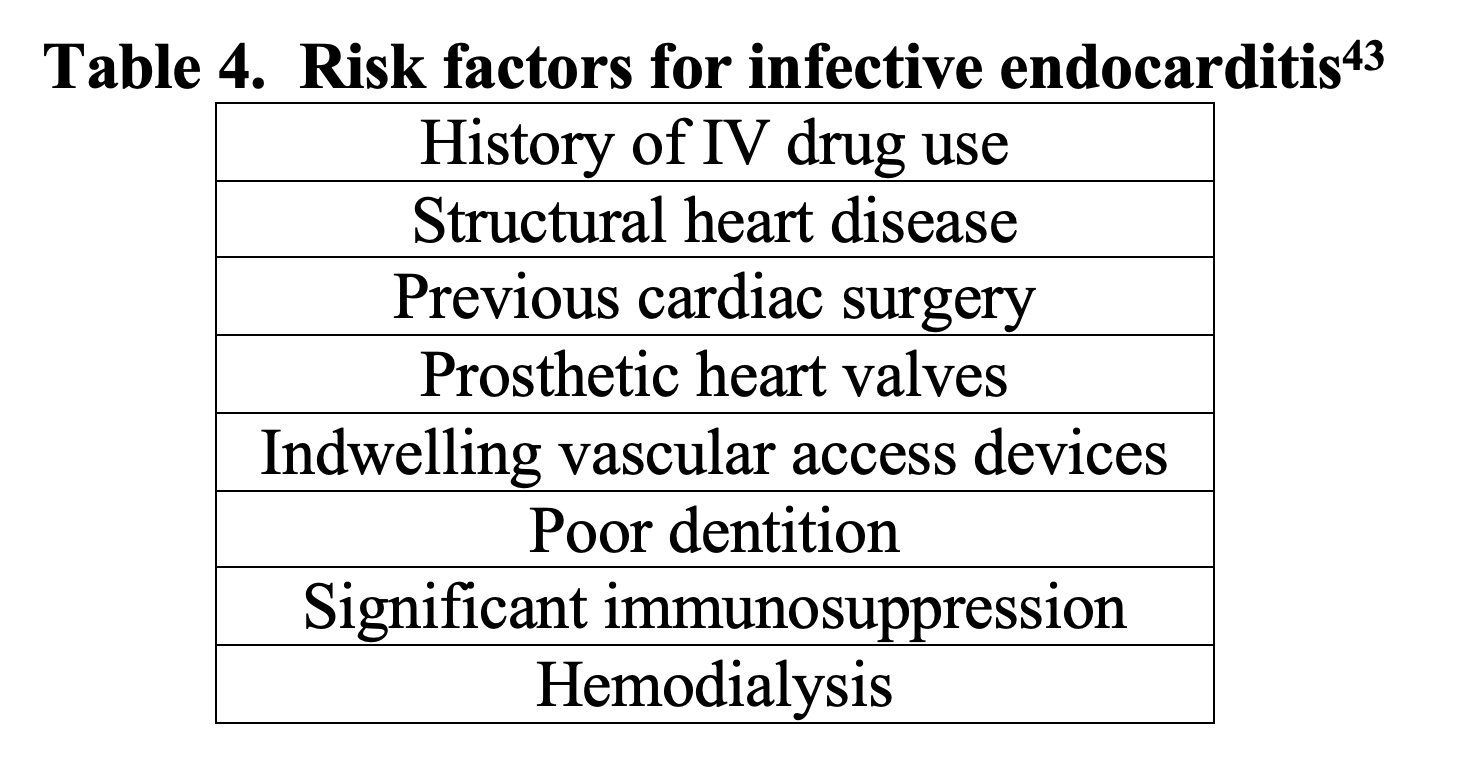

Diagnosis of IE is made with the Duke Criteria, which relies on positive blood cultures and echo findings consistent with IE.47 Blood cultures are a cornerstone of the diagnosis and are positive in over 95% of cases.44,45 If concern for endocarditis, the recommendation is to draw 3 sets of blood cultures, ideally drawn 30-60 minutes apart.48 After blood cultures have been drawn, broad spectrum intravenous antimicrobial therapy should be initiated. When evaluating a patient for possible COVID-19 infection, consider risk factors for IE, and if clinically concerned, obtain blood cultures and initiate broad spectrum antibiotics.
For more information on endocarditis, see this previous emDOCs post.
Heart Failure Exacerbation
Per the American Heart Association, an estimated 6.5 million Americans suffer from heart failure.49 Heart failure (HF) encompasses a wide spectrum of disease, and can include systolic or diastolic HF, acute or chronic HF, and left or right sided HF. Acute HF (AHF) exacerbations can mimic COVID-19 infections, as patients may primarily complain of dyspnea. AHF may occur with incidental COVID-19 infections or may occur in a patient without a previous history of heart disease as a result of severe COVID-19 infection.
AHF must be considered when any patient with a history of HF presents with respiratory complaints. Common risk factors for the development of HF include coronary artery disease, long-standing hypertension, diabetes, smoking, or valvular pathology.50 AHF commonly presents with worsening symptoms over several days to weeks with dyspnea associated with worsening lower extremity edema. Additionally, patients may complain of worsening exercise tolerance, orthopnea, paroxysmal nocturnal dyspnea, fatigue, chest discomfort, and weight gain.51,52 Patients with AHF may be tachycardic, and an S3 heart sound is very specific for AHF.51,52 Severe AHF exacerbations present with tachypnea, increased work of breathing, bilateral crackles on pulmonary auscultation, wheezing, or diminished air entry.51,52 They may have increased jugular venous distension and increasing lower extremity edema, which is usually bilateral and symmetric.51,52
B-type natriuretic peptide (BNP) or pro-BNP is often utilized to evaluate for AHF. A BNP < 100 or pro-BNP < 300 indicates that AHF is unlikely, with a negative LR of 0.1.52 BNP > 1000 or pro-BNP > 30,000 indicates that AHF is likely, with a positive LR of 6 and 3.3, respectively.52 However, BNP levels may be difficult to interpret in patients with chronic HF, as these patients have BNP levels that are chronically elevated. Additionally, BNP may also be elevated in conditions other than AHF, such as COPD, PE, or pulmonary hypertension, or any other condition that exerts stress on the right heart.
The CXR in AHF may include prominent interstitial markings, cardiomegaly, and pleural effusions.53 US is helpful in the evaluation of potential AHF and may show B-lines on pulmonary exam (Figure 6). Lung US alone with presence of greater than two B-lines in greater than two bilateral thoracic lung zones has a positive likelihood ratio of 7.4, with a sensitivity approaching over 90%, and a specificity of 92.7%.52,54-56 However, B lines may also be seen in acute COVID-19 infection, and this finding on US may be insufficient to differentiate the two conditions. US can also be utilized to evaluate the heart and may show a dilated heart with poor left ventricular function.

Therapy for AHF focuses on afterload and preload reduction with nitrates if the patient is hypertensive with pulmonary edema, in addition to noninvasive positive pressure ventilation (NIPPV) for patients with any respiratory distress. NIPPV is associated with a reduced rate of intubation (NNT of 8) and decreased mortality in patients with acute pulmonary edema from AHF (NNT of 13).57,58 In patients with more chronic overload, diuretics should be administered.
When considering AHF and acute COVID-19, there are several factors to consider. Preexisting heart failure or coronary artery disease may lead to high risk of severe COVID-19 infection and respiratory failure. Additionally, a patient may have AHF exacerbation secondary to severe COVID-19 infection. Zhou et al reported heart failure was found in 23% of patients with COVID-19 presentations.27 The Emergency Physician may not be able to reliably distinguish between AHF and COVID-19 infection in the ED, as patients may present with both pathologies. Given the effectiveness of nitrates and NIPPV in patients with AHF and respiratory distress, these therapies should not be withheld if there is a strong suspicion for AHF causing the patient’s presentation.
For more information on acute heart failure, see these two previous emDOCs posts: Heart Failure Myths: Part I – ED Eval, Heart Failure Myths: Part II – ED Management.
Chronic Obstructive Pulmonary Disease Exacerbation
Chronic Obstructive Pulmonary Disease (COPD) is common, affecting 5% of the population and is the 3rd most common cause of death in the United States after heart disease and cancer.59 Patients presenting with COPD exacerbations often have a history of COPD, smoking or smoke exposure, or are on home oxygen therapy.59 An exacerbation is defined by the Global Initiative for COPD (GOLD) as a change in baseline dyspnea, cough, and/or sputum production beyond daily variations and is acute in onset.60 70% of COPD exacerbations are triggered by a viral or bacterial etiology.59,61,62 Unfortunately, a COPD exacerbation may be impossible to distinguish from acute COVID-19 infection, as COVID-19 infection may lead to a COPD exacerbation.
COPD is a clinical diagnosis guided by the history and exam, but CXR may be helpful in excluding alternative diagnoses including bacterial or viral pneumonia or AHF. CXR in patients with COPD often shows radiolucent and hyperinflated lungs, flat diaphragm, and a narrow heart shadow.63 A sample chest radiograph is shown in Figure 7.

Treatment for COPD exacerbations include inhaled bronchodilators, supplemental oxygen, steroids, and respiratory support in the form of NIPPV.60,64,65 Antibiotics are utilized in moderate to severe exacerbations and have a limited role in mild exacerbations.66 While steroids are not recommended in early COVID-19 infection due to the fear of increased viral shedding, they are crucial in reducing lung inflammation associated with COPD exacerbations. The GOLD COVID-19 provides the following statement: “GOLD is not aware of any scientific evidence that inhaled or oral corticosteroids should be avoided in patients with COPD during the COVID-19 epidemic.”67
Similar to AHF, NIPPV has been associated with decreased mortality and need for intubation for patients in severe respiratory distress secondary to COPD exacerbations.65 For patients presenting with respiratory distress from a presumed COPD exacerbation, NIPPV should not be withheld even in current COVID-19 pandemic.
For more information on COPD, see this previous emDOCs post.
Acute Coronary Syndrome
Acute coronary syndrome (ACS) must be considered when evaluating the potential COVID-19 patient, as COVID-19 infection may rarely present primarily as chest pain. There is a case report of a patient in Italy presenting with chest pain and EKG changes concerning for acute myocardial infarction. On cardiac catheterization, the patient was free of obstructive coronary disease but tested positive for COVID-19.68
ACS may mimic COVID-19 as it may present atypically without chest pain. Up to one third of patients with ACS will have silent or atypical symptoms such as shortness of breath, malaise, fatigue, syncope, or nausea and vomiting.69,70 The GRACE trial found that 8.4% of patients presented without chest pain and over half of these patients complained primarily of dyspnea.71 Patients that more often present atypically are female patients, patients with diabetes, older age, or history of heart failure.72 In elderly patients, chest pain is only present in 50% of patients with ACS.73,74 See this emDOCs post for more information on missed myocardial infarctions.
When evaluating the patient for COVID-19, ACS versus COVID-19 infection may be difficult to differentiate, as severe COVID-19 infection may lead to myocardial injury.28 Elevated troponin levels may occur in 7-17% of patients hospitalized with COVID-19 and is a marker of severe illness.27,75 There have also been reports of fulminant myocarditis secondary to severe COVID-19 infection.76,77 It is also thought that the hyperinflammatory and prothrombotic state of COVID-19 may increase the risk of plaque rupture leading to acute coronary syndrome.27 It is imperative that part of the workup for the COVID-19 patient includes electrocardiogram and troponin, to both assess for ACS or myocardial injury secondary to acute COVID-19 infection.
According to the Journal of the American College of Cardiology, percutaneous coronary intervention remains the standard of care for acute myocardial infection, including for patients with confirmed COVID-19.78 If PCI is not available in the setting of a patient with acute MI, thrombolytics may be utilized. However, as acute COVID-19 infection can lead to perimyocarditis, it is prudent to first conduct a bedside cardiac ultrasound to evaluate for a pericardial effusion, as thrombolytic administration in this setting can lead to a hemorrhagic pericardial effusion and cardiac tamponade.79,80 When evaluating possible myocardial infarction in the setting of COVID-19, the case should be discussed with the cardiologist and joint decision making should be utilized to optimize patient care. See this emDOCs post for further information on managing the COVID-19 patient with acute MI.
Acute Respiratory Distress Syndrome
Acute Respiratory Distress Syndrome (ARDS) is defined as a condition of acute inflammatory lung injury that leads to non-cardiogenic pulmonary edema and respiratory failure. ARDS stems from diffuse alveolar injury and lung capillary endothelial injury, which leads to increased capillary permeability and pulmonary edema. The Berlin criteria is used for the Diagnosis of ARDS (Table 5).81,82

While COVID-19 may lead to ARDS, there are a multitude of underlying causes to consider. The most common cause of ARDS is sepsis.83 Other causes include aspiration pneumonitis, infectious pneumonia, severe trauma or multiple fractures, severe burns, smoke inhalation, pulmonary contusions, transfusion-associated acute lung injury, pancreatitis, drowning, thoracic or other major surgery, or drug exposures or overdose.83
ARDS commonly presents with respiratory distress, with patients often displaying significant dyspnea, tachycardia, tachypnea, and hypoxemia.84 Chest radiography is helpful in the diagnosis, which will show bilateral opacities (Figure 8).84
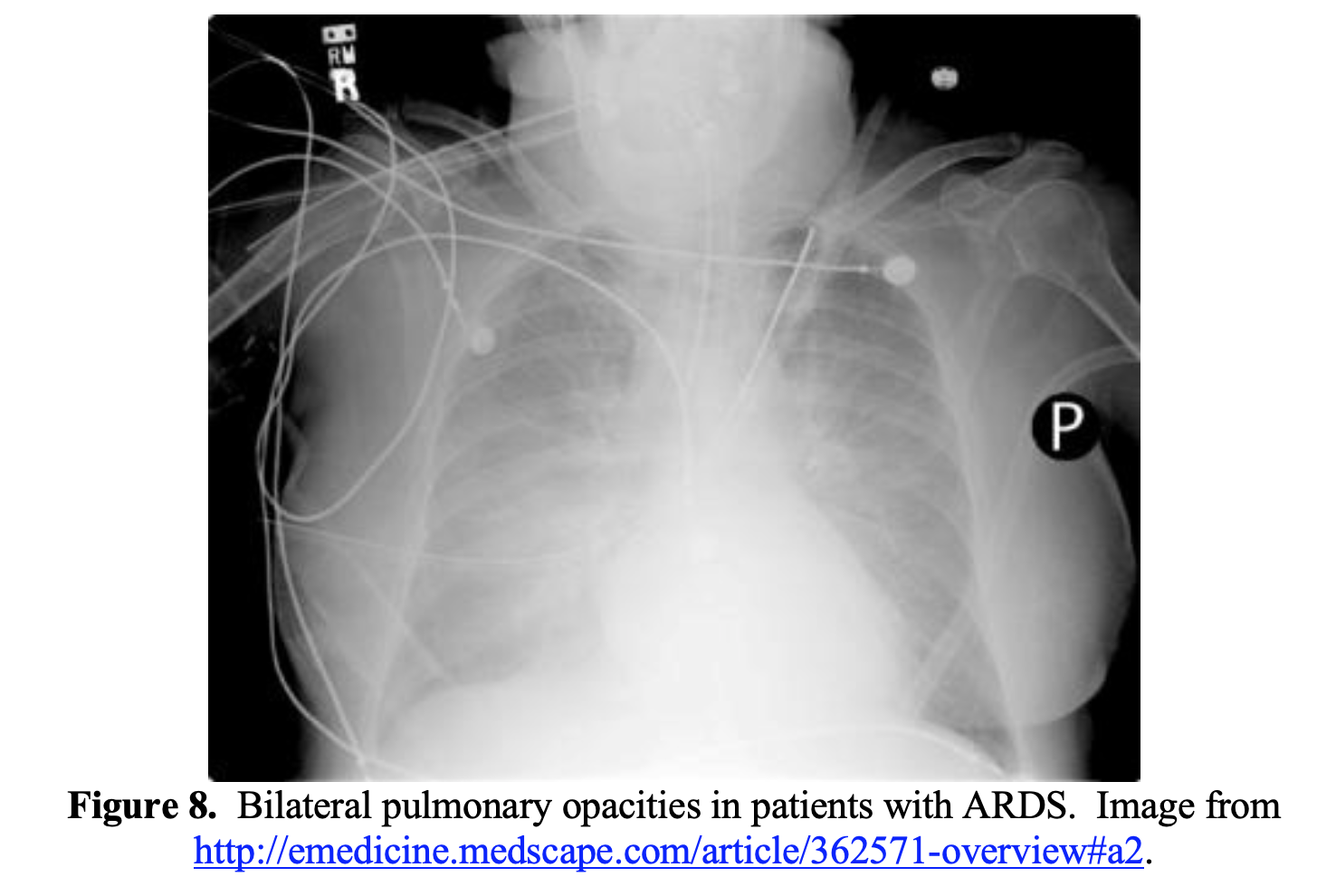
Two phenotypes for COVID-19 pneumonia have been proposed: Type L and Type H.85 The Type L phenotype is characterized by low elastance, normal compliance, and low ventilation to perfusion ratios.85 At this stage, the patient may display significant hypoxemia without marked respiratory distress. Patients may be more responsive to non-invasive ventilation options such as high-flow nasal cannula or NIPPV. If intubated, these patients can be ventilated with volumes greater than the classic 6 mL/kg, as their lungs are highly compliant. The Type H phenotype is characterized by high elastance (decreased gas volume due to increased pulmonary edema), high degree of right-to-left shunt, and high lung recruitability.85 Patients with Type H phenotype often represent classic ARDS: hypoxemia, bilateral infiltrates, and decreased compliance.85 Patients with Type H phenotype may be managed with respiratory support similar to other causes of ARDS, with a lung-protective ventilation strategy. However, other evidence suggests these phenotypes may not be present, and they should not form the foundation of your management.
See this emDOCs post for further information on ARDS.
Interstitial Lung Disease
Interstitial lung disease (ILD) includes a group of diffuse parenchymal lung diseases that cause progressive scarring of lung tissue.86 A broad categorization of ILD is idiopathic or secondary.86 Causes of idiopathic ILD include idiopathic pulmonary fibrosis (most common), acute interstitial pneumonia, cryptogenic organizing pneumonia, and several others.86,87 Secondary causes of ILD have a known etiology, some of which are listed in Table 6.
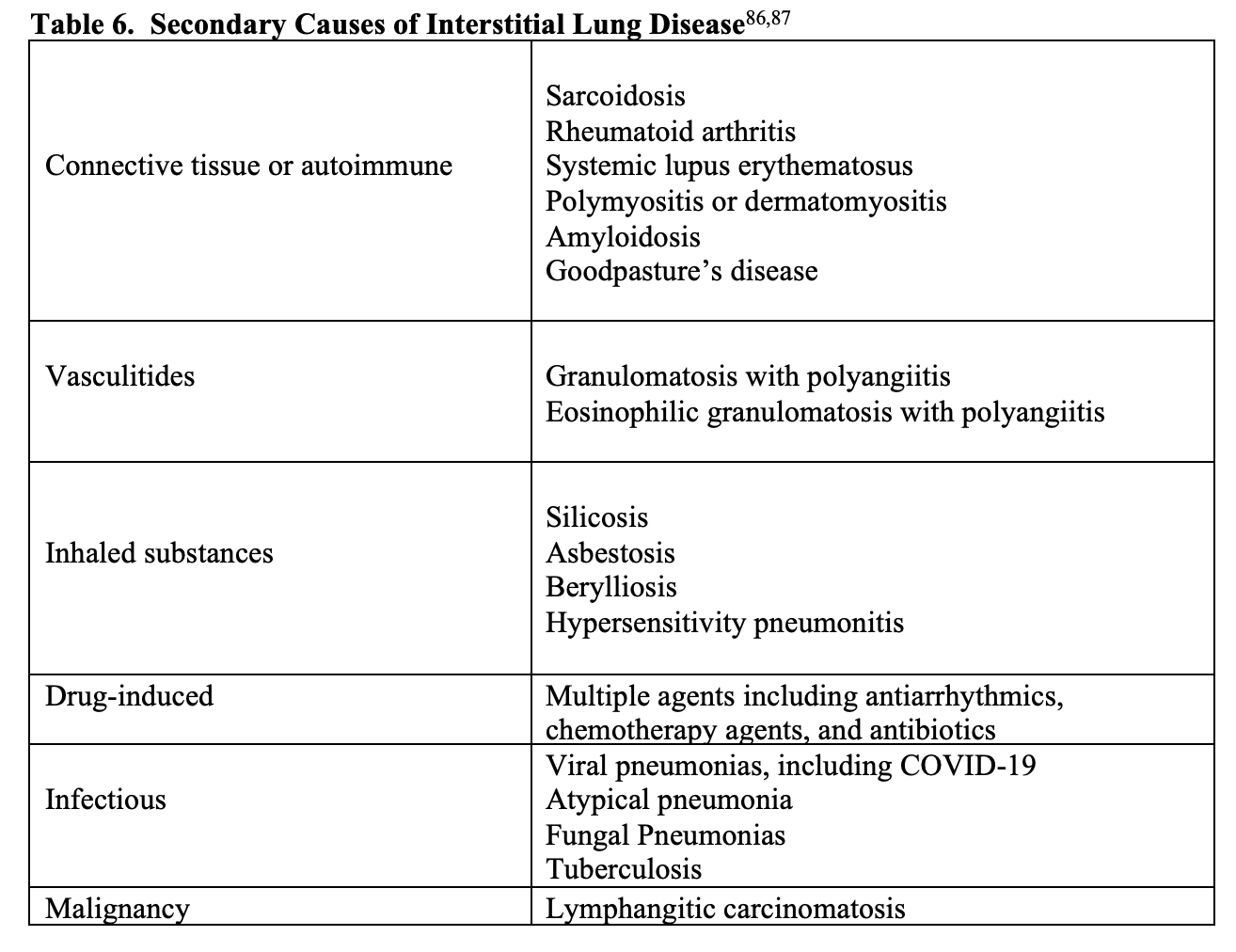
ILD often presents gradually over weeks to months, with worsening shortness of breath, dyspnea on exertion, persistent nonproductive cough.86 Patient may also have a history of occupational exposure, such as exposure to asbestosis or radiation therapy.86,88 There are numerous drugs that can lead to ILD, including methotrexate, azathioprine, rituximab, amiodarone, procainamide, hydralazine, isoniazid, and many others.89 Other important historical factors include long history of smoking, radiation therapy, or family history of ILD.86,87
The most common abnormality on CXR is a reticular pattern, but X-ray may also show a nodular pattern or increased interstitial marking.90 Chest CT is helpful for the diagnosis and can show multiple findings, including bilateral symmetric hilar adenopathy, reticular opacities, pleural plaques, centrilobular nodules, cysts, reticular opacities, and ground glass opacities.91
While ILD may present similarly to COVID-19, symptoms are likely to be present for a much longer period of time than the typical course of COVID-19. Historical factors, such as occupational exposure or exposure to radiation therapy, may also be helpful in distinguishing the two clinical entities.
Influenza/other respiratory viruses
Testing for influenza and other common respiratory viruses may be helpful when evaluating a patient for COVID-19. However, coinfection may occur with reported rates up to 20% of cases of COVID-19 having coexisting influenza infection.92 A patient having a positive test for influenza or a different respiratory virus does not rule out acute COVID-19 infection.
Case 1 Resolution
For the 62 year-old male with cough and shortness of breath, you do a bedside US exam, which is significant for multiple B-lines bilaterally in multiple thoracic fields. Cardiac US is notable for small pericardial effusion with an enlarged left ventricle and decreased ejection fraction. Concerned for an acute heart failure exacerbation, you start the patient on a high dose nitroglycerin drip and NIPPV. On re-evaluation 20 minutes later, the patient’s work of breathing is significantly improved, and he feels much better. Prior to admission, you send a rapid COVID-19 test, which is negative, and the patient is admitted for an acute heart failure exacerbation.
Case 2 Resolution
On re-examination of the 36 year-old female with fever and shortness of breath, you note multiple track marks on her arms, and she admits she had a relapse and recently used intravenous heroin. Concerned for endocarditis given her fever and multiple complaints, you tell the nurses to get 3 sets of blood cultures over the next hour and order broad spectrum antibiotics to begin after blood cultures have been obtained. Chest X-ray is concerning for septic pulmonary emboli, and after a negative rapid COVID-19 test the patient is admitted to the ICU with the cardiology and infectious disease services following.
References
- Lu H, Stratton CW, Tang YW. Outbreak of pneumonia of unknown etiology in Wuhan China: the mystery and the miracle. J Med Virol. 2020 Apr;92(4):401-402.
- Wang D, Hu B, Hu C, et al. Clinical characteristic of 138 hospitalized patients with novel coronavirus-infected pneumonia in Wuhan, China. 2020 Feb 7.
- Del Rio C, Malani PN. COVID-19-New Insights on a Rapidly Changing Epidemic JAMA. 28 Feb 2020.
- Centers for Disease Control and Prevention. Coronavirus Disease 2019 (COVID-19): Cases in the US. https://www.cdc.gov/coronavirus/2019-ncov/cases-updates/cases-in-us.html. Accessed 26 April 2020.
- Johns Hopkins Coronavirus Resource Center. COVID-19 Dashboard by the Center for Systems Science and Engineering. https://coronavirus.jhu.edu/map.html. Accessed 26 April 2020.
- Van Doremalen N, Bushmaker T, Morris DH, et al. Aerosol and Surface Stability of SARS-CoV-2 as Compared with SARS-CoV-1. N Eng J Med. 16 April 2020.
- Lauer SA, Grantz KH, Bi Q, et al. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Ann Intern Med. 2020 Mar 10.
- 2019 Novel Coronavirus (2019-nCoV) clinical care. Centers for Disease Control and Prevention. https://www.cdc.gov/coronavirus/2019-ncov/hcp/clinical-guidance-management-patients.html. Published February 11, 2020. Accessed March 30, 2020.
- Sanche S, Lin YT, Xu C, et al. The novel coronavirus, 2019-nCoV, is highly contagious and more infectious than initially estimated. arXiv. 9 Feb 2020.
- Wu Z, McGoogan JM. Characteristics of and Important Lessons from the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72,314 Cases From the Chinese Center for Disease Control and Prevention. 2020 Feb 24.
- National Center for Immunization and Respiratory Diseases (NCIRD), Division of Viral Diseases. Coronavirus Disease 2019 (COVID-19). People at Risk for Serious Illness from COVID-19. CDC 24-7 Organization website.https://www.cdc.gov/coronavirus/2019-ncov/specific-groups/high-risk-complications.html. Updated March 10, 2020. Accessed April 11, 2020.
- Huang C, Wang Y, Li X, et al.Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. 2020 Feb 15;395(10223):497-506.
- Guan WJ, Ni ZY, Hu Y, et al. Clinical Characteristics of Coronavirus Disease 2019 in China [published online ahead of print, 2020 Feb 28]. N Engl J Med. 2020;10.1056/NEJMoa2002032.
- Xie J, Tong Z, Guan X, et al. Critical care crisis and some recommendations during the COVID-19 epidemic in China. Int Care Med. 2 March 2020.
- Siddiqi HK, Mehra MR. COVID-19 Illness in native and immunosuppressed states: a clinical-therapeutic staging proposal. Journal of Heart and Lung Transplantation. 3 March 2020.
- Chen N, Zhou M, Dong X, et al. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. 30 January 2020.
- Pan L, Mu M, Yang P, et al. Clinical characteristics of COVID-19 patients with digestive symptoms in Hubei, China: a descriptive, cross-sectional, multicenter study. Am J Gastroenterol. Accessed April 18, 2020.
- American Academy of Otolaryngology – Head and Neck Surgery Website. AAO-HNS: Anosmia, Hyposmia, and Dysgeusia symptoms of coronavirus disease. Updated March 23, 2020. Accessed April 23, 2020.
- Ruan Q, Yang K, Wang W, et al. Clinical predictors of mortality due to COVID-19 based on an analysis of data of 150 patients from Wuhan, China. Int Care Med. 03 March 2020.
- Wong HYF, Lam HYS, Fong AH, et al. Frequency and Distribution of Chest Radiographic Findings in COVID-19 Positive Patients. Radiology. 2019 Mar 27:201160.
- Rodrigues, J.C.L. et al. An update on COVID-19 for the radiologist – A British society of Thoracic Imaging statement. Clin Radiol. 2020 May;75(5):323-325. Epub 2020 Mar 23.
- Li Y, Xia L. Coronavirus Disease 2019 (COVID-19): Role of chest CT in diagnosis and management. AJR AM J Roentgenol. 2020 Mar 4:1-7.
- Feng P, Ye T, Peng S, et al. Time course of lung changes on chest CT during recovery from 2019 novel coronavirus (COVID-19) pneumonia. 13 Feb 2020.
- Weerakkody Y, Bell DJ, et al. COVID-19. https://radiopaedia.org/articles/covid-19-3. Accessed 25 April 2020.
- Miller WT, Shah RM. Isolated diffuse ground-glass opacity in thoracic CT: causes and clinical presentations. AJR Am J Roentgenol. 2005;184 (2): 613-22.
- Huang Y, Wang S, Liu Y, et al. A preliminary study on the ultrasonic manifestations of peripulmonary lesions of non-critical vovel coronavirus pneumonia (COVID-19). Clin Radiol. 2020 Feb 26.
- Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. 2020 Mar 28;395(10229):1054-1062.
- Driggin E, Madhavan MV, Bikdeli B, et al. Cardiovascular considerations for patients, health care workers, and health systems during the coronavirus disease 2019 (COVID-19) pandemic. J Am Coll Cardiol. 2020 Mar 18.
- Lippi G, Plebani M. Procalcitonin in patients with severe coronavirus disease 2019 (COVID-19): A meta-analysis. Clin Chim Acta. Epub 2020 Mar 4.
- Ai T, Yang Z, Hou H, et al. Correlation of chest CT and RT-PCR testing in coronavirus disease 2019 (COVID-19) in China: A report of 1014 cases. Radiology. 2020;200642.
- Wang W, Xu Y, Gao R, et al. Detection of SARS-CoV-2 in different types of clinical specimens. JAMA.11 March 2020.
- Anderson FA Jr, Spencer FA. Risk factors for venous thromboembolism. 2003;107(23 Suppl 1):I9–I16.
- Miniati M, Prediletto R, Formichi B, et al. Accuracy of clinical assessment in the diagnosis of pulmonary embolism. Am J Respir Crit Care Med. 1999;159(3):864–871.
- Arora S, Menchine M. EMA July 1999: Accuracy Of Clinical Assessment In The Diagnosis Of Pulmonary Embolism. EM:RAP. https://www.emrap.org/episode/ema-1999-7/abstract37. Updated September 20, 2017. Accessed April 15, 2020.
- Lim W, Le Gal G, Bates SM, et al. American Society of Hematology 2018 guidelines for management of venous thromboembolism: diagnosis of venous thromboembolism. Blood Adv. 2018;2(22):3226–3256.
- The top 10 causes of death. World Health Organization website.https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death. Accessed April 15, 2020.
- American Thoracic Society. Top 20 pneumonia facts – 2018.https://www.thoracic.org/patients/patient-resources/resources/top-pneumonia-facts.pdf. Accessed April 15, 2020.
- Long B, Long D, Koyfman A. Emergency Medicine Evaluation of Community-Acquired Pneumonia: History, Examination, Imaging and Laboratory Assessment, and Risk Scores. J Emerg Med. 2017;53(5):642–652.
- Metlay JP, Fine MJ, Schulz R, et al. Measuring symptomatic and functional recovery in patients with community-acquired pneumonia. J Gen Intern Med. 1997;12(7):423–430.
- Wunderink RG, Waterer G. Advances in the causes and management of community acquired pneumonia in adults. BMJ. 2017 Jul 10;358:j2471.
- Kalil AC, Metersky ML, Klompas M, et al. Management of Adults With Hospital-acquired and Ventilator-associated Pneumonia: 2016 Clinical Practice Guidelines by the Infectious Diseases Society of America and the American Thoracic Society [published correction appears in Clin Infect Dis. 2017 May 1;64(9):1298] [published correction appears in Clin Infect Dis. 2017 Oct 15;65(8):1435] [published correction appears in Clin Infect Dis. 2017 Nov 29;65(12):2161]. Clin Infect Dis. 2016;63(5):e61–e111.
- Franquet T. Imaging of pneumonia: trends and algorithms. Eur Respir J. 2001 Jul;18(1):196-208.
- Schauer SG, Pfaff JA, Cuenca PJ. Emergency department management of acute infective endocarditis. Emerg Med Pract. 2014 Nov;16(11):1-17; quiz 17-8. Epub 2014 Nov 1.
- Cahill TJ, Prendergast BD. Infective endocarditis. Lancet. 2016 Feb 27;387(20021):882-93.
- Crawford MH, Durack DT. Clinical presentation of infective endocarditis. Cardiol Clin. 2003 May;21(2):159-66.
- Wang A, Gaca JG, Chu VH. Management considerations in infective endocarditis: A review. JAMA. 2018 Jul 3;320(1):72-83.
- Durack DT, Lukes AS, Bright DK. New criteria for diagnosis of infective endocarditis: utilization of specific echocardiographic findings. Duke Endocarditis Service. Am J Med. 1994 Mar;96(3):200-9.
- Lee A, Mirrett S, Reller LB, Weinstein MP. Detection of bloodstream infections in adults: how many blood cultures are needed?. J Clin Microbiol. 2007 Nov;45(11):3546-8. Epub 2007 Sep 19.
- Benjamin EJ, Blaha MJ, Chiuve SE, et al. Heart Disease and Stroke Statistics-2017 Update: A Report From the American Heart Association [published correction appears in Circulation. 2017 Mar 7;135(10 ):e646] [published correction appears in Circulation. 2017 Sep 5;136(10 ):e196]. Circulation. 2017;135(10):e146–e603.
- Ho KK, Pinsky JL, Kannel WB, Levy D. The epidemiology of heart failure: the Framingham Study. J Am Coll Cardiol. 1993 Oct;22(4 Suppl A):6A-13A.
- Jessup M, Brozena S. Heart failure. N Engl J Med. 2003 May 15;348(30):2007-18.
- Martindale JL, Wakai A, Collins SP, et al. Diagnosing Acute Heart Failure in the Emergency Department: A Systematic Review and Meta-analysis. Acad Emerg Med. 2016 Mar;23(3):223-42.
- Mueller-Lenke N, Rudez J, Staub D, et al. Use of chest radiography in the emergency diagnosis of acute congestive heart failure. Heart. 2006;92(5):695–696.
- Jambrik Z, Monti S, Coppola V, et al. Usefulness of ultrasound lung comets as a nonradiologic sign of extravascular lung water. Am J Cardiol. 2004;93(10): 1265–70.
- Mallamaci F, Benedetto FA, Tripepi R, et al. Detection of pulmonary congestion by chest ultrasound in dialysis patients. JACC Cardiovasc Imaging. 2010;3(6): 586–94.
- Anderson KL, Fields JM, Panebianco NL, et al. Inter-rater reliability of quantifying pleural B-lines using multiple counting methods. J Ultrasound Med. 2013;32(1): 115–20.
- Marik PE, Flemmer M. Narrative review: the management of acute decompensated heart failure. J Intensive Care Med. 2012;27:343–53.
- Vital FMR, Saconato H, Ladeira MT, et al. Non-invasive positive pressure ventilation (CPAP or bilevel NPPV) for cardiogenic pulmonary edema. Cochrane database of systematic reviews; 2008 Issue 3. No.: CD005351
- Mannino DM, Watt G, Hole D, et al. The natural history of chronic obstructive pulmonary disease. Eur Respir J. 2006;27(3):627–643.
- Global Initiative for Chronic Obstructive Lung Diseases. Global strategy for the diagnosis, management and prevention of chronic obstructive pulmonary disease: 2019 report.https://goldcopd.org/wp-content/uploads/2018/11/GOLD-2019-v1.7-FINAL-14Nov2018-WMS.pdf. Accessed April 20, 2020.
- Wilson R. Bacteria and airway inflammation in chronic obstructive pulmonary disease: more evidence. Am J Respir Crit Care Med. 2005;172(2):147–148.
- Wu X, Chen D, Gu X, Su X, Song Y, Shi Y. Prevalence and risk of viral infection in patients with acute exacerbation of chronic obstructive pulmonary disease: a meta-analysis. Mol Biol Rep. 2014;41(7):4743–4751.
- O’brien C, Guest PJ, Hill SL et-al. Physiological and radiological characterization of patients diagnosed with chronic obstructive pulmonary disease in primary care. Thorax. 2000;55 (8): 635-42.
- Nair S, Thomas E, Pearson SB, Henry MT. A randomized controlled trial to assess the optimal dose and effect of nebulized albuterol in acute exacerbations of COPD. Chest. 2005;128(1):48–54. doi:10.1378/chest.128.1.48
- Ram FS, Picot J, Lightowler J, Wedzicha JA. Non-invasive positive pressure ventilation for treatment of respiratory failure due to exacerbations of chronic obstructive pulmonary disease. Cochrane Database Syst Rev. 2004;(3):CD004104.
- Miravitlles M, Moragas A, Hernández S, Bayona C, Llor C. Is it possible to identify exacerbations of mild to moderate COPD that do not require antibiotic treatment?. Chest. 2013;144(5):1571–1577.
- Global Initiative for Chronic Obstructive Lung Diseases. GOLD COVID-19 Guidance. https://goldcopd.org/gold-covid-19-guidance/. Accessed April 20, 2020.
- Wood S. TCT the Heat Beat: COVID-19 and the Heart: Insights from the Front Lines. https://www.tctmd.com/news/covid-19-and-heart-insights-front-lines. Accessed April 15, 2020.
- Brieger D, et al. Acute coronary syndromes without chest pain, an underdiagnosed and undertreated high-risk group. Insights from the Global Registry of Acute Coronary Events. Chest 2004;126(2):461-9.
- Dorsh MF, et al. Poor prognosis of patients presenting with symptomatic myocardial infarction but without chest pain. Heart 2001;86(5):494-8.
- Fox KA, Goodman SG, Klein W, et al. Management of acute coronary syndromes. Variations in practice and outcome; findings from the Global Registry of Acute Coronary Events (GRACE). Eur Heart J. 2002;23:1177-89.
- Body R, McDowell G, Carley S, et al. Do risk factors for chronic coronary heart disease help diagnose acute myocardial infarction in the Emergency Department? Resuscitation. 2008;79:41-45.
- Alexander KP, et al. Acute coronary care in the elderly, part I: Non-ST-segment elevation acute coronary syndromes. Circulation. 2007;115:2549-2569.
- Samaras, N, Chevalley, T, et al. Older patients in the emergency department: a review. Ann Emerg Med. 2010;56:261-269.
- Zhou P, Yang XL, Wang XG et al. A pneumonia outbreak associated with a new coronavirus of probable bat origin. Nature. 2020 Mar;579(7798):270-273.
- Liu K, Fang YY, Deng Y et al. Clinical characteristics of novel coronavirus cases in tertiary hospitals in Hubei Province. Chin Med J (Engl). 2020 Feb 7.
- Xu Z, Shi L, Wang Y et al. Pathological findings of COVID-19 associated with acute respiratory distress syndrome. Lancet Respir Med. 2020 Apr;8(4):420-422.
- Mahmud E, Dauerman HL, Welt FGP, et al. Management of acute myocardial infarction during the COVID-19 pandemic. J Am Coll Cardiol. Apr 2020.
- Inciardi RM, Lupi L, Zaccone G, et al. Cardiac involvement in a patient with coronavirus disease 2019 (COVID-19). JAMA. 27 Mar 2020.
- Li K, Wu J, Li C et al. The clinical and chest CT features associated with severe and critical COVID-19 pneumonia. Invest Radiol. 29 Feb 2020.
- The ARDS Definition Task Force. Acute Respiratory Distress Syndrome: The Berlin Definition. JAMA. May 21, 2012.
- Ferguson ND, Fan E, Camporota L, et al. The Berlin definition of ARDS: an expanded rationale, justification, and supplementary material. Intensive Care Med. 2012;38:1573.
- Hudson LD, Milberg JA, Anardi D, Maunder RJ. Clinical risks for development of the acute respiratory distress syndrome. Am J Respir Crit Care Med. 1995;151:293.
- Fan E, Brodie D, Slutsky AS. Acute Respiratory Distress Syndrome: Advances in diagnosis and treatment. JAMA. 2018 Feb 20;319(7):698-710.
- Gattinoni L, Chiumello D, Caironi P, et al. COVID-19 pneumonia: different respiratory treatment for different phenotypes? Int Care med. Apr 2020.
- Schwartz M, King Jr TE. Interstitial Lung disease, 5th ed, People’s Medical Clearing House, Shelton, CT 2011.
- Raghu G, Collard HR, Egan JJ, et al. An official ATS/ERS/JRS/ALAT statement: idiopathic pulmonary fibrosis: evidence-based guidelines for diagnosis and management. Am J Respir Crit Care Med. 2011;183:788.
- Glazer CS, Newman LS. Occupational interstitial lung disease. Clin Chest Med. 2004;25:467.
- Camus P. Drug-induced interstitial lung disease: In: Interstitial Lung Disease, 4th ed, King TE Jr, Schwarz MI (Eds), B.C. Decker, Hamilton, ON, Canada 2003. P485.
- Mueller-mang C, Grosse C, Schmid K et-al. What every radiologist should know about idiopathic interstitial pneumonias. Radiographics. 27 (3): 595-615.
- Ferguson EC, Berkowitz EA. Lung CT: Part 2, The interstitial pneumonias-clinical, histologic, and CT manifestations. AJR Am J Roentgenol. 2012;199 (4): W464-76.
- Wang M, Wu Q, Xu W, et al. Clinical diagnosis of 8274 samples with 2019-novel coronavirus in Wuhan. medRxiv. 2020 Jan 1.








