Author: Nick Crain, MD (Flight Surgeon, USAF) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UT Southwestern Medical Center / Parkland Memorial Hospital) and Brit Long, MD (@long brit)
Case
A 35-year-old women presents with severe left-sided flank pain. The pain started about 7 hours ago as a vague, mild sensation in her left flank, but it has been gradually increasing in severity in a waxing and waning pattern and now is radiating to her labia. Her pain is unrelieved with over-the-counter Ibuprofen and is associated with mild nausea. She noticed blood in her urine the last 2 times she urinated. She has a history of hypertension treated with Lisinopril and Roux-en-Y gastric bypass surgery last year.
Her vital signs are normal. Examination is notable for a patient in distress, left-sided costovertebral angle tenderness, and mild tenderness to the left side of her abdomen without rebound tenderness or guarding. Urinalysis is positive for blood, while complete blood count, complete metabolic panel, and the remainder of the urinalysis are normal. Non-contrast CT scan reveals a 4mm non-obstructing stone in the left ureter.
Background
Nephrolithiasis is a common diagnosis in the ED. Evidence over the past 2 decades suggests an increasing prevalence of kidney stones, citing an increase from 3.8% between 1976 and 1980 to 8.4% between 2007 and 2010.1 This increase was seen particularly in women, with an overall male-to-female ratio decreasing from 3:1 to less than 2:1 over the past 25 years.2
A large number of factors may contribute to kidney stone formation. Known risk factors include high urine calcium, high urine oxalate, low urine citrate, and low urine volume. Dietary factors such as low intake of fluid, calcium, potassium, and phytate or high intake of oxalate, sodium, sucrose, fructose, and animal protein also predispose patients to stones.3 Additionally, a number of medical conditions increase the risk of nephrolithiasis, chiefly primary hyperparathyroidism, obesity, gout, diabetes, and medullary sponge kidney.
Nephrolithiasis can be asymptomatic. Roughly 70% of asymptomatic cases remain asymptomatic after 31 months.4 Patients who experience symptoms often complain of pain ranging from a mild ache to extremely severe pain requiring parenteral analgesia. This pain typically resolves following passage of the stone. Pain is accompanied by hematuria in the majority of patients, particularly early in presentation. Roughly 95% of patients have either gross or microscopic hematuria on day 1 of pain, while on day 3 the percentage of those with hematuria decreases to 65%.5 Other symptoms include nausea and vomiting, as well as dysuria and urgency if the stone is in the distal ureter. Definitive diagnosis can be made with non-contrast CT scan. Patients with signs and symptoms of urosepsis and acute kidney injury should be emergently decompressed by Urology. Stones >10mm should be evaluated by Urology and potentially treated via extracorporeal shock wave lithotripsy or ureteroscopy.6,7 Smaller stones can be expectantly managed by treating symptoms and straining urine to detect passage. An alpha-blocker such as tamsulosin 0.4mg given once daily for up to 4 weeks has been found to expedite stone passage in some patients, though the data are controversial. Stones greater than 5mm may benefit the most from tamsulosin, but stones smaller than this likely do not pass faster with medications.8 Patients with acute renal failure, associated urinary tract infection, intolerable pain, PO intolerance, and stone with moderate/severe hydronephrosis require urology consultation and consideration for admission.

Many conditions can present similarly to nephrolithiasis, some of which are life-threatening.9 Luckily, there are distinguishing features with these alternative diagnoses that can usually be teased out with a focused history, physical exam, lab studies, and in most cases, imaging.
Upper urinary tract infections
Urinary tract infections (UTI’s) are much more common in women than in men due to a shorter urethral length, a higher urethral colonization rate, and lack of prostatic fluid which has antimicrobial properties.10,11 UTI’s are classified as either lower UTI’s (cystitis) or upper UTI’s (pyelonephritis) and can occur alone or in conjunction with one another. UTI’s are further classified into uncomplicated and complicated. They are considered complicated when the affected individual has a condition that renders them more likely to fail treatment. These include underlying kidney disease, pregnancy, hospital acquired infections, diabetes mellitus, male gender, and anatomic abnormalities of the urinary tract. Specific to the upper urinary tract, acute complicated pyelonephritis describes progression of the UTI to emphysematous pyelonephritis, renal corticomedullary abscess, perinephric abscess, or papillary necrosis. For the most part, clinical manifestations of these entities mirror severe acute pyelonephritis. Delineation is usually made with a CT scan.12
Uncomplicated and complicated pyelonephritis overlap considerably in their presentation with renal colic. Both diagnoses may present with flank pain, costovertebral angle tenderness, nausea, and vomiting. Additionally, dysuria and urgency are common in both distal ureteral stones and ascending UTI’s that include the lower urinary tract. Hematuria is common in nephrolithiasis as well as UTI’s that include the lower urinary tract. Fever is a distinguishing feature of urinary tract infections, particularly pyelonephritis. Fever is rare in nephrolithiasis unless there is a concomitant infectious process. A patient who presents with sepsis and fever warrants further evaluation. A CT scan, combined with CBC, metabolic panel, and urinalysis, narrows the diagnosis in cases of uncertainty.
Ectopic pregnancy
Approximately 98% of extrauterine pregnancies occur in the fallopian tube.13 Risk factors for ectopic pregnancy include any previous fallopian tube procedure/pathology (especially past ectopic pregnancy), infertility, history of cervicitis, current IUD use, multiple sexual partners, and smoking, to name a few.14 The most common presentation of ectopic pregnancy is first-trimester abdominal pain and vaginal bleeding, particularly 6-8 weeks after the last menstrual period.15 Pain is typically localized to the pelvic region. Ectopic pregnancies are classified ruptured or unruptured. Patients with tubal rupture may present with symptoms of profound blood loss, such as light-headedness, loss of consciousness, and hypotension/shock. Other potential features of rupture include shoulder pain due to referred pain of diaphragmatic irritation, peritoneal signs, and free fluid in the pelvic cul-de-sac and/or abdominal cavity.
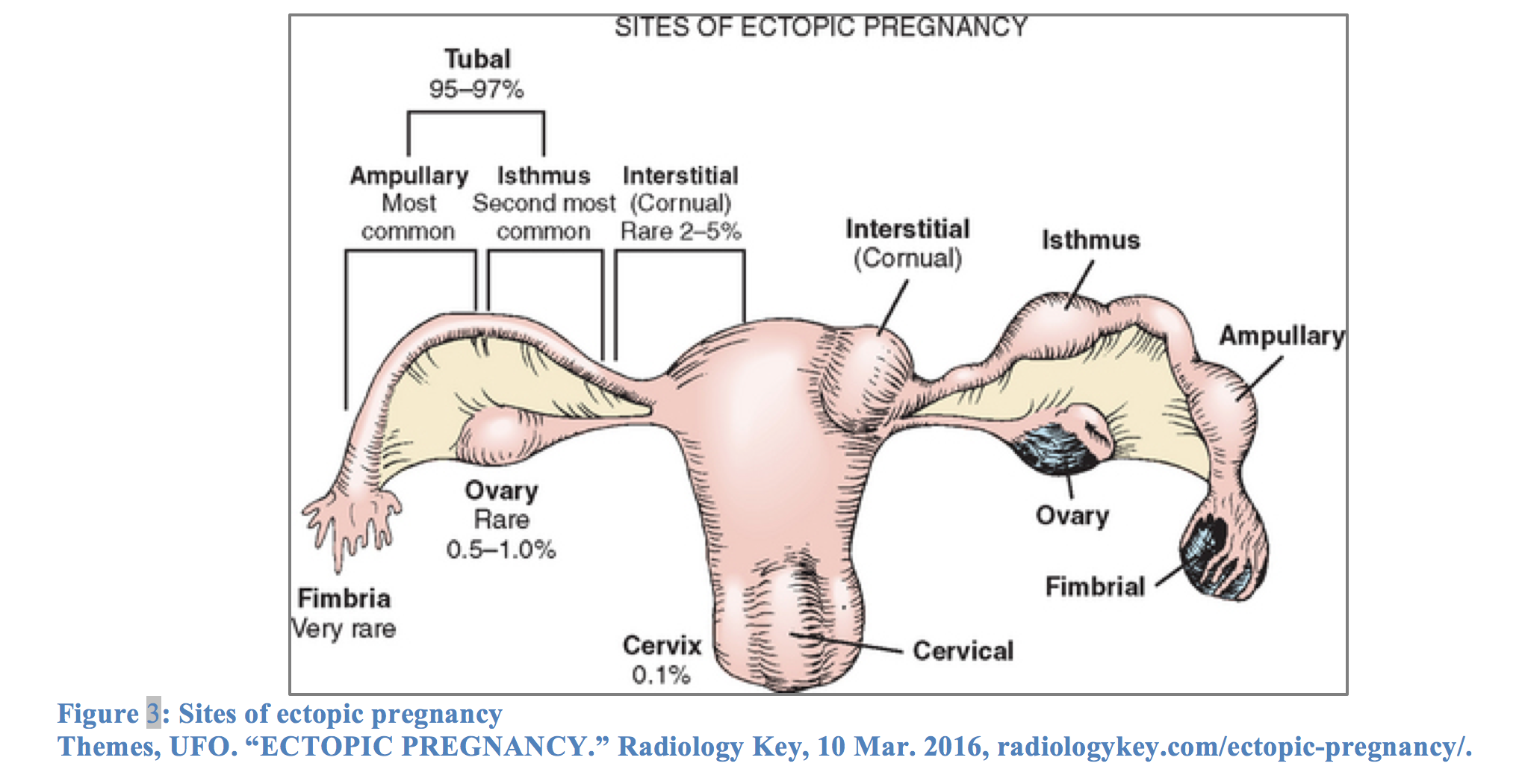
Because of the potential for rupture, all women of child-bearing age with abdominal pain and/or vaginal bleeding require evaluation for ectopic pregnancy. Location of the patient’s pain may overlap with typical nephrolithiasis, and vaginal bleeding may be mistaken for hematuria. Diagnosis of ectopic pregnancy is confirmed with transvaginal ultrasound (TVUS) revealing an extrauterine pregnancy and positive B-hCG.
However, diagnosis can be complicated in cases of positive B-hCG and an inconclusive TVUS. In these situations, a serum quantitative B-hCG level is then measured. If this value is above the level at which TVUS may identify intrauterine pregnancy, also known as the “discriminatory zone,” then ectopic pregnancy is assumed. However, complicating matters further is that while most facilities use a value of 1000-2000, the discriminatory zone has not been definitively established by the literature. Also, a serum quantitative B-hCG level by itself is not of value in diagnosis because ectopic pregnancies can occur at virtually any B-hCG level. For these reasons, patients with inconclusive ultrasounds should receive OBGYN evaluation with serial B-hCG levels measured roughly 48 hours apart.16
Ovarian torsion
Ovarian torsion results from partial or complete rotation of the ovary, resulting in reduction of blood flow to the organ. If left untreated, it may result in necrosis and permanent dysfunction of the affected ovary. Torsion affects women of every age but is most common in women of reproductive age. The primary risk factor for ovarian torsion is an ovarian mass. This risk increases with the diameter of the mass, with size greater than 5cm being of particularly high risk.17 However, torsion can present in females without a preexisting mass, especially in the pediatric population. A definitive diagnosis of ovarian torsion can be difficult to make because it is only confirmed when a rotated ovary is visualized during surgery or pelvic ultrasound. Pelvic US cannot definitively rule out the diagnosis. Women may experience intermittent symptoms as the ovary torses and detorses. Therefore, the decision to go to surgery is usually based on a number of signs and symptoms as well as exclusion of other diagnoses.18
Ovarian torsion can present as sharp, stabbing flank or pelvic pain in a waxing and waning pattern, similar to renal colic. The most common features experienced with ovarian torsion, in order, are pelvic pain, presence of an adnexal mass, nausea/vomiting, fever, and abnormal gynecologic bleeding.19 When fever is present, it may indicate ovarian necrosis, particularly in the setting of concomitant leukocytosis. Hematuria is typically not present. Because of the common difficulty in making a confident diagnosis of ovarian torsion, alternative diagnoses, including renal colic, should be excluded based on clinical symptoms and imaging if appropriate.
Adnexal masses
Adnexal masses can represent a wide variety of conditions. Etiologies range from benign ovarian cysts to more severe causes such as ovarian cancer and urgent conditions such as ectopic pregnancy, adnexal torsion, and tuboovarian abscess. The primary role of the emergency physician is to rule out urgent conditions. Ectopic pregnancy and adnexal torsion were discussed above. The other two primary urgent conditions are tuboovarian abscess and an ovarian cyst (ruptured or hemorrhagic).
Tuboovarian abscess should be suspected in any female who presents with lower abdominal pain, fever/chills, vaginal discharge, and an adnexal mass. A pelvic ultrasound commonly shows a complex multilocular mass that obliterates normal adnexal architecture.20 Tuboovarian abscess should be managed quickly to reduce the risk of damage to fallopian tubes and infertility. Patients with signs of sepsis and peritonitis should be assumed to have an abscess that has ruptured and receive immediate surgical drainage. Additionally, tuboovarian abscess in postmenopausal women should raise the suspicion for concurrent ovarian malignancy. Otherwise, most premenopausal women are initially treated with antibiotic therapy alone.21
The classic presentation of a ruptured ovarian cyst is the abrupt onset of severe unilateral lower abdominal pain in mid-menstrual cycle immediately following sexual intercourse or other physical activity. They are most often physiologic, follicular cysts which form mid-cycle and are followed by the corpus luteum. Diagnosis is aided with a pelvic ultrasound which may show an ovarian cyst with free fluid in the pelvis. An ovarian cyst may not be visible if it has already ruptured; however lack of cyst visualization on ultrasound should prompt further evaluation of other conditions. Patients may also present with hemorrhagic cysts without preceding activity. In both ruptured ovarian cysts and hemorrhagic ovarian cysts, hemodynamically stable women are managed conservatively. Patients should be hospitalized in the presence of a significant hemoperitoneum and a drop in blood count to rule out ongoing blood loss via frequent monitoring of vital signs and blood counts. Most patients with stable vital signs and hematocrit are managed conservatively without surgery. Fever, leukocytosis, or possible malignancy are other indications for hospitalization.22
Testicular torsion
Testicular torsion, along with acute epididymitis, is one of the most common scrotal emergencies in men and classically presents with acute scrotal pain with swelling, diffuse tenderness, a “high-riding testicle,” and absent cremasteric reflex. The testicle is often found in a transverse plane, contrasting with normal longitudinal lie. However, the absence or presence of cremasteric reflex is not reliable. Relief of pain with attempted detorsion away from the midline suggests torsion (“Open the book”). Suspected cases should be urgently surgically corrected, even if attempted manual torsion was apparently successful, so that the testicle can be secured to the scrotal wall to prevent recurrence and definitively correct the torsion. In cases of uncertainty, Doppler ultrasound with abnormal or absent testicular blood flow can confirm the diagnosis.23 US is also not 100% sensitive. Delay of treatment may result in irreversible testicular damage and infertility.24
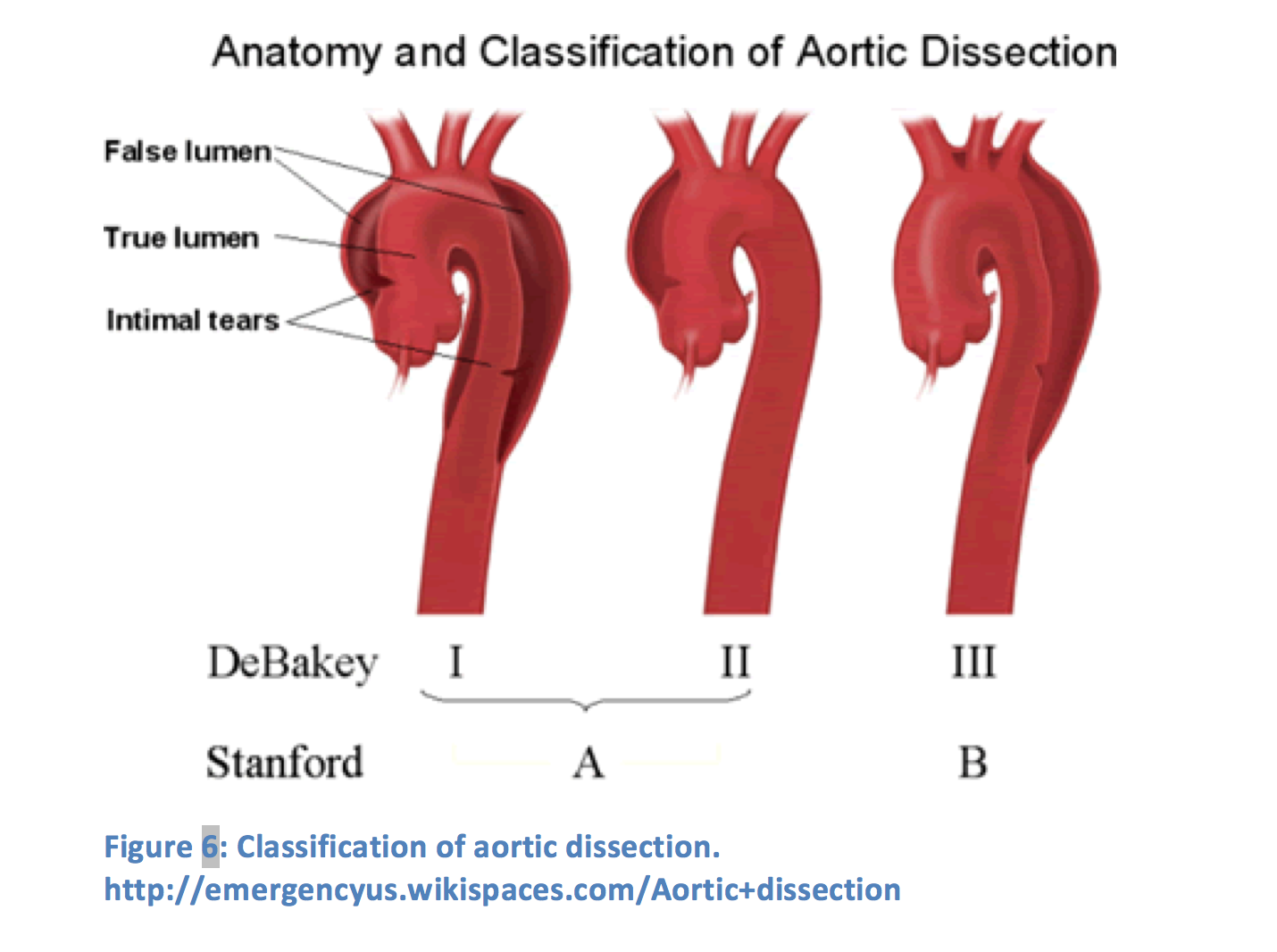
Acute aortic syndromes
Acute aortic dissections are the most common acute aortic syndrome and are nearly twice as common in the ascending aorta as the descending aorta.25 Either variant may mimic renal colic, particularly dissections in the descending abdominal aorta, as they may cause severe acute back pain. Obviously, these are potentially life-threatening events that should be considered, particularly in any individual with advanced age, male sex, systemic hypertension, a preexisting aortic aneurysm, and risk factors for atherosclerosis.26 While pain is the most common sign and may occur without other symptoms, it may be accompanied by syncope, myocardial infarction, signs of stroke, hemodynamic instability, or other evidence of end-organ/ischemia.27 Other acute aortic syndromes include aortic intramural hematoma, penetrating aortic ulcer, intimal tear without hematoma, and periaortic hematoma. Symptoms of all acute aortic syndromes are similar and nonspecific; therefore, angiographic imaging modalities, particularly CT angiography, are relied upon to confirm the diagnosis.28
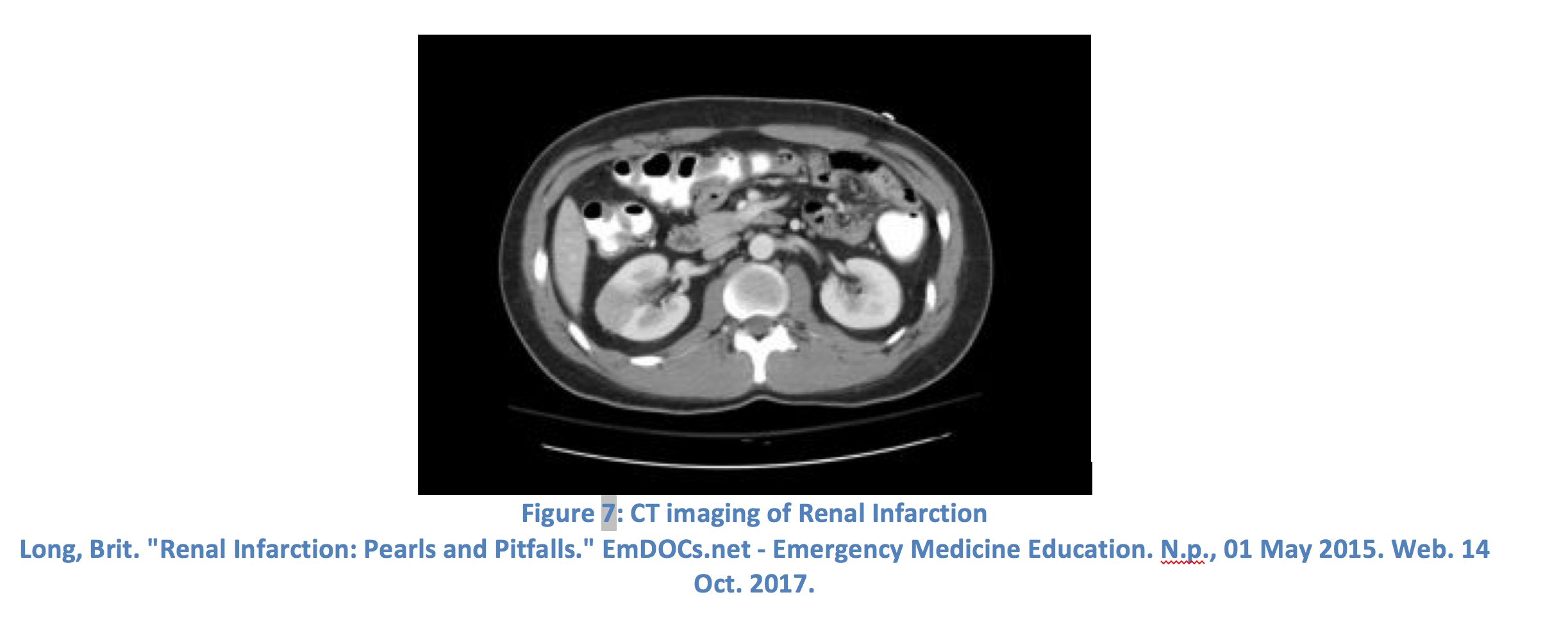
Renal infarction
The 2 most common causes of renal infarction are cardioembolic disease and renal arterial injury, such as dissection.31 Hypercoagulable states also account for a significant percentage of renal infarction, although roughly 30% are idiopathic.32 Symptoms closely mirror that of nephrolithiasis and consist of flank pain, nausea, vomiting, fever, elevated blood pressure (due to increased renin release), and hematuria, although hematuria is not as common as in renal colic. Labs classically reveal microscopic hematuria, elevated LDH, and normal liver enzymes.33 Symptoms and patient characteristics often mirror that of AAA, so a bedside ultrasound evaluating the aorta should be the first step in diagnosis. Non-contrast CT should then be performed to evaluate for nephrolithiasis, followed by a CT with IV contrast to evaluate for infarction if no stone is found and infarction is still presumed likely.34 Renal infarction is classically identified as a wedge-shaped perfusion defect.
Splenic infarct
The classic presenting symptoms of splenic infarct is upper left abdominal pain and tenderness.35 However, many other presentations are possible. Other common findings include nausea/vomiting, splenomegaly, elevated LDH, fever, and leukocytosis. Splenic infarct may appear in a variety of settings, including hypercoagulable states, embolic disease, myeloproliferative neoplasms, hemoglobinopathies such as sickle cell disease, splenomegaly of any cause, trauma, splenic artery torsion, and rarely, mononucleosis.36 Diagnosis is confirmed with a CT scan. Treatment should be directed at the underlying cause.
Bowel obstruction
Bowel obstruction has a wide variety of causes. Both the small and large bowel may become obstructed, and the obstruction may be functional, due to abnormalities in intestinal physiology, or mechanical, due to a physical barrier preventing movement of intestinal contents. The most common cause of mechanical bowel obstruction is adhesions from prior surgery. Patients will typically present with acute nausea, vomiting, abdominal pain, and chronic postprandial discomfort or distension. These symptoms may occur in a waxing and waning pattern, similar to renal colic.37 When bowel obstruction is suspected, routine lab studies in addition to lactate with abdominal plain film should be early steps in the evaluation. Abdominal plain film can show important features such as air-fluid levels or pneumoperitoneum, though sensitivity is poor.38 Abdominal CT can specify the location of the bowel obstruction in less urgent cases. US can also be used for diagnosis. Unlike nephrolithiasis, hematuria is not characteristic of bowel obstruction, and profound abdominal tenderness is relatively uncommon in renal colic compared to bowel obstruction.
Diverticulitis
While diverticulitis is most common among the elderly, it can occur at any age. Although right-sided diverticulitis occurs, 85% of cases will occur on the left. Left-sided abdominal pain is the most common complaint, usually constant in character and present for several days prior to presentation.39 The diagnosis should be suspected in anyone presenting to the ED with left-sided abdominal pain, particularly with nausea, vomiting, diarrhea, constipation, leukocytosis, and/or low-grade fever. Diverticulitis is confirmed with CT scan. Complicated cases, defined by the presence of abscess, perforation, obstruction, and fistulization, as well as high-risk patients are admitted. Patients are considered high risk if they are immunosuppressed, have fever greater than 102.5o F, significant leukocytosis, severe abdominal pain, peritonitis, advanced age, significant comorbidities, intolerance to oral intake, poor follow-up or support system, history of non-compliance, or failure of outpatient management. Candidates for outpatient treatment often receive a course of Ciprofloxacin 500mg q12h combined with Metronidazole 500mg q8h for 7-10 days.
Appendicitis
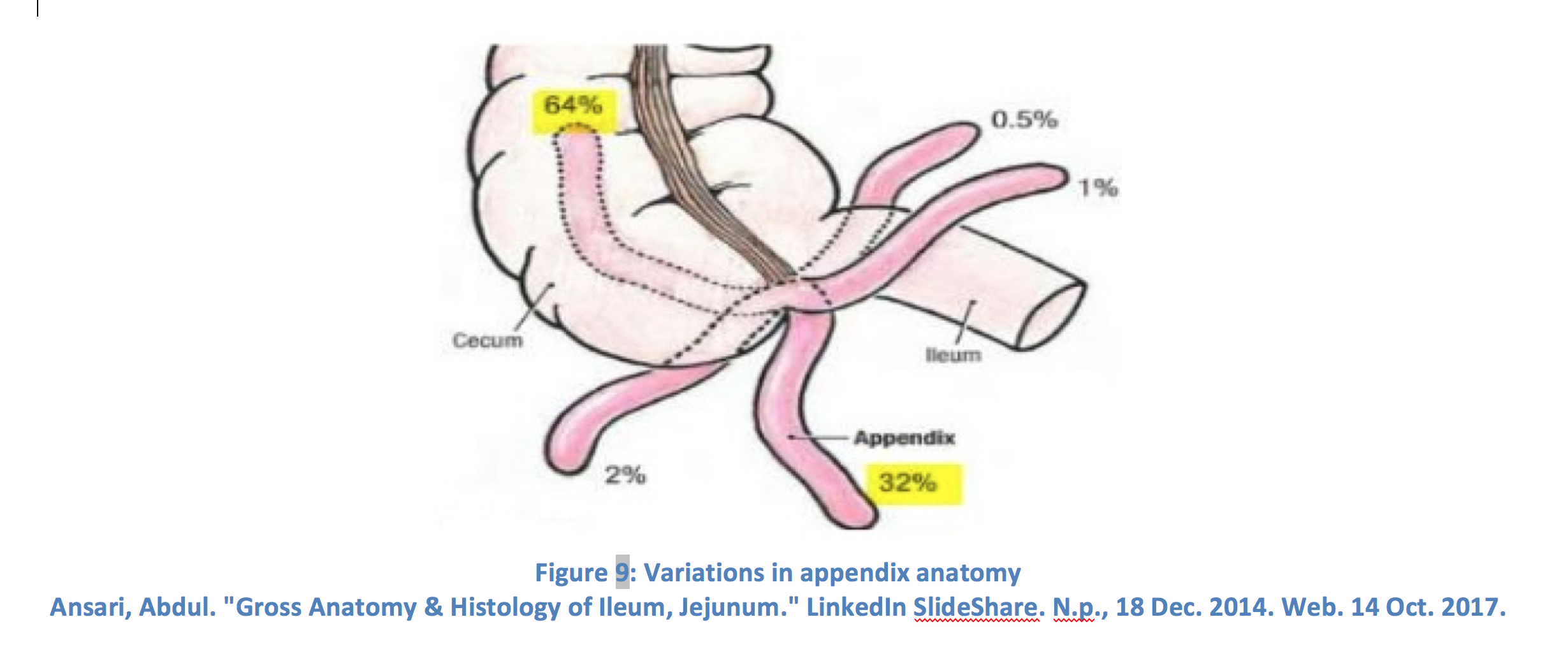
Appendicitis is one of the most common causes of acute abdomen and a frequent indication for an emergent abdominal surgical procedure.40 The classic presentation is periumbilical abdominal pain with subsequent migration to the right-lower quadrant. Associated symptoms include nausea, vomiting, anorexia, and fever. Other nonspecific findings include indigestion, flatulence, irregular bowels, and malaise.41 Pain in the right lower quadrant can mimic that of a stone in the right ureter. It is important to note that the classic migratory pain is not seen in nearly half of patients, and appendix anatomy is variable.42,43 Appendicitis may be more likely to mimic renal colic if the tip of the appendix is found in the pelvis, in which case patients may have pain below McBurney’s point and experience dysuria and urgency.44 CT scan with IV contrast is the preferred imaging modality for diagnosis.
Cholecystitis/Biliary colic
The classic presentation of gallstone disease is an intense, dull discomfort located in the right upper quadrant or epigastrium that may radiate to the back, commonly associated with nausea/vomiting and diaphoresis. Eating a fatty meal is a common, but not uniformly experienced trigger.45 Physical exam is usually normal. Acute cholecystitis should be suspected when symptoms of biliary colic are present in the setting of fever and/or leukocytosis or when right upper quadrant pain lasts for greater than 6 hours without waning as would be typical of uncomplicated gallstone disease.46 Abdominal ultrasound is the imaging method of choice when either disease is suspected. Ultrasound can both visualize gallstones and detect gallbladder wall thickening and edema caused by acute cholecystitis. Biliary colic and cholecystitis may mimic renal colic in that they can present with flank pain. However, hematuria would not be a manifestation of gall bladder disease.9
Acute intestinal ischemia
Intestinal ischemia can be acute or chronic and occur in both the small (mesenteric ischemia) and large intestine (colonic ischemia). Risk factors for intestinal ischemia are many and include cardiac disease, aortic surgery/instrumentation, peripheral artery disease, hemodialysis, vasoconstrictive medications, hypercoagulable conditions, and infection.47 History is of particular importance in the diagnosis of intestinal ischemia. A previous embolic event is present in about one-third of patients, and a personal or family history of DVT or PE is present in about one-half of patients with acute mesenteric venous thrombosis.48 Classic presentation is “pain out of proportion to physical exam” and a history of postprandial abdominal pain, anorexia, and unintentional weight loss. Lab work is nonspecific. Lactate is commonly elevated late in the disease course. For unstable patients, abdominal plain film is the initial test of choice while CT with IV contrast is preferred for stable patients in the emergency department.49 Anticoagulation is needed.
Pneumonia
Patients with pneumonia occasionally mimic renal colic, especially with lower lobe involvement. Pleuritic pain in those with pneumonia is often severe enough to restrict respiratory movements.50 Location of pain is dependent on which side is affected and can be located near the flank in many patients. Pneumonia’s classic presentation of fever, chills, sputum production, and pleuritic pain is seen most often in younger populations. The elderly, in which pneumonia is much more common, typically have more subtle symptoms. Hematuria would be highly unusual. An infiltrate on a chest radiograph is often the most common imaging finding, though US is often helpful.51
Pulmonary Embolism
PE may also present with pleuritic pain located near the flank, thus mimicking renal colic. Pleuritic pain is the second most common symptom, preceded only by dyspnea.52 Other symptoms found in PE in order of frequency include cough, orthopnea, wheezing, and hemoptysis. If DVT is present, the affected leg may be swollen. Less common presentations are arrhythmias due to right ventricular strain, syncope/pre-syncope, and hemodynamic collapse. The most common signs are (in order) tachypnea, exam suggestive of lower extremity DVT, tachycardia, rales, decreased breath sounds, accentuated pulmonic component of second heart sound, JVD, and fever. Tachypnea and tachycardia combined with pleuritic flank pain is entirely possible as a presentation of both PE and renal colic. Risk factors for DVT and renal colic should be elicited during the history, as these can help narrow the diagnosis. Diagnosis consists of CT of the pulmonary vasculature, with anticoagulation for management.
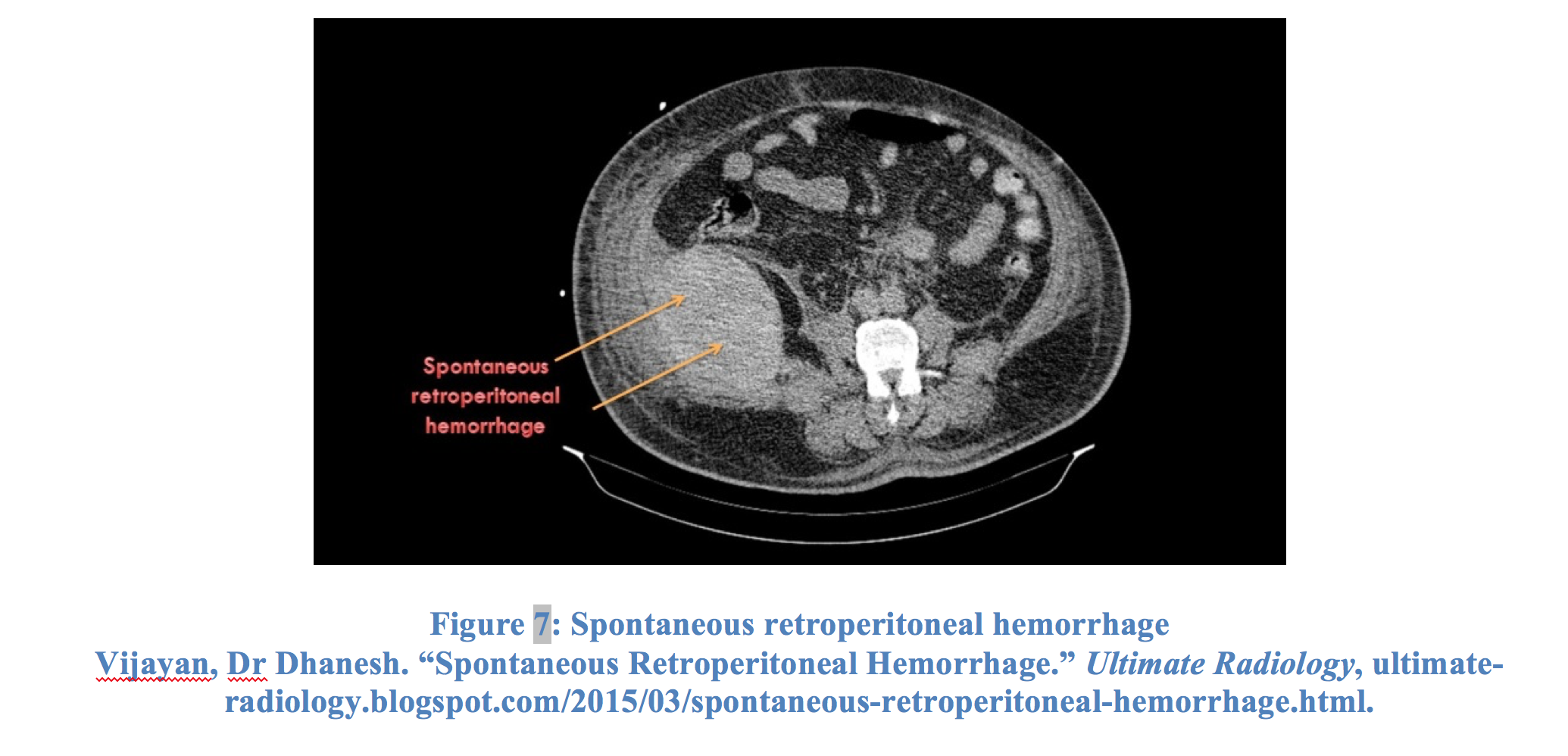
Retroperitoneal hematoma
Bleeding into the potential space behind the abdominal cavity can result from trauma or a ruptured aneurysm of the aorta or one of its branches. Structures that lie in the retroperitoneum include the aorta, IVC, kidneys and their vessels, adrenal glands, ascending and transverse colon, pancreas, and part of the duodenum. Injury to any of these structures can potentially cause flank pain and mimic renal colic. Injury to either kidney could cause hematuria, further mirroring the symptoms of nephrolithiasis.
Retroperitoneal hemorrhage, either from trauma or aortic rupture, represents a potentially life-threatening situation requiring urgent intervention. Patients may have abdominal tenderness and ecchymosis on the abdomen, flanks, scrotum, or perineum, but overall history and physical are largely unreliable.53 Hemodynamically stable patients should receive CT scan with IV contrast for diagnosis confirmation. Unstable trauma patients should receive a FAST and surgical exploration of the abdomen. A negative FAST should not dissuade surgery, and a negative FAST in a hypotensive patient suggests retroperitoneal bleeding.54
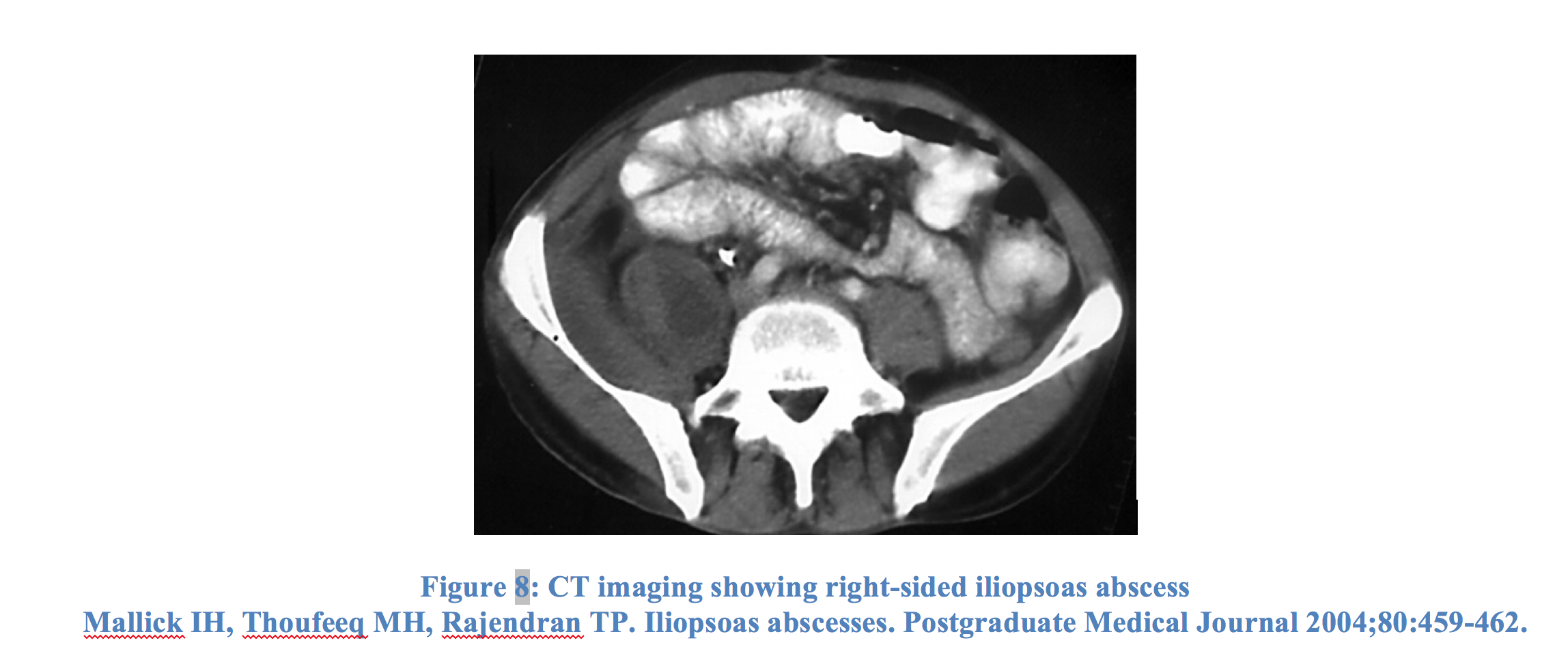
Iliopsoas abscess
An iliopsoas (or psoas) abscess is a collection of pus in the iliopsoas muscle compartment.55 It may arise from contiguous spread from adjacent structures or by hematogenous spread from a distant site. Staph aureus, including MRSA, is the most commonly isolated organism. Symptoms include back/flank pain, fever, an inguinal mass, limping, anorexia, and weight loss. Pain may also be present in the lower abdomen and is often exacerbated when performing movements in which the psoas muscle is stretched or extended (“psoas sign”). Limitation of hip movement is common and patients will often hold their hip in flexion for less discomfort.56 Diagnosis is confirmed with CT scan, though sensitivity is limited early in the disease course.55
Summary:
- Flank pain and hematuria are the hallmarks of renal colic, however the presentation is variable.
- Diagnosis can be confirmed with CT scan, which will show most other potential items in the differential if the scan is negative for a stone.
- Patients with a good story for stone who are young and healthy at baseline can receive renal US to evaluate for hydronephrosis, rather than CT.
- Nephrolithiasis and nearly all of its mimics are confirmed with imaging.
- Some mimics of renal colic that may arrive at the ED include: upper UTI’s, ectopic pregnancies, ovarian torsion, adnexal masses, testicular torsion, acute aortic syndromes, renal artery aneurysms, renal infarction, splenic infarction, bowel obstruction, diverticulitis, appendicitis, biliary colic, cholecystitis, acute intestinal ischemia, pneumonia, pulmonary embolism, retroperitoneal hematoma, iliopsoas abscess.
- Focused history and physical exam are paramount due to the multitude of structures in the area and potentially dangerous conditions that may mimic renal colic.
References/Further Reading:
- Scales CD Jr, Smith AC, Hanley JM, et al. Prevalence of kidney stones in the United States. Eur Urol 2012; 62:160.
- Scales CD Jr, Curtis LH, Norris RD, et al. Changing gender prevalence of stone disease. J Urol 2007; 177:979.
- Coe FL, Parks JH, Asplin JR. The pathogenesis and treatment of kidney stones. N Engl J Med 1992; 327:1141.
- Glowacki LS, Beecroft ML, Cook RJ, et al. The natural history of asymptomatic urolithiasis. J Urol 1992; 147:319.
- Kobayashi T, Nishizawa K, Mitsumori K, Ogura K. Impact of date of onset on the absence of hematuria in patients with acute renal colic. J Urol 2003; 170:1093.
- Preminger GM, Tiselius HG, Assimos DG, et al. 2007 guideline for the management of ureteral calculi. J Urol 2007; 178:2418.
- Assimos D, Krambeck A, Miller NL, et al. Surgical Management of Stones: American Urological Association/Endourological Society Guideline, PART I. J Urol 2016; 196:1153.
- Campschroer T, Zhu Y, Duijvesz D, et al. Alpha-blockers as medical expulsive therapy for ureteral stones. Cochrane Database Syst Rev 2014; :CD008509.
- Manjunath A, Skinner R, Probert J. Assessment and management of renal colic. BMJ 2013; 346:f985.
- Krieger JN, Ross SO, Simonsen JM. Urinary tract infections in healthy university men. J Urol 1993; 149:1046.
- Vorland LH, Carlson K, Aalen O. An epidemiological survey of urinary tract infections among outpatients in Northern Norway. Scand J Infect Dis 1985; 17:277
- Kawashima A, LeRoy AJ. Radiologic evaluation of patients with renal infections. Infect Dis Clin North Am 2003; 17:433.
- Bouyer J, Coste J, Fernandez H, et al. Sites of ectopic pregnancy: a 10 year population-based study of 1800 cases. Hum Reprod 2002; 17:3224.
- Bouyer J, Coste J, Shojaei T, et al. Risk factors for ectopic pregnancy: a comprehensive analysis based on a large case-control, population-based study in France. Am J Epidemiol 2003; 157:185.
- Alkatout I, Honemeyer U, Strauss A, et al. Clinical diagnosis and treatment of ectopic pregnancy. Obstet Gynecol Surv 2013; 68:571.
- Hahn SA, Lavonas EJ, Mace SE, et al. Clinical policy: Critical issues in the initial evaluation and management of patients presenting to the emergency department in early pregnancy. Ann Emerg Med 2012; 60:381-390.
- McWilliams GD, Hill MJ, Dietrich CS 3rd. Gynecologic emergencies. Surg Clin North Am 2008; 88:265.
- Argenta PA, Yeagley TJ, Ott G, Sondheimer SJ. Torsion of the uterine adnexa. Pathologic correlations and current management trends. J Reprod Med 2000; 45:831.
- Houry D, Abbott JT. Ovarian torsion: a fifteen-year review. Ann Emerg Med 2001; 38:156.
- Lareau SM, Beigi RH. Pelvic inflammatory disease and tubo-ovarian abscess. Infect Dis Clin North Am 2008; 22:693.
- Wiesenfeld HC, Sweet RL. Progress in the management of tuboovarian abscesses. Clin Obstet Gynecol 1993; 36:433.
- Barnes AD. Nonsurgical management of a large hemoperitoneum from a ruptured corpus luteum: A 15-year study. The Female Patient 2005; 30:29.
- Pepe P, Panella P, Pennisi M, Aragona F. Does color Doppler sonography improve the clinical assessment of patients with acute scrotum? Eur J Radiol 2006; 60:120.
- Wampler SM, Llanes M. Common scrotal and testicular problems. Prim Care 2010; 37:613.
- Harris KM, Braverman AC, Eagle KA, et al. Acute aortic intramural hematoma: an analysis from the International Registry of Acute Aortic Dissection. Circulation 2012; 126:S91.
- Erbel R, Aboyans V, Boileau C, et al. Corrigendum to: 2014 ESC Guidelines on the diagnosis and treatment of aortic diseases. Eur Heart J 2015; 36:2779.
- Pape LA, Awais M, Woznicki EM, et al. Presentation, Diagnosis, and Outcomes of Acute Aortic Dissection: 17-Year Trends From the International Registry of Acute Aortic Dissection. J Am Coll Cardiol 2015; 66:350.
- Maraj R, Rerkpattanapipat P, Jacobs LE, et al. Meta-analysis of 143 reported cases of aortic intramural hematoma. Am J Cardiol 2000; 86:664.
- Hidai H, Kinoshita Y, Murayama T, et al. Rupture of renal artery aneurysm. Eur Urol. 1985. 11(4):249-53.
- Down LA, Papavassiliou DV, O’Rear EA. Arterial deformation with renal artery aneurysm as a basis for secondary hypertension. Biorheology. 2013 Jan 1. 50(1):17-31
- Bourgault M, Grimbert P, Verret C, et al. Acute renal infarction: a case series. Clin J Am Soc Nephrol 2013; 8:392.
- Oh YK, Yang CW, Kim YL, et al. Clinical Characteristics and Outcomes of Renal Infarction. Am J Kidney Dis 2016; 67:243.
- Bourgault M, Grimbert P, Verret C, et al. Acute renal infarction: a case series. Clin J Am Soc Nephrol 2013; 8:392.
- Hazanov N, Somin M, Attali M, et al. Acute renal embolism. Forty-four cases of renal infarction in patients with atrial fibrillation. Medicine (Baltimore) 2004; 83:292.
- Lawrence YR, Pokroy R, Berlowitz D, et al. Splenic infarction: an update on William Osler’s observations. Isr Med Assoc J 2010; 12:362.
- Nores M, Phillips EH, Morgenstern L, Hiatt JR. The clinical spectrum of splenic infarction. Am Surg 1998; 64:182.
- Markogiannakis H, Messaris E, Dardamanis D, et al. Acute mechanical bowel obstruction: clinical presentation, etiology, management and outcome. World J Gastroenterol 2007; 13:432.
- Catena F, Di Saverio S, Kelly MD, et al. Bologna Guidelines for Diagnosis and Management of Adhesive Small Bowel Obstruction (ASBO): 2010 Evidence-Based Guidelines of the World Society of Emergency Surgery. World J Emerg Surg 2011; 6:5.
- Rodkey GV, Welch CE. Changing patterns in the surgical treatment of diverticular disease. Ann Surg 1984; 200:466.
- Williams GR. Presidential Address: a history of appendicitis. With anecdotes illustrating its importance. Ann Surg 1983; 197:495.
- Lee SL, Walsh AJ, Ho HS. Computed tomography and ultrasonography do not improve and may delay the diagnosis and treatment of acute appendicitis. Arch Surg 2001; 136:556.
- Birnbaum BA, Wilson SR. Appendicitis at the millennium. Radiology 2000; 215:337.
- Guidry SP, Poole GV. The anatomy of appendicitis. Am Surg 1994; 60:68.
- Buschard K, Kjaeldgaard A. Investigation and analysis of the position, fixation, length and embryology of the vermiform appendix. Acta Chir Scand 1973; 139:293.
- Festi D, Sottili S, Colecchia A, et al. Clinical manifestations of gallstone disease: evidence from the multicenter Italian study on cholelithiasis (MICOL). Hepatology 1999; 30:839.
- Trowbridge RL, Rutkowski NK, Shojania KG. Does this patient have acute cholecystitis? JAMA 2003; 289:80.
- Acosta S, Alhadad A, Svensson P, Ekberg O. Epidemiology, risk and prognostic factors in mesenteric venous thrombosis. Br J Surg 2008; 95:1245.
- Harward TR, Green D, Bergan JJ, et al. Mesenteric venous thrombosis. J Vasc Surg 1989; 9:328.
- Laissy JP, Trillaud H, Douek P. MR angiography: noninvasive vascular imaging of the abdomen. Abdom Imaging 2002; 27:488.
- Metlay JP, Schulz R, Li YH, et al. Influence of age on symptoms at presentation in patients with community-acquired pneumonia. Arch Intern Med 1997; 157:1453.
- Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis 2007; 44 Suppl 2:S27.
- Stein PD, Terrin ML, Hales CA, et al. Clinical, laboratory, roentgenographic, and electrocardiographic findings in patients with acute pulmonary embolism and no pre-existing cardiac or pulmonary disease. Chest 1991; 100:598.
- Tintinalli, Judith E. “Aortic Dissection and Aneurysms.” Emergency Medicine: a Comprehensive Study Guide, 5th ed., McGraw-Hill, 1999, pp. 413
- Shanmuganathan K, Mirvis SE, Sherbourne CD, et al. Hemoperitoneum as the sole indicator of abdominal visceral injuries: a potential limitation of screening abdominal US for trauma. Radiology 1999; 212:423.
- Mallick IH, Thoufeeq MH, Rajendran TP. Iliopsoas abscesses. Postgrad Med J 2004; 80:459.
- Stefanich RJ, Moskowitz A. Hip flexion deformity secondary to acute pyogenic psoas abscess. Orthop Rev 1987; 16:67.






