Authors: Joe Penna, DO (EM Resident, UMass Chan-Worcester); Aaron Prugar, MD (EM Fellow Toxicology, UMass Chan-Worcester); Simon Ostrowski, MD (Assistant Professor of EM/Attending Physician, UMass Chan-Worcester); Cassandra Mackey, MD (Assistant Professor of EM/Attending Physician, UMass Chan-Worcester); Jeffrey Lai, MD (Associate Professor of EM/Attending Physician, UMass Chan-Worcester) // Reviewed by: Tony Spadaro, MD, MPH (Assistant Professor of Emergency Medicine, Medical Toxicology, and Addiction Medicine at University of Pennsylvania); Brit Long, MD (@long_brit)
Welcome to EM@3AM, an emDOCs series designed to foster your working knowledge by providing an expedited review of clinical basics. We’ll keep it short, while you keep that EM brain sharp.
A 42-year-old male with a past medical history of substance use presents to the ED for chest pain and shortness of breath. The patient also endorses nausea and vomiting. He reported snorting fentanyl two days ago and has a remote history of IV drug use.
Initial vital signs include temperature of 98.8°F, HR 150, RR 23, SpO2 98% on RA, and BP of 180/100. On examination, he is anxious-appearing and diaphoretic, with dilated pupils (6mm, equal, round, and reactive) and full body rigors; ankle clonus is absent. Labs demonstrate a potassium of 2.8 mEq/L and a lactate of 3 mg/dL. He receives electrolyte repletion, fluid resuscitation, and broad-spectrum antibiotics.
Concerned for opioid withdrawal, the treating physicians orders multiple doses of buprenorphine and full opioid agonists, without evidence of clinical improvement. Toxicology is consulted; they recommend sub-dissociative dose ketamine and dexmedetomidine infusion. The patient is admitted to the ICU for further management and observation.
What is the diagnosis?
Answer: Alpha-2 Agonist Withdrawal
Background/Epidemiology:
- The opioid epidemic has shifted to a fourth wave where fentanyl is adulterated with psychoactive substances. In some regions, alpha-2 agonists, such as xylazine and medetomidine, are common adulterants.1
- These adulterants are often added due to their sedative effects and to cut costs. Many of these medications, including medetomidine and xylazine, were intended for veterinary medicine.
- A common street name for xylazine is “Tranq.”2
- It first became prominent in Puerto Rico, but soon became prominent in states such as Ohio and Pennsylvania. It is now seen across much of the United States.3
- The most common combination is fentanyl and xylazine or fentanyl plus medetomidine, but some studies suggest patients have been exposed to all three.4
- A recent DEA report showed that in 2022, approximately 23% of fentanyl powder and 7% of fentanyl pills seized by the DEA contained xylazine.2 In April 2023, the Biden Administration labeled fentanyl laced with xylazine as an emerging drug threat.5
Pharmacokinetics:
Xylazine
- Half-life of 5 hours, with a duration of action of 4-8 hours
- Hepatically cleared4
Medetomidine
- Half-life of 1.5 to 2 hours
- Hepatically cleared via hydroxylation6
Pharmacodynamics:
- Compounds such as xylazine and medetomidine are all alpha-2 agonists.
- Xylazine is lipophilic. Thus it can cross the blood-brain barrier easily, allowing it to bind to peripheral and central alpha-2 receptors.8
- Alpha-2 agonists work by mimicking the action of the neurotransmitter norepinephrine at alpha-2 receptors.7
- This causes decreased sympathetic outflow from the central nervous system.
- By activating these receptors, they inhibit the release of norepinephrine and other neurotransmitters, leading to a range of effects, including:
- Decreased heart rate
- Decreased blood pressure
- Sedation and drowsiness
- Reduced anxiety and agitation
- Relief from pain
- Decreased gastrointestinal motility8
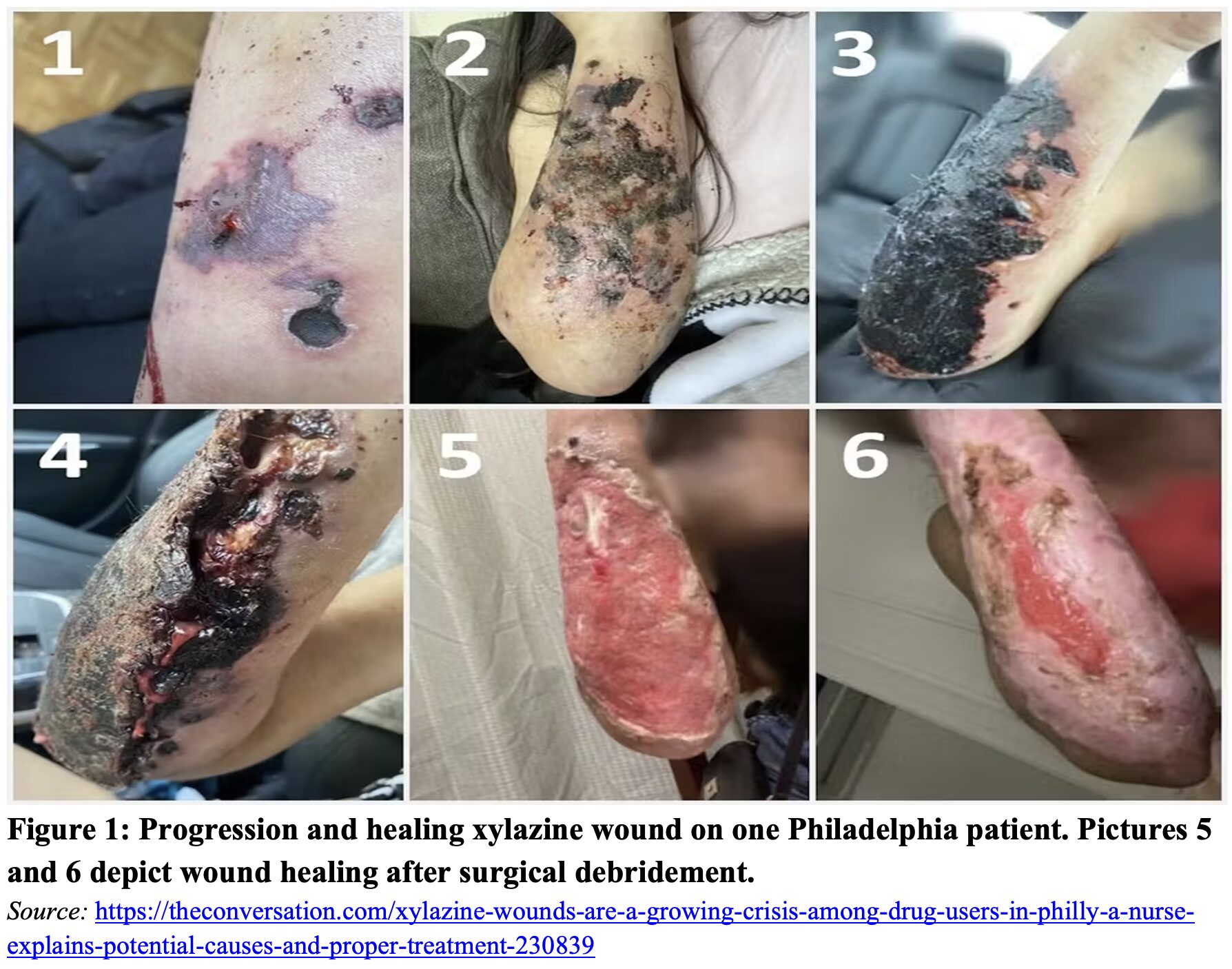
- Peripheral alpha-2 agonism can cause vasoconstriction, which is one mechanism thought to lead to severe wounds as seen primarily in xylazine use.9
- Medetomidine also works on imidazoline receptors. The activation of these imidazoline receptors can lead to:
- Hypotension
- Bradycardia8,10
Clinical Presentation:
Alpha-2 Agonist Intoxication4
- Miosis
- Sinus bradycardia
- Hypotension
- Sedation/lethargy
- Decreased respirations
- Skin lesions from chronic use (specifically with xylazine)
Alpha-2 Agonist Withdrawal4
- Mydriasis
- Sinus tachycardia, ~130-150 bpm
- Hypertension
- Nausea/vomiting (often severe and refractory to ondansetron)
- Diaphoresis
- Anxiety
- Large amplitude tremors (not clonus)
- Pain
- Skin lesions from chronic use
Evaluation/diagnosis:
- Diagnosis made primarily through history and exam.
- Consider alpha-2 agonist withdrawal in patients when severe autonomic hyperactivity (i.e., tachycardia, hypertension, diaphoresis) and psychomotor restlessness are present in those who use illicit opioids. Patients may have severe nausea and vomiting that does not respond to ondansetron.
- Failure to respond to conventional treatment for opioid withdrawal should further increase suspicion.
- Also consider alpha-2 agonist intoxication in patients with suspected opioid overdose (i.e., miosis, lethargy, bradycardia, and hypotension) who have an improvement in respiratory rate but remain sedated in response to naloxone.
- Differential diagnosis (not all inclusive):
- Toxicity: Opioid overdose, benzodiazepine overdose, ETOH intoxication, hypoglycemia, hypercarbia, hypoxia, hypothermia, hypothyroidism, infectious process, seizure, stroke, traumatic injury, beta blocker or calcium channel blocker overdose, myxedema coma
- Withdrawal: Opioid withdrawal, sympathomimetic toxidrome, sedative-hypnotic withdrawal, ETOH withdrawal, infectious process, pulmonary embolism, coronary ischemia, serotonin syndrome, neuroleptic malignant syndrome, malignant hyperthermia, anticholinergic toxidrome, aortic dissection
- Laboratory evaluation will be non-specific. May find a variety of nonspecific lab abnormalities such as electrolyte disturbances, AKI, myocardial injury, rhabdomyolysis, and lactate elevation.4
- Gas chromatography and mass spectrometry can detect these agents, but they will not return quickly and are not readily available.
- Rapid strip tests for xylazine are becoming more common now, but medetomidine rapid strip tests are not readily available in most EDs in the United States.8
- Consider obtaining basic labs:
- Point-of-care glucose (to rule out hypo/hyperglycemia causing altered mentation and agitation)
- CBC
- BMP (to check for electrolyte abnormalities)
- Troponin (if severe hypertension or if tachycardia is observed)
- VBG (assess patient’s ventilation status)
- If there are concerns for self-harm, obtain basic toxicology labs (i.e., acetaminophen, salicylate, and ethanol)
- Obtain ECG to assess for arrhythmia.8
Treatment:
Intoxication11,12
- The mainstay of treatment is supportive, focusing on supporting the patient’s airway and providing hemodynamic support.
- The duration of toxicity varies.
- Patients should be monitored until vital signs and mental status improve.
- If patients do not return to baseline after 6-8 hours, it may be reasonable to pursue hospital admission.
- Bradycardia in alpha-2 agonist intoxication is common.
- Bradycardia in alpha-2 agonist often does not need to be treated. Atropine should be avoided if possible. Bradycardia resolve as the compounds are metabolized.4
- Naloxone should be reserved for the treatment of respiratory insufficiency in someone with expected opioid exposure.
- Failure to respond to adequate doses of naloxone should raise concern for concomitant intoxication, such as alpha-2 agonists, benzodiazepines, etc.
- Naloxone should NOT be used to reverse drowsiness or bradycardia in the absence of respiratory insufficiency (i.e., bradypnea or hypercapnia).
- It will not reverse alpha-2 agonists and will likely cause acute opioid withdrawal.4
- Begin with conservative respiratory support, including capnography, nasal cannula, and NPA. May need to escalate respiratory support (i.e., intubation) for some patients.
Withdrawal13-15
- Consultation with in-hospital toxicologists and/or regional poison control centers can be a valuable resource in guiding the treatment of alpha-2 agonist withdrawal.
- Oral clonidine can be used for less severe alpha-2 agonist withdrawal or as step-down therapy.
- The typical maximum dose for clonidine is 2.4 mg per day. Starting doses include 0.1-0.2 mg oral for those with mild withdrawal or 0.3-0.5 mg oral for severe withdrawal. This can be repeated every 2-4 hours.
- Patients in alpha-2 agonist withdrawal may require significantly higher dosages, with some institutions utilizing 0.6-0.9 mg Q1H.15
- For more severely ill patients or those who cannot tolerate oral therapy, dexmedetomidine IV infusion may be necessary.
- This is often needed for 1-3 days.
- No optimal dose or established protocol for dexmedetomidine. Consider using infusion 0.2-0.5 mcg/kg/hr IV. Loading dose 1 mcg/kg can be considered for severe cases.
- Severe cases have been observed to require high doses of IV dexmedetomidine, sometimes exceeding “maximum doses” established per hospital protocol.15,16
- Medetomidine withdrawal closely resembles opioid withdrawal and can artificially inflate Clinical Opiate Withdrawal Scale (COWS) scores.
- If a patient has recently used illicit opioids (e.g., within 12-24 hours) or if they are maintained on methadone, they may still be at risk for buprenorphine-precipitated opioid withdrawal despite COWS scores >8-12.
- If the patient has recently used opioids or there is concern for risk of precipitated withdrawal, methadone or other full agonist opioids (e.g., hydromorphone, oxycodone) may be appropriate to manage opioid withdrawal physiology.
- Exercise caution in using methadone in those with prolonged QTc, which can be seen in medetomidine withdrawal.
- For acute opioid withdrawal superimposed with alpha-2 agonist withdrawal, consider the University of Pennsylvania Protocol.17
- Treat nausea and vomiting with metoclopramide or prochlorperazine.
- Tachycardia and hypertension should resolve with treatment of withdrawal symptoms.4
Prognosis/disposition:
- Patients displaying persistent alpha 2 agonist toxicity or severe withdrawal require hospital admission and intensive medical care.
- One study cited that ~90% of patients treated were admitted to the ICU on dexmedetomidine infusions.18
- Patients often improve after 24-48 hours.8
- Patients suspected of alpha-2 agonist consumption, asymptomatic patients, or patients who return to their baseline while being observed in the ED can be safely discharged.
Pearls:
- With this new wave of the opioid epidemic, consider alpha-2 agonist withdrawal/intoxication.
- Xylazine can cause severe, necrotic wounds in those who are chronically exposed.
- Alpha-2 agonist intoxication can cause sedation, bradycardia, and occasionally hypotension.
- Alpha-2 agonist withdrawal can cause a sympathomimetic-like toxidrome.
- Consider alpha-2 agonist withdrawal in patients not improving with standard opioid withdrawal treatment.
- Treat opioid withdrawal with short-acting opioids, and the mainstay treatment of alpha-2 agonist withdrawal centers around clonidine or dexmedetomidine, likely with consultation with toxicology or poison center.

A 3-year-old child is brought to the ED after ingesting an unknown quantity of clonidine tablets approximately 2 hours ago. The child is somnolent with an HR of 58 bpm, BP of 92/48 mm Hg, and RR of 10/min. Fingerstick glucose is 68 mg/dL. Which of the following is the most appropriate next step in management?
A) Administer flumazenil
B) Administer glucagon
C) Administer naloxone
D) Perform gastric lavage
E) Start intravenous fluids and observe
Correct answer: C
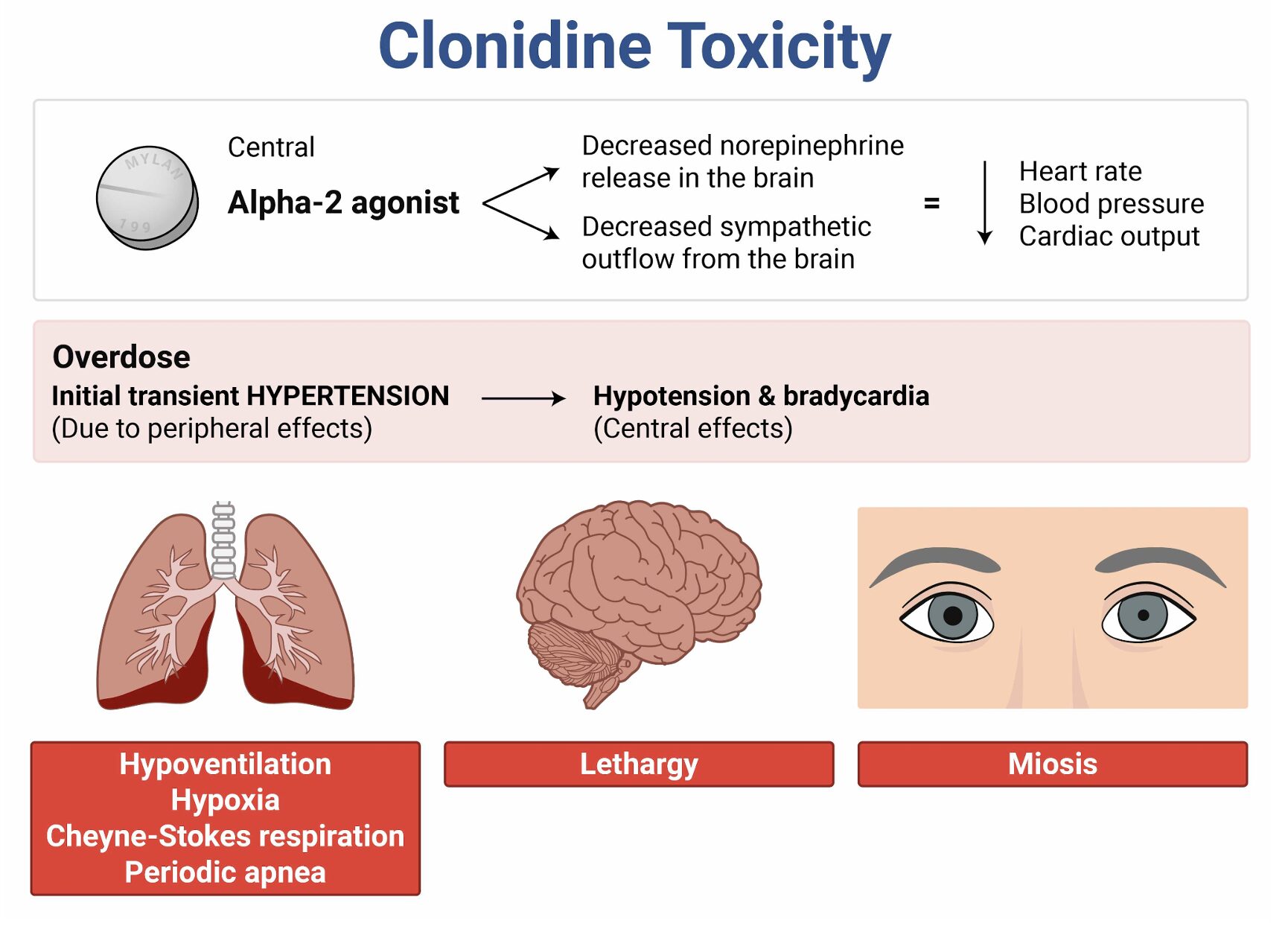
Naloxone is an opioid antagonist with off-label use in reversing CNS and respiratory depression caused by clonidine overdose. Although clonidine is an alpha-2 adrenergic agonist, it can produce opioid-like effects, including bradycardia, hypotension, respiratory depression, and somnolence. In pediatric patients, ingestion of even a single clonidine tablet can result in significant toxicity. Administering naloxone can counteract these effects by reversing the depression of the medullary centers responsible for respiratory and CNS alertness.
Diagnosis of clonidine toxicity is clinical, supported by a history of exposure and characteristic signs and symptoms. Sometimes, patients develop transient hypertension and tachycardia, but this quickly changes to hypotension and bradycardia. Laboratory studies, such as fingerstick glucose and blood gas analysis, can rule out other causes of altered mental status and guide therapy.
The mainstay of treatment for hypotension, especially in alert patients, is IV crystalloid therapy, starting at 60 mL/kg. Often, this is the only treatment necessary. The use of naloxone in adult patients is controversial, as improvement is inconsistent and rare in adults. However, the evidence is more robust in children. One study showed that nearly 80% of children with clonidine toxicity responded to naloxone. Thus, this is the treatment of choice in the pediatric population, especially if the child is somnolent, like this patient. The initial dose of naloxone is typically 0.01 mg/kg IV, titrated to effect. If a response to naloxone is incomplete, additional doses or other supportive measures, including atropine for bradycardia or vasopressors for refractory hypotension, may be needed. Patients should be admitted for observation, as symptoms of clonidine toxicity can recur due to its long half-life.
Flumazenil (A) is a benzodiazepine antagonist and is not indicated in the treatment of clonidine overdose. Using it could provoke seizures, especially in patients with a history of benzodiazepine use or dependence. Flumazenil would not address the alpha-2 adrenergic agonism caused by clonidine.
While glucagon (B) is effective for beta-blocker overdoses, it is not a first-line treatment for clonidine toxicity. The bradycardia and hypotension seen in clonidine overdose result from alpha-2 receptor stimulation, not beta-blockade.
Gastric lavage (D) is rarely indicated in pediatric patients due to the risk of complications such as aspiration or esophageal injury. It is most effective within 1 hour of ingestion, but the risks generally outweigh the benefits, particularly when safer and more effective treatments like naloxone are available. Additionally, clonidine is absorbed rapidly, and lavage is unlikely to remove significant amounts of the drug after 2 hours.
Although intravenous fluids (E) are a supportive measure for hypotension and should be started in this patient, they are not the best treatment as monotherapy for significant clonidine toxicity in a somnolent child. The bradycardia and respiratory depression require more specific interventions, such as naloxone, to directly address the underlying cause of the patient’s mental status changes. IV fluids would be the correct choice as the initial intervention in adult patients, but naloxone should be given first in pediatric patients.
Further reading:
Further FOAMed
- https://emcrit.org/ibcc/oa2a/
- https://www.emdocs.net/toxcard-medetomidine/
- https://www.emdocs.net/toxcard-xylazine/
References:
- Pieters T. The imperative of regulation: The co-creation of a medical and non-medical US opioid crisis.Psychoactives. 2023;2(4):317-336. doi:10.3390/psychoactives2040020
- Drug Enforcement Administration, US Department of Justice. The growing threat of xylazine and its mixture with illicit drugs. https://www.dea.gov. Accessed December 12, 2022.
- Moraff C. “Tranq dope” – The heroin combo that’s been putting Philly to sleep. Filter. April 2, 2019.https://filtermag.org/tranq-dope-the-heroin-combo-thats-been-putting-philly-to-sleep/. Accessed December 12, 2022.
- Farkas J. Xylazine & medetomidine: Opioid plus alpha-2 agonist (OA2A) toxidromes. EMCrit Project. August 28, 2025. https://emcrit.org/ibcc/oa2a/. Accessed September 16, 2025.
- Weixel N. White House says fentanyl laced with “tranq” drug is “emerging threat”. The Hill. April 12, 2023. https://thehill.com. Accessed April 13, 2023.
- Farkas J. Analgesia and sedation for the critically ill patient. EMCrit Project. June 29, 2024.https://emcrit.org/ibcc/pain/#dexmedetomidine. Accessed September 16, 2025.
- Ruiz-Colón K, Chavez-Arias C, Díaz-Alcalá JE, Martínez MA. Xylazine intoxication in humans and its importance as an emerging adulterant in abused drugs: A comprehensive review of the literature. Forensic Sci Int. 2014;240:1-8. doi:10.1016/j.forsciint.2014.03.015
- Wang S. Clonidine and related imidazoline poisoning. UpToDate. 2025. https://www.uptodate.com/contents/clonidine-and-related-imidazoline-poisoning. Accessed September 16, 2025.
- McFadden R, Wallace-Keeshen S, Petrillo Straub K, et al. Xylazine-associated wounds: Clinical experience from a low-barrier wound care clinic in Philadelphia. J Addict Med. 2024;18(1):9-12. doi:10.1097/ADM.0000000000001245
- Ghanavatian S, Derian A. Tizanidine. In: StatPearls [Internet]. Treasure Island, FL: StatPearls Publishing; January 2025–. Updated August 28, 2023. https://www.ncbi.nlm.nih.gov/books/NBK519505/
- Nayani Z, Reese T, Armstrong A, Walker A, Tani F, Fitzgerald R. Medetomidine-Associated Opioid Overdoses in Chicago, Illinois: A Report of 3 Cases. J Addict Med. 2025 Jul-Aug 01;19(4):497-500. doi: 10.1097/ADM.0000000000001500. Epub 2025 Apr 21. PMID: 40257097.
- Nham A, Le JN, Thomas SA, Gressick K, Ussery EN, Ko JY, Gladden RM, Mikosz CA, Schier JG, Vivolo-Kantor A, Fiorillo M, McMaster M, Magana DN, Verklan-McInnes L, Wahl M, Wood T, Adams A, Krotulski A, Trecki J, Ellison R, Gerona R, Arunkumar P, Mir M, Wise LM, Betancourt E, Monty K, Gulmatico J, Pojas A, Fitzgerald R, Hua M. Overdoses Involving Medetomidine Mixed with Opioids – Chicago, Illinois, May 2024. MMWR Morb Mortal Wkly Rep. 2025 May 1;74(15):258-265. doi: 10.15585/mmwr.mm7415a1. PMID: 40310760; PMCID: PMC12045484.
- London KS, Huo S, Murphy L, Warrick-Stone T, Goodstein D, Montesi M, Carter M, Butt S, Alexander K, Satz W, Tasillo A, Xu L, Arora M, Casey E, McKeever R, Lowenstein M, Durney P, Hart B, Perrone J. Severe Fentanyl Withdrawal Associated With Medetomidine Adulteration: A Multicenter Study From Philadelphia, PA. J Addict Med. 2025 Aug 1. doi: 10.1097/ADM.0000000000001560. Epub ahead of print. PMID: 40747932.
- Huo S, London K, Murphy L, Casey E, Durney P, Arora M, McKeever R, Tasillo A, Goodstein D, Hart B, Perrone J. Notes from the Field: Suspected Medetomidine Withdrawal Syndrome Among Fentanyl-Exposed Patients – Philadelphia, Pennsylvania, September 2024-January 2025. MMWR Morb Mortal Wkly Rep. 2025 May 1;74(15):266-268. doi: 10.15585/mmwr.mm7415a2. Erratum in: MMWR Morb Mortal Wkly Rep. 2025 May 15;74(17):296. doi: 10.15585/mmwr.mm7417a3. PMID: 40310762; PMCID: PMC12045483.
- Durney P, Paquin E, Fitzpatrick G, Lockstein D, Warrick-Stone T, Montesi M, Patel-Francis SH, Rashid J, Vaughan-Ogunlusi O, Goodsell K, et al. Profound Opioid and Medetomidine Withdrawal: A Case Series and Narrative Review of Available Literature. Psychoactives. 2025; 4(4):37. https://doi.org/10.3390/psychoactives4040037
- Durney P, Paquin E, Fitzpatrick G, Lockstein D, Warrick-Stone T, Montesi M, Patel-Francis SH, Rashid J, Vaughan-Ogunlusi O, Goodsell K, et al. Profound Opioid and Medetomidine Withdrawal: A Case Series and Narrative Review of Available Literature. Psychoactives. 2025; 4(4):37. https://doi.org/10.3390/psychoactives4040037
- Medetomidine – CAMP. PENN CAMP. April 23, 2025. https://penncamp.org/medetomidine/. Accessed September 16, 2025.
- Ostrowski SJ, Tamama K, Trautman WJ, Stratton DL, Lynch MJ. Notes from the Field: Severe Medetomidine Withdrawal Syndrome in Patients Using Illegally Manufactured Opioids – Pittsburgh, Pennsylvania, October 2024-March 2025. MMWR Morb Mortal Wkly Rep. 2025 May 1;74(15):269-271. doi: 10.15585/mmwr.mm7415a3. PMID: 40310861; PMCID: PMC12045482.
Images:
- Figure 1. McFadden R. Xylazine wounds are a growing crisis among drug users in Philly – a nurse explains potential causes and proper treatment. The Conversation. Published July 24, 2024. Accessed October 22, 2025.https://theconversation.com/xylazine-wounds-are-a-growing-crisis-among-drug-users-in-philly-a-nurse-explains-potential-causes-and-proper-treatment-230839