Author: Rachel Bridwell, MD (@rebridwell, EM Resident Physician, San Antonio, TX) // Edited by: Brit Long, MD (@long_brit, EM Attending Physician, San Antonio, TX) and Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital)
Welcome to EM@3AM, an emDOCs series designed to foster your working knowledge by providing an expedited review of clinical basics. We’ll keep it short, while you keep that EM brain sharp.
A 65-year-old male with a history of paroxysmal atrial fibrillation, peripheral vascular disease, and HTN presents to the ED with diffuse abdominal pain. The pain started after dinner last night and has progressed since with nausea and emesis. He’s previously had these symptoms after eating, but they have usually resolve. The patient is not currently on any anticoagulation, but takes HCTZ-lisinopril. Social history pertinent for 48 ppy tobacco use. ROS pertinent for noted blood in stool.
Exam: Vitals: HR 112, BP 98/61, RR 22, T 101.1, SpO2 98% on RA
General: Older male, in mild distress; CV: Tachycardic, irregularly irregular, no murmur, 2+ pulses in UE, 1+ in LE BL; Abdomen: Diminished BS, extremely tender diffusely with light palpation; Integumentary: Lower extremities without hair; Rectal: Bloody stool
What do you need to consider, and what’s the next step in your evaluation and treatment?
Answer: Mesenteric Ischemia1-21
Epidemiology:
- 0.09% incidence annually1
- Mortality within first 24 hours 60-80%1–3
- Time is crucial in mortality
- Mortality 70% if time to diagnosis > 24 hours4
- Mortality 14% if surgical intervention without 12 hours of symptom onset3,5
- Comorbidity Prevalence:
- Peripheral Arterial Disease 27%6
- Coronary Artery Disease 46%6
- HTN, HLD, DM, and obesity also common7
- 14%/year risk of mesenteric ischemia secondary to atrial fibrillation CITE (2.3%/year stroke risk)
Anatomy and Physiology:
- Superior mesenteric artery (SMA) most commonly affected for embolic events secondary to 45 degree angle departure from aorta, which perfuses duodenum to transverse colon4
- Infarction occurs from mucosa and propagates outward through muscularis and serosa8
- Pain starts viscerally, only affecting mucosa and once the muscularis and serosal layers are ischemic, the classic pain out of proportion peritonitic findings occur9
- Patients may present without pain due to hypoperfused intramural nociceptors9
- Bowel: high bacterial load and poor blood supply, and during gut ischemia, entry of digestive enzymes into gut wall10
Etiologies: Four categories
- Acute mesenteric arterial embolus: 40-50% cases, consider atrial fibrillation and endocarditis as sources7,9
- Mesenteric Thrombosis: 20-35% cases, thrombosis of previously atherosclerotic vessel7,9
- MI of the gut
- Dissection or inflammation in<5% cases7
- Mesenteric Venous Thrombosis: 5-15% cases, secondary to hypercoagulable state: Antithrombin-III, protein C or S deficiency, Factor V Leiden, pancreatitis, trauma, polyarteritis nodosa4,6,7,9,11,12
- Non-occlusive: 5-15% cases, occurs secondary to low perfusion states, both a cause of sepsis and result of sepsis, any hypoperfusion, CHF, recent surgery, hemodialysis2,4,5,7,9,13,14
Clinical Presentation: sensitivity of the following presenting signs and symptoms1
- Abdominal pain: 60-100%
- GI bleeding: 12-48%
- Diarrhea: 18-48%
- Nausea and vomiting: 39-93%
- Post prandial abdominal pain 30-60 minutes7
- Food avoidance7
- Hypotension: 5.2-54%
- Tachycardia: 31%
Evaluation:
- Assess ABCs and VS—may be normal on early presentation but will progress to tachycardia, hypotension, and fever.13
- Perform a complete physical examination:
- Abdominal: absent bowel sounds, pain out of proportion to exam only 45-54% sensitive1
- Vascular: Signs and symptoms of peripheral vascular disease
- CV: New murmur (endocarditis with septic emboli), irregularly irregular (Afib)
- Rectal exam: Guaiac positive stool 5.9-23% sensitive1
- Laboratory evaluation:
- CBC, CMP, ECG, L-Lactate, D-dimer
- Academic pursuits: D-lactate (bacterial lactate production), intestinal fatty acid binding protein, which is 90% sensitive and 89% specific15
- Most common lab abnormality is leukocytosis, 75% of patients will have a WBC count exceeding 15,000, with a sensitivity of 80% and specificity of 50%6,12
- Lactate: pool SN and SP: 73-95% and 32-55%1
- D-dimer: pooled SN and SP, 89-99% and 33-47% 1
- Elevated pancreatic amylase in 27% cases, and normal lactate in 48% of cases13
- Troponin: 47% of patients with troponin I above clinical level13
- Associated with increased cardiology consult, which was associated with poorer outcomes13
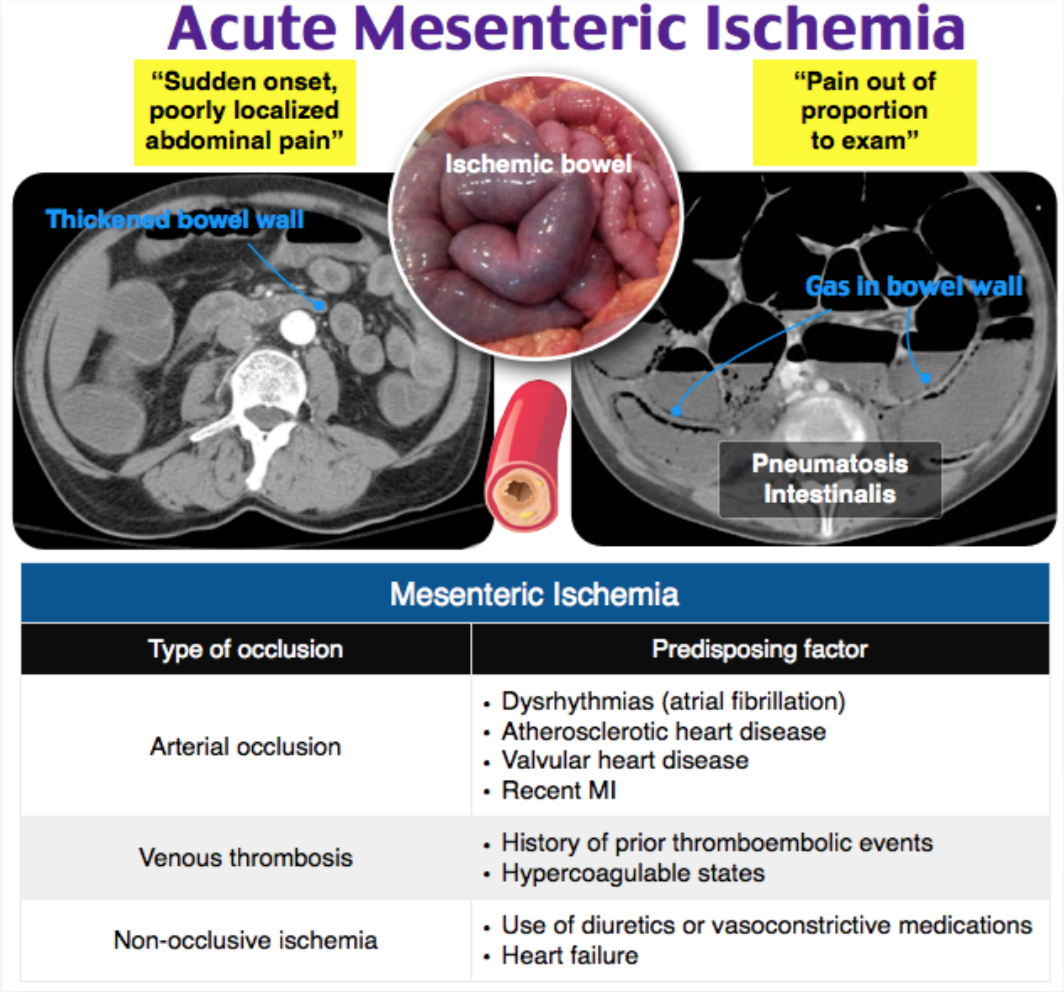
- Plain film: Consider plain films if patient too unstable for transport to CT. May see: bowel dilation, thumb printing, ileus, which is often misinterpreted as mechanical bowel obstruction), though pneumatosis in severe cases.16
- Normal AXR does not rule out mesenteric ischemia, 25% of patients with AMI have a normal AXR16
- Ultrasound:
- Arterial thrombus: Doppler assessment of celiac artery and SMA as they take off from aorta—Sensitivity 92-100%, specificity 70-89%16
- Non-occlusive thrombus or distal occlusion: Limited utility 17
- Venous thrombus: Portal or SMV thrombus, thickening of bowel wall, decreased peristalsis, or very late intraluminal air18
- CT Angiography:
- Pooled sensitivity and specificity 90-97% and 93-97% respectively, +LR 17.5(5.99-51.29) and –LR 0.09 (0.05-0.17)1
- Early non-specific CT Findings: Ascites, bowel wall thickening, mesenteric edema12
- Late CT Findings: portal gas, pneumoperitoneum, pneumatosis12
- Protocoling: Speak with radiologist in order to protocol the etiology for which you are concerned.
- Venogram – if suspicion of venous thrombosis
- Arteriogram – if suspicion of arterial emboli
- Triple phase (plain, venous and arterial phase CTs) – increased sensitivity for mesenteric ischemia; but, increased radiation exposure
Treatment:
- NPO and fluid resuscitate with crystalloid and correct electrolyte and base deficit7
- Profound capillary leak and third spacing
- Broad spectrum antibiotics: ceftriaxone and metronidazole or piperacillin/tazobactam19
- Associated with improved outcomes in critically ill patients7
- If persistently hypotensive after fluid resuscitation, vasopressors
- Very tricky with gut ischemia as many vasopressors cause splanchnic vasoconstriction20
- Dobutamine, low dose dopamine, and milrinone have decreased splanchnic vasoconstriction as compared to epinephrine and phenylephrine20
- Ask surgeons if they want heparin started
- If mesenteric venous thrombosis confirmed on CT venography, systemically anticoagulate with unfractionated heparin8
- Postoperative intra-arterial papaverin infusion into SMA to reduce vasospasm2
- Very tricky with gut ischemia as many vasopressors cause splanchnic vasoconstriction20
Disposition:
- Early surgical consultation
- AMI secondary to SMV thrombosis => heparin infusion and to MICU
- May involve IR for endovascular thrombectomy
- Endovascular thrombectomy success rates vs open
- In a review or 23,744 cases with matched severity of illness and comorbidity, increased survival with endovascular therapy versus open 39.3% vs 14.9%3
- In a case series of 70 consecutive patients, endovascular thrombectomy has been reported to have 87% technical success rate and decreased the amount of bowel resected (52 cm to 160 cm)21
- Further increases the risk of requiring TPN3
- Bowel resection reduces mortality to 30% though the need for bowel resection reflects the degree of ischemia and was found to be an independent mortality predictor3,5
- Increased rates of ARDS and lactic acidosis in open repair versus endovascular3
- Endovascular thrombectomy success rates vs open
Pearls:
- Early surgical consultation
- AMI secondary to SMV thrombosis => heparin infusion and admission to MICU
- May involve IR for endovascular thrombectomy
- Force function to consider with every older patient with abdominal pain, infrequent but very dangerous disease
- No one laboratory test can rule in or rule out mesenteric ischemia
- Always consider cardiovascular and thromboembolism risk factors in these patients
- Often with subacute onset
- Deceptive 3-6 hour periods with pain as pain receptions are hypoperfused9
- CT Angiography is the imaging modality of choice
- Be sure to optimize contrast protocol (triple phase CT and specify concern to radiologist)
- Acidosis is a univariant marker of mortality in these patients7
Pitfalls:
- Normal D-dimer—makes less likely but does not rule out
- Do not be reassured by a normal lactate—cannot rule in or out
- Elevated pancreatic amylase commonly seen in AMI, may be red herring to delay or miss diagnosis
- Elevated troponin in AMI with known cardiovascular risk factors, which may delay dx
- Increased morbidity and mortality when funnel disposition to inappropriate consultant13

A 76-year-old man with a history of atrial fibrillation, hypertension and prior stroke presents with acute onset of diffuse abdominal pain and vomiting. On examination, he is tachycardic and yelling in pain but his abdomen is soft with no rebound or guarding. An acute abdominal series shows pneumatosis intestinalis. Which of the following is the most likely site of acute mesenteric arterial occlusion?
A) Celiac trunk
B) Inferior mesenteric artery
C) Left colic artery
D) Superior mesenteric artery
Answer: D
The majority of cases of acute mesenteric ischemia are the result of acute arterial occlusion resulting from embolization from the heart. Risk factors include dysrhythmias (particularly atrial fibrillation), atherosclerosis, valvular heart disease and recent myocardial infarction. The superior mesenteric artery is most frequently affected in acute arterial occlusion. This artery supplies the distal half of the duodenum to the proximal two-thirds of the transverse colon. It is likely the most commonly involved artery due to its large caliber and narrow takeoff angle from the aorta. In addition to an acute embolic event, mesenteric ischemia can be due to progression of atherosclerotic disease with arterial or venous thrombosis, or nonocclusive ischemia due to hypoperfusion or vasospasm. Patients with acute mesenteric ischemia due to arterial embolism present with sudden onset diffuse abdominal pain, vomiting and diarrhea. Early on, the pain is out of proportion to physical exam findings. As the ischemia progresses and infarct occurs, pain becomes more localized and peritoneal signs can be present. CT angiography is the imaging study of choice for diagnosis with a sensitivity and specificity of over 90%. Management includes fluid resuscitation, broad spectrum IV antibiotics and emergent surgical consultation.
The celiac trunk (A) branches from the anterior aspect of the abdominal aorta and gives rise to the common hepatic, splenic, and left gastric arteries which supply the distal esophagus to the duodenum. The inferior mesenteric artery (B) branches from the aorta about 6 cm below the origin of the superior mesenteric artery. Via its branches, which include the left colic artery (C), sigmoid arteries and hemorrhoidal arteries, the inferior mesenteric artery provides circulation from the distal third of the transverse colon to the rectum. The celiac trunk, inferior mesenteric and left colic artery are all less likely to be occluded by an embolism than the superior mesenteric artery.
FOAMed:
References:
- Cudnik MT, Darbha S, Jones J, Macedo J, Stockton SW, Hiestand BC. The Diagnosis of Acute Mesenteric Ischemia: A Systematic Review and Meta-analysis. Jones AE, ed. Acad Emerg Med. 2013;20(11):1087-1100. doi:10.1111/acem.12254
- Award AK, Ryer EJ, Kalra M, et al. Revascularization for acute mesenteric ischemia. YMVA. 2012;55:1682-1689. doi:10.1016/j.jvs.2011.12.017
- Beaulieu RJ, Dean Arnaoutakis K, Abularrage CJ, Efron DT, Schneider E, Black III JH. Comparison of open and endovascular treatment of acute mesenteric ischemia. J Vasc Surg. 2014;59:159-164. doi:10.1016/j.jvs.2013.06.084
- Oldenburg WA, Lau LL, Rodenberg TJ, Edmonds HJ, Burger CD. Acute Mesenteric Ischemia. Arch Intern Med. 2004;164(10):1054. doi:10.1001/archinte.164.10.1054
- Kougias P, Lau D, El Sayed HF, Zhou W, Huynh TT, Lin PH. Determinants of mortality and treatment outcome following surgical interventions for acute mesenteric ischemia. J Vasc Surg. 2007;46(3):467-474. doi:10.1016/J.JVS.2007.04.045
- Carver TW, Vora RS, Taneja A. Mesenteric Ischemia. Crit Care Clin. 2016;32(2):155-171. doi:10.1016/j.ccc.2015.11.001
- Clair DG, Beach JM. Mesenteric Ischemia. Campion EW, ed. N Engl J Med. 2016;374(10):959-968. doi:10.1056/NEJMra1503884
- Acosta S, Björck M. Modern treatment of acute mesenteric ischaemia. Br J Surg. 2014;101(1):e100-e108. doi:10.1002/bjs.9330
- Singh M, Long B, Koyfman A. Mesenteric Ischemia. Emerg Med Clin North Am. 2017;35(4):879-888. doi:10.1016/j.emc.2017.07.005
- Chang M, Kistler EB, Schmid-Schönbein GW. Disruption of the Mucosal Barrier During Gut Ischemia Allows Entry of Digestive Enzymes Into the Intestinal Wall. Shock. 2012;37(3):297-305. doi:10.1097/SHK.0b013e318240b59b
- Acosta S. Mesenteric ischemia. Curr Opin Crit Care. 2015;21(2):171-178. doi:10.1097/MCC.0000000000000189
- Heijkant TC van den. Challenges in diagnosing mesenteric ischemia. World J Gastroenterol. 2013;19(9):1338. doi:10.3748/wjg.v19.i9.1338
- Acosta S, Block T, Björnsson S, Resch T, Björck M, Nilsson T. Diagnostic Pitfalls at Admission in Patients with Acute Superior Mesenteric Artery Occlusion. J Emerg Med. 2012;42(6):635-641. doi:10.1016/J.JEMERMED.2011.03.036
- Quiroga B, Verde E, Abad S, et al. Detection of patients at high risk for non-occlusive mesenteric ischemia in hemodialysis. J Surg Res. 2013;180(1):51-55. doi:10.1016/j.jss.2012.10.008
- Sun D-L, Cen Y-Y, Li S-M, Li W-M, Lu Q-P, Xu P-Y. Accuracy of the serum intestinal fatty-acid-binding protein for diagnosis of acute intestinal ischemia: a meta-analysis. Sci Rep. 2016;6(1):34371. doi:10.1038/srep34371
- Mccarthy E, Little M, Briggs J, et al. Radiology and mesenteric ischaemia. Clin Radiol. 2015;70:698-705. doi:10.1016/j.crad.2015.02.012
- Oliva IB, Davarpanah AH, Rybicki FJ, et al. ACR appropriateness criteria® imaging of mesenteric ischemia. Abdom Imaging. 2013;38(4):714-719. doi:10.1007/s00261-012-9975-2
- Reginelli A, Genovese E, Cappabianca S, et al. Intestinal Ischemia: US-CT findings correlations. Crit Ultrasound J. 2013;5(Suppl 1):S7. doi:10.1186/2036-7902-5-S1-S7
- Frishman WH, Novak S, Brandt LJ, et al. Pharmacologic Management of Mesenteric Occlusive Disease. Cardiol Rev. 2008;16(2):59-68. doi:10.1097/CRD.0b013e31815a6600
- Tilsed JVT, Casamassima · A, Kurihara · H, et al. ESTES guidelines: acute mesenteric ischaemia. Eur J Trauma Emerg Surg. 2016;42:253-270. doi:10.1007/s00068-016-0634-0
- Arthurs ZM, Titus J, Bannazadeh M, et al. A comparison of endovascular revascularization with traditional therapy for the treatment of acute mesenteric ischemia. J Vasc Surg. 2011;53(3):698-704; discussion 704-5. doi:10.1016/j.jvs.2010.09.049