Author: Brit Long, MD (@long_brit, EM Attending Physician, San Antonio, TX) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital)
Lower gastrointestinal bleeding (LGIB) is a common ED chief complaint. Similar to many other conditions, LGIB may be associated with a variety of presentations and diseases ranging from benign to life-threatening. While most LGIB is due to diverticulosis, other conditions associated with LGIB include colitis, polyps, cancer, hemorrhoids, fissures, and others. Mortality is usually due to comorbidities, rather than exsanguination. The emergency clinician must be able to resuscitate the patient if needed and select the appropriate testing and disposition.
The British Society of Gastroenterology recently released their guidelines on LGIB bleeding evaluation and management in the BMJ. This post will provide you with the key takeaways. For more details and the evidence behind the recommendations, see the open access article.

Recommendations:
1. Categorize the patient as stable versus unstable (defined as shock index > 1). Patients with an unstable bleed should be resuscitated and undergo CTA. Patients with stable bleeds and shock index < 1 are less likely to have severe, active bleeding. These patients can be assessed with scoring systems, such as the Oakland score (weak recommendation, moderate quality evidence). The guidelines provide a nice algorithm with the recommendations:

Wait… What’s the Oakland score? This score is a tool comprised of 7 variables used to classify stable patients as major vs. minor LGIB. Variables include age, gender, prior LGIB admission, digital rectal exam, HR, SBP, and hemoglobin. A score < 8 predicts a 95% chance of safe discharge, or a minor bleed. Scores > 8 predict major bleed, with admission recommended. This score was derived and validated in the UK, and it has not been evaluated in other countries.

Other tools include the Glasgow-Blatchford score, which can be utilized as well. While typically used in UGIB, there is literature for its use in LGIB.
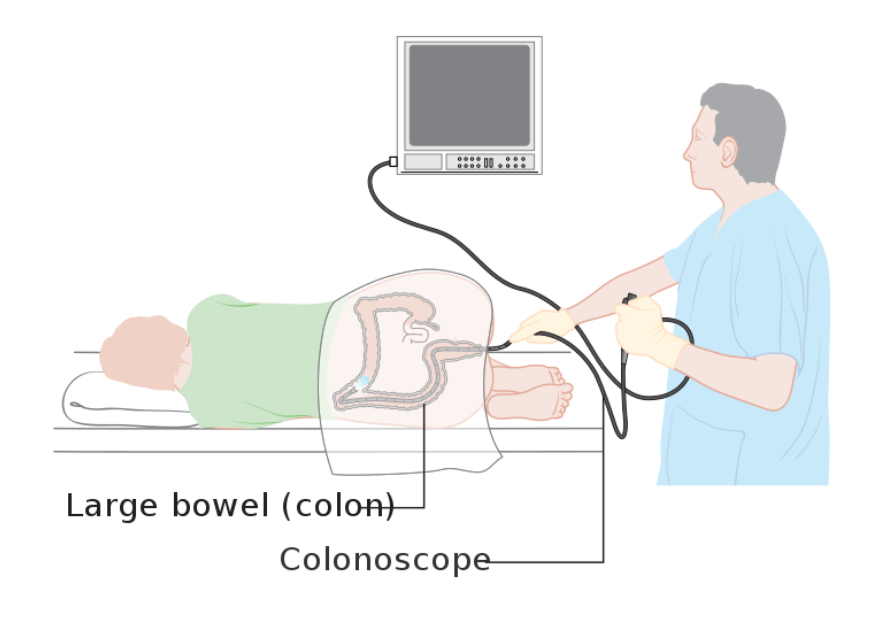
2. Patients with minor, self-terminating bleed with no other indications for admission can be discharged with urgent outpatient assessment (strong recommendation, moderate quality evidence). Colonoscopy is the recommended test, though endoscopy may be needed in higher risk patients.

3. Patients with major bleeding should be admitted for colonoscopy (strong recommendation, moderate quality evidence). This modality allows for diagnosis and therapy, with diagnostic yields ranging from 42-90%. Timing of colonoscopy is controversial, and patients with LGIB should “have an inpatient colonoscopy on the next available list.”
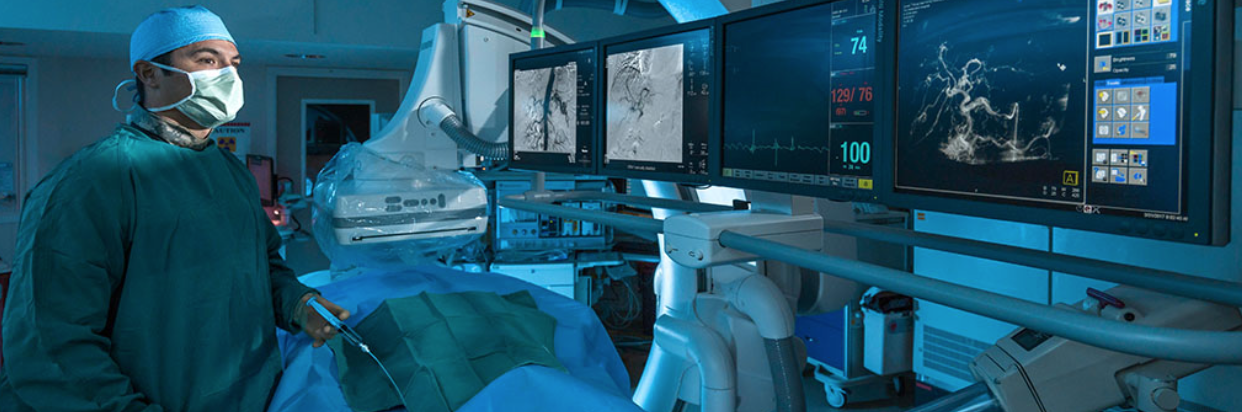
4. If the patient is hemodynamically unstable or has a shock index > 1 after resuscitation or active bleeding is suspected, obtain CTA. This modality provides the fastest and most accurate means to finding the sits of bleeding and allows for radiological intervention (strong recommendation, low quality evidence). CTA sensitivity and specificity range from 79-95% and 95-100%, respectively.

5. Upper endoscopy should be performed if CTA does not identify a source of bleeding, as LGIB with hemodynamic instability may be due to an UGIB source. Approximately 11-15% of patients thought to have a LGIB source actually have an UGIB source. NG placement for suspected UGIB is not recommended on a routine basis. Gastroscopy may be utilized if the patient stabilizes (strong recommendation, moderate evidence quality).

6. If CTA is positive, catheter angiography with embolization is recommended as soon as possible. In centers with 24/7 interventional radiology service, this should be completed within 1 hour for hemodynamically unstable patients (strong recommendation, low quality evidence).
Diagnosis: If no cause is found on CTA or endoscopy/gastroscopy, then other options include video capsule endoscopy.
Therapy: Most cases of LGIB stop spontaneously. However, treatment modalities include endoscopic therapy, embolization, and surgery.
7. Emergent laparotomy is the last line of therapy, which should be completed only after all other radiological and endoscopic modalities, except under exceptional circumstances (strong recommendation, low quality evidence).

8. Restrictive transfusion thresholds of 7 g/dL are recommended, with a Hb target of 7-9 g/dL after transfusion. In patients with cardiovascular disease, use a trigger of 8 g/dL and target of 10 g/dL (strong recommendation, low quality evidence). FFP and platelet transfusion is uncommon in these patients, with no randomized data for LGIB. They are a component of massive transfusion protocols, however.
9. Interrupt warfarin therapy at presentation (weak recommendation, low quality evidence). If unstable LGIB, reverse anticoagulation with PCC and vitamin K (strong recommendation, moderate quality evidence).

Recommendations 10-13 deal with patients with high thrombotic risk and heparin, aspirin, and dual antiplatelet therapy.
14. For patients taking DOACs, stop therapy at presentation (strong recommendation, low quality evidence). Consider inhibitors such as idarucizumab or andexanet for life-threatening bleeding (strong recommendation, moderate quality evidence). Restart DOAC therapy at a maximum of 7 days after bleeding (weak recommendation, very low quality evidence).

TXA may benefit in acute GI bleeding, but the guidelines do not make any clear recommendations pending the HALT-IT trial.
15-17. The final recommendations state hospitals should have access to a GI bleeding lead and agreed pathways for management, colonoscopy access, and interventional radiology access (either on site or through transfer).
While there is plenty of data still required, such as scoring systems, DOAC reversal, TXA use, etc., these guidelines provide several important updates for the evaluation and management of LGIB.
Reference:







2 thoughts on “Lower GI Bleeding Guidelines Update”
Pingback: Nedre GI-blödning – Mind palace of an ER doc
The BMJ also just published an open access review on LGIB, covering resuscitation, transfusion in LGIB, antithrombotic drug reversal, and definitive therapies. Read the article at https://www.bmj.com/content/364/bmj.l536.