Author: Brit Long, MD (@long_brit, EM Chief Resident at SAUSHEC, USAF) // Edited by: Alex Koyfman, MD (@EMHighAK, EM Attending Physician, UTSW / Parkland Memorial Hospital) & Justin Bright, MD (@JBright2021)
You are in the midst of caring for a 62 year-old male who is tachycardic with HR 120, BP 88/42, T 101.2 oF, RR 26, and SpO2 98%. He was brought in by EMS for fever and myalgias, and with one look at his vital signs, he triggered the protocol for SIRS.
Your initial exam showed similar vital signs, with dry mucous membranes but otherwise normal HEENT exam, clear lungs, normal mental status, nontender abdomen, normal skin and genitourinary exams, and normal extremities/back. Due to his vital signs and SIRS criteria, you were concerned and ordered CBC, RFP, LFT, lactate, blood cultures, urinalysis/culture, and chest xray. You started 1 L NS, and his VS did not improve.
A lactate returns at 5.2, with WBC of 14,000 and platelets 88,000. Chest Xray and urine are normal. You start empiric antibiotics for presumed sepsis, provide another liter of NS, but have no improvement of VS. Your US shows a flat IVC with respiratory variation after the 2L NS. You provide another 2L of NS, start norepinephrine through a peripheral line, and begin your preparation for a central line. However, his VS do not improve, even with the norepinephrine. His RR is increasing, and his capillary refill and mental status are beginning to decline.
This patient meets criteria for septic shock unresponsive to fluids and pressors.
What could you be missing? What steps should be taken next?
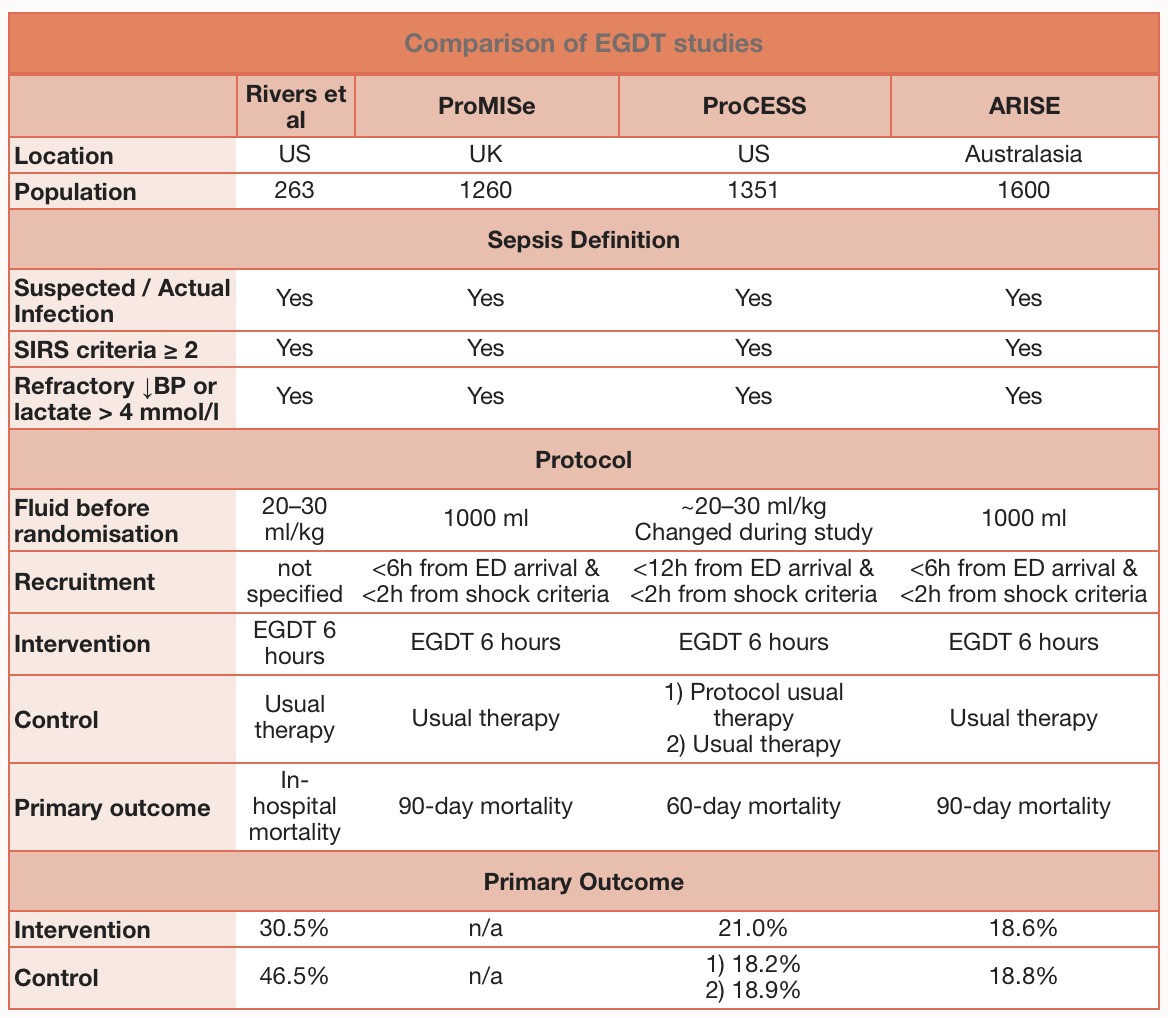
The emergency medicine community has had numerous updates in the literature with ProCESS, ARISE, ProMISe, and TRISS. A summary, from The Bottom Line, is shown below:
Background: Emergency Physicians are well versed in the care of the septic patient, with 3 million visits per year to EDs related to sepsis.1-3 The majority of patients will respond to treatment including antibiotics and fluid resuscitation. However, many of us have cared for the patient who does not respond to our initial treatments. The measures we take at this point can be life-saving, but unfortunately, there is not much literature that provides a well-defined algorithm. This post will seek to educate on what steps we need to take in the ED to avoid the patient deteriorating and potentially dying.
This post will utilize the following for care of the septic shock patient who is unresponsive to treatment: source consideration, assessment for adequate preload and fluid resuscitation, antibiotic coverage and dosing, ultrasound (US) with RUSH protocol and evaluation for reversible causes, vasopressor choice, metabolic/endocrine concerns, abdominal compartment syndrome (ACS), transfusion of blood products, and respiratory status. With a patient unresponsive to your treatments, cognitively going through these issues may save the patient!
The following is a summary of sepsis, severe sepsis, and septic shock:

1. Source control
Identifying the source of infection is paramount in care for the sepsis patient in order to adequately treat the patient. If the patient is not responding to your treatment, question whether you have discovered the source. Grouping sources using LUCCASSS can be helpful: Lung, Urine, CNS, Cardiac, Abdomen, Skin, Spine, Septic Arthritis. This targets the likely sources. Lung followed by urine is most likely, so ensure you obtain a chest Xray and urinalysis.3-5
L: Lung infection involving pneumonia is the most common source. Older patients or neutropenic patients may not display an infiltrate early in pneumonia, so if respiratory symptoms are present, treat empirically for pneumonia. Look for tachypnea, as this is often the earliest finding of sepsis in elderly and immunosuppressed patients.6,7
U: Urine is the second most common source.4,5 Obtaining a urinalysis can be an issue in the nursing home patient, as asymptomatic bacteriuria is very common in this population. Older adults in the community have rates as high as 10%, while those in nursing homes have rates that can reach 50%! Symptoms include fever, CVA tenderness, new suprapubic pain, hematuria, new/increased incontinence, new/increased urgency, and new/increased frequency. If the patient has symptoms, check a urinalysis or urine dipstick.8 If this is positive, treat for presumed urinary tract infection.
C: CNS infections may be subtle. If the patient is altered, has a headache, or has meningeal signs but is unstable, hold on performing lumbar puncture. Instead, provide empiric coverage for suspected meningitis (Please see prior emDocs post on meningitis: http://www.emdocs.net/meningitis-clinical-pearls-pitfalls/). Be mindful of HSV encephalitis (focal deficit, characteristic rash, elevated LFTs) and be ready to utilize acyclovir. Please see prior emDocs post on HSV encephalitis: http://www.emdocs.net/hsv-encephalitis/
C: Endocarditis can be difficult to diagnose, as patients often present with myalgias and fever. Listen carefully for murmurs on cardiac exam, check for Janeway lesions and Osler nodes, evaluate for splenomegaly, and look for splinter hemorrhages. Unfortunately these classic findings are not common on initial presentation. Pay close attention to endocarditis risk factors such as prosthetic/mechanical heart valve, infection involving multiple organ systems, indwelling lines, hemodialysis, degenerative valvular problems, history of rheumatic heart disease, recent instrumentation, or intravenous drug use. Please see prior emDocs post on a complicated endocarditis case: http://www.emdocs.net/endocarditis-complicated-case/
A: An abdominal source should be suspected with any symptoms such as abdominal pain, obstipation, nausea, vomiting, diarrhea, and in immunosuppressed/older/diabetic patients. Do not hesitate to utilize your ultrasound to evaluate the gallbladder in these patients, as they may not display classic symptoms of cholecystitis. The majority of elderly/diabetic patients with acute biliary disease will have no fever or vomiting, and 5% will present with only mental status changes! A normal WBC is present in 40% of patients. If the disease progresses to emphysematous cholecystitis, mortality reaches upwards of 25%! If US is normal, consider CT abdomen/pelvis.6,7
S: It is essential to complete a full skin exam including the back, buttocks, and lower extremities. Cellulitis may be subtle, especially in elderly patients, and you must not miss necrotizing fasciitis!6 Also evaluate for the presence of central line or PORT. If an intravascular device is present, look for erythema and warmth over the site and consider removing the line if either is present. Please see prior emDocs post on necrotizing fasciitis: http://www.emdocs.net/necrotizing-fasciitis/
S: Epidural abscess, osteomyelitis, and discitis can present with myalgias and fever, so you must beware of the patient with back pain, fever, neurologic symptoms and risk factors similar to those of endocarditis (see above). Please see prior emDocs post on epidural abscess: http://www.emdocs.net/spinal-epidural-abscess/ and osteomyelitis: http://www.emdocs.net/osteomyelitis-making-diagnosis/
S: Septic Arthritis can affect any joint, but the knee is most common. S. aureus and N. gonorrhea are the most common bacteria, and this infection is more common in patients with prosthetic joint, rheumatoid arthritis, HIV, and recent surgery. Only 50% of patients will have fever, and WBC, ESR, and CRP do not have cutoffs that are sensitive enough to exclude the diagnosis. You must evaluate the joints for redness, effusion, or pain. If there are concerning clinical signs/symptoms, perform arthrocentesis. Synovial lactate has shown promise in diagnosis.9 Please see prior emDocs post on septic arthritis:
http://www.emdocs.net/septic-joint-reminders-updates-and-pitfalls/
2. Assessment of Fluid Status and Fluid Loading
An important premise besides source control and antibiotic choice includes ensuring adequate preload, and if fluid tolerant, fluid loading.10-12 Just because the patient is in septic shock does not necessarily mean the patient requires liters and liters of fluid. Septic shock is often a combination of venodilation, arterial dilation, intravascular volume depletion, and sometimes septic cardiomyopathy (studies show a large range of 10-80%!).13,14 Utilizing US to look at the IVC with respiratory variation can be beneficial.13 This is important in order to avoid fluid over-resuscitation.
A great deal of literature exists on fluids in sepsis, and patients in sepsis are often hypovolemic and require rapid fluid resuscitation. Most support the use of crystalloids such as lactated ringers or normal saline in the initial resuscitation.15,16 Avoid pentastarch or hydroxyethyl starch.17 Needless to say, 1L is probably not enough. Ensure you are properly loading the tank. US can be helpful for this in terms of evaluating fluid tolerance. Fluid responsiveness is a different issue and can be evaluated with passive leg raise, pulse pressure variation, and other markers. Static targets such as CVP possess poor ability to predict volume responsiveness.18 When providing fluid, give well-defined boluses such as 500ml or 1L at a time and evaluate the IVC using US, mental status, distal perfusion, blood pressure, and urine output. A combination is better than one marker alone.2,18
3. Antibiotic Choice and Dosing
Every hour of delay can increase mortality by 7.6%, and time to antibiotic initiation has been shown to be the strongest predictor of patient outcome!19,20 In a Canadian study involving patients with meningitis, a delay of antibiotics by 6 hours increased mortality 8 fold.21 Factors involved in antibiotic selection include assessment of the likely source, host/patient factors, and local antibiotic resistance patterns (consulting the local antibiogram is essential). Gram-positive infections account for over 50% of cases, with gram-negative accounting for approximately 35%. The important aspect is covering for suspected organisms. If the patient is unresponsive, evaluate the antibiotic regimen and ensure you have adequate coverage. Broadening antibiotics early is better than too narrow of coverage.21,22
Targeted antibiotic provision early is essential, but there are multiple factors in antibiotic choice including the patient, the microbe(s), pharmacokinetic and pharmacodynamic effects, and suspected source of infection.21,22
Factors in Treatment with Antibiotics
Patient factors – Chronic immunosuppression, neutropenia, alcoholism, active malignancy, older age, transplant related, intravenous drug use, instrumentation, nursing facility, indwelling device, and hemodialysis impact the antibiotic choice and dosing. These factors increase the risk of multidrug resistant organisms (MDRO).2,21,22
Microbe factors – Consulting your institution’s antibiogram is vital to adequately treat your suspected source. Drug resistant S. pneumonia and methicillin resistant S. aureus have increased drastically in the recent years. Risk factors for MDRO include recent hospitalization, nursing home resident, prior infection with MDRO, and recent antibiotic treatment.2,21,22
Pharmacodynamic and Pharmacokinetic Effects – When providing antibiotics to a patient with sepsis and shock, several pharmacokinetic and pharmacodynamic processes affect the distribution, absorption, metabolism, and disposition of the medications. First, for the pharmacokinetic effects, fluid shifts can lead to increased volume of distribution of hydrophilic antimicrobials such as aminoglycosides, beta-lactams, vancomycin, and linezolid. The vasodilation and interstitial edema that sets in cause large changes in volume of distribution, thus requiring increased initial dosing of these medications. For antibiotics that are highly protein bound to serum albumin, hypoalbuminemia results in higher than normal unbound fractions of medication, which is an advantage for these patients.24-27
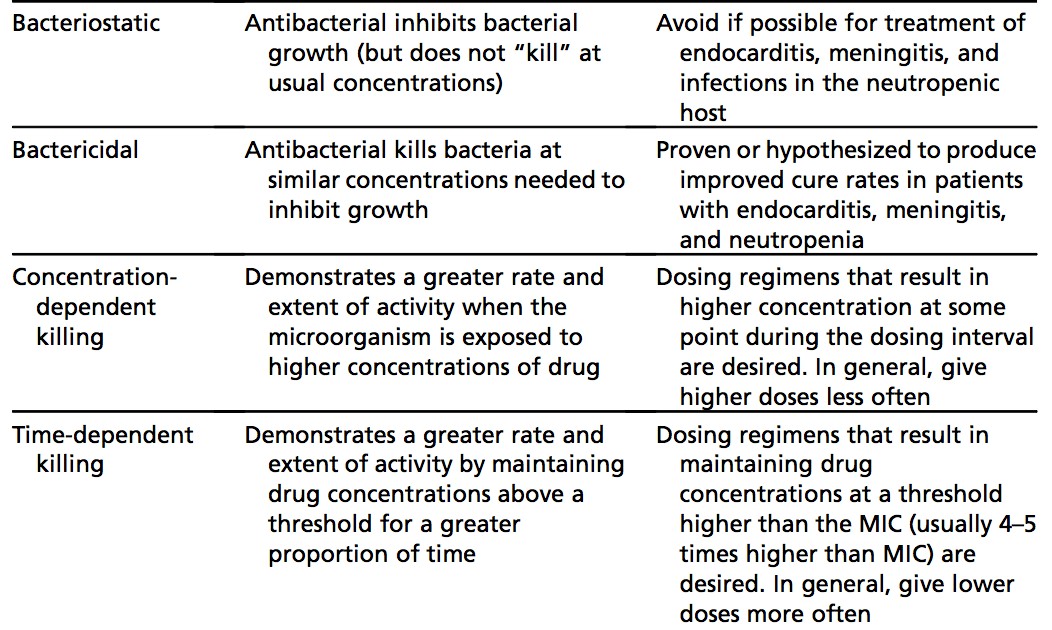
On the pharmacodynamics side, antibiotics that require higher concentrations to kill microbes than inhibit growth are bacteriostatic, while those that kill at concentrations similar to those required to inhibit growth are bactericidal. This is important to know in certain subpopulations such as endocarditis, meningitis, and severely immunocompromised patients such as in neutropenia, where bactericidal agents are necessary.24-27
Concentration-dependent antibiotics include aminoglycosides, as these medications have a greater rate and extent of killing with higher concentrations, so one high dose of the medication is given. On the other hand, time-dependent antimicrobials like penicillin, carbapenems, and cephalosporins kill microbes based on the time above the minimum inhibitory concentration, which requires more frequent dosing.24-27
Base your antibiotic coverage on local antibiogram, suspected infection source and avoid recently used antibiotics.2,20,22 Below is a listing of empiric therapy:
- Vancomycin 25-30mg/kg IV (max 2g) AND
- Zosyn 4.5g IV or Ceftriaxone 2g IV or Cefepime 2g IV or Meropenem 500mg-1g IV or Imipenem 500mg IV
- For additional gram-negative coverage in septic shock add: Tobramycin 5mg/kg, Gentamicin 5mg/kg IV, OR Levofloxacin 750mg IV.
- If vancomycin resistant enterococcus, use Linezolid 600mg IV
- If multi-drug resistant Pseudomonas, Acinetobacter, or Klebsiella is suspected, use Colistimethate 5mg/kg ideal body weight IV.
4. Ultrasound (US) for RUSH and Reversible Causes
US is valuable in the setting of a patient unresponsive to your treatment for several reasons. First, it provides a framework for the rapid and systematic evaluation of the cause for hypotension via a designated protocol (such as the RUSH protocol). Second, it evaluates for potentially reversible causes of shock such as pneumothorax, pulmonary embolism, pericardial effusion, hemorrhage, dissection, poor LV contractility, and pleural effusion. Finally, as discussed previously, it helps you evaluate part of “the tank,” or the IVC for fluid tolerance as discussed above.
I will cover the RUSH protocol here, but other US protocols exist. RUSH is “Rapid Ultrasound for Shock” and evaluates the pump (cardiac), the tank (volume), and the pipes (vascular) using phased array and linear probes. Please see prior emDocs post on the RUSH protocol: http://www.emdocs.net/rush-protocol/
In summary, your pump evaluation looks for pericardial effusion, assesses LV contractility, and assesses RV strain. The tank evaluation looks at the IVC, FAST and lung US, assesses for pleural effusion/pulmonary edema, and evaluates for pneumothorax. The pipe evaluation looks at the aorta for AAA, evaluates for aortic dissection, and evaluates for DVT.28,29
5. Choice of Vasopressor and need for Balanced Resuscitation
Your first-line vasopressor should be norepinephrine at 2-4mcg/min (usual dosage is 8-12mcg/min, but no true maximum on dosing), and dopamine should be avoided in the adult population (though it is still used first line in pediatrics).2 Early use of norepinephrine in septic shock improves venous return and preload while fluid loading the patient. Again, septic shock may not be only due to hypovolemia, and norepinephrine has numerous beneficial effects in early septic shock such as venoconstriction, arterial constriction, positive inotropy, improved cardiac output, and renal perfusion. Recent literature supports a “balanced resuscitation” approach with norepinephrine augmenting preload while volume is administered. This should be started early in combination with fluid resuscitation. Multiple studies have found improved mortality in early norepinephrine provision, as opposed to delayed after fluid loading.30-32 Norepinephrine can be given peripherally for a limited time while volume challenging.33
If you are using norepinephrine with fluid loading and perfusion remains poor, evaluate the heart and contractility with your trusty US. If the heart has poor contractility, consider starting epinephrine at 1-4mcg/min (usual dose varies from 2-20mcg/min), but if the heart has “good squeeze,” use vasopressin at a fixed dose of 0.04 units/min.34 The addition of vasopressin to norepinephrine in the VASST trial found no overall mortality difference in groups with vasopressin versus norepinephrine.35 However, it did show that patients randomized to vasopressin had significantly decreased norepinephrine requirements. This beneficial effect was consistent in both the more and the less-severe shock subgroups, as well as demonstrating improved renal outcomes.35,36 While vasopressin is at the fixed dose and the vitals have stabilized, attempt to titrate down the norepinephrine.2
An inotrope can be added in the presence of myocardial dysfunction (elevated cardiac filling pressures and low cardiac output) or ongoing signs of hypoperfusion despite adequate fluid loading. Dobutamine at 2.5mcg/kg/min IV titrated by 2.5mcg/kg/min to max of 20mcg/kg/min can be used.2,20 Ensure you have a vasopressor on board before you begin dobutamine, which can cause peripheral vasodilation due to beta-agonism and a further drop in blood pressure.
6. Metabolic Concerns
A number of metabolic complications can occur in the setting of septic shock and reduce clinical response to treatment. First, adequate calcium stores are vital for maintaining vascular tone and myocardial function. Thus, check a calcium level (ionized is preferable).34 If the ionized Ca is <1.1mmol/L (4.8mg/dL), or if the patient is symptomatic (seizures, long QT interval, cardiac arrhythmia, positive Chvostek/Trousseau signs), then provide Ca gluconate 1amp (peripheral line) or Ca chloride 1 amp (central line). Correcting the calcium can improve the patient’s clinical course.
Addressing adrenal insufficiency is vital. This condition is commonly associated with fluid and catecholamine resistant septic shock. If the patient is resistant to fluids and vasopressors, provide stress dose steroids of hydrocortisone 50-100mg IV. These patients may not have the classic hyperkalemia and hyponatremia unfortunately, making diagnosis even more difficult. The cosyntropin test is no longer recommended to evaluate for adrenal insufficiency.2,20 Please see a prior emDocs post on adrenal insufficiency: http://www.emdocs.net/adrenal-crisis-in-the-ed/
Steroids have been used for years in treating sepsis, with studies dating back to the 1960s. Two studies after 2002 focused on the provision of physiologic doses of steroids. Annane in 2002 provided steroids to patients with vasopressor resistant septic shock in the first 8 hours of presentation and found an improved adjusted mortality rate in the steroid group, while the CORTICUS trial enrolled patients with sepsis with evidence of shock that persisted for 1-72 hours, and found no difference between groups given hydrocortisone versus placebo.37,38 A meta analysis in 2009 found no mortality difference, but decreased time to resolution of vasopressor dependent shock and no increased risk of superinfection. It appears that steroids improve hemodynamic stability and allow earlier removal of vasopressors, decreasing the duration of shock.39
In summary, in patients on two vasopressors but still with poor perfusion, consider adrenal insufficiency and provide hydrocortisone 50-100mg IV X1. Both the Annane study and CORTICUS support corticosteroids decreasing the duration of vasopressor dependent shock. These studies do not support increased risk of superinfection. Prompt resolution of shock will improve organ perfusion and function.
Thyroid disease can also contribute, so do not hesitate to check TSH. Myxedema coma or thyroid storm can be triggered by infection that results in sepsis. If the TSH is low and the patient is tachycardic, febrile, and tremulous, consider thyroid storm. If the TSH is high and the patient hypothermic, bradycardic, and hypotensive, think hypothyroidism. These patients will also be hyponatremic, have altered mental status, and have delayed return of deep tendon reflexes. In this case, provide levothyroxine at 500-800 micrograms IV (T3 is controversial, but your ICU may want this). Providing thyroid hormone and vasopressors at the same time can result in severe arrhythmias due to the vasopressor and inotropic effects of thyroid hormone, so carefully monitor the patient as you begin the thyroid hormone replacement.39 Elderly patients and patients with history of coronary disease require close monitoring while receiving T4.
7. Abdominal Compartment Syndrome (ACS)
ACS is defined by intra-abdominal pressures greater than 20mmHg with concomitant organ dysfunction. This affects up to 7% of critically ill patients! It is broken into primary (abdominal/pelvic injury) or secondary (no abdominal/pelvic injury). Risk factors include massive fluid resuscitation, burns, gastroparesis, ileus, obstruction, and acidosis. ACS can worsen intracranial pressure, reduce preload by compressing the IVC, cause compression atelectasis and worsen ventilation/oxygenation, cause bowel ischemia, and reduce renal blood flow. Diagnosis relies on bladder pressures. Definitive treatment is laparotomy, but temporizing measures include nasogastric and rectal tubes, providing prokinetic agents, draining ascetic fluid or abscess, neuromuscular blockage, and increased analgesia/sedation.41,42
8. Transfusion of Blood Products
With no treatment response, consider if the patient could be surreptitiously bleeding. Check the stool for blood if suspicious. With the recent TRISS trial demonstrating no mortality difference at 90 days in septic shock patients with hemoglobin of 7g/dL vs. 9g/dL and transfusion complication risk, a threshold of 7g/dL is recommended unless the patient is experiencing myocardial ischemia, severe hypoxemia, or severe hemorrhage.43 Fresh frozen plasma should not be used just to correct laboratory abnormalities. Platelets should be used only if the count is < 10,000/mm3 without bleeding, < 20,000/mm3 if risk of bleeding present, or < 50,000/mm3 and bleeding is present/invasive procedure planned. Erythropoietin is not recommended.2 With that being said, please evaluate the patient closely for signs of bleeding. If needed, transfuse products and closely evaluate the patient for any response.34
9. Respiratory Status
Septic shock is often associated with metabolic acidosis, resulting in respiratory compensation. This respiratory compensation with tachypnea requires increased energy devotion to the muscles of respiration. If the patient is tiring or tachypneic, consider taking away this requirement by intubating the patient and placing on mechanical ventilation.34 Remember, ARDSnet respiratory settings should be utilized, with tidal volumes of 6-8ml/kg ideal body weight, not actual body weight.44 By removing the strenuous respiratory effort, the patient can provide ATP where it’s truly needed to fight off infection and perfuse distal organs.
Summary
In patients unresponsive to initial treatments, consider the following:
- Source identification – LUCCASSS.
- Ensure adequate preload, and determine whether further fluid resuscitation warranted.
- Antibiotic regimen coverage and dosage – beware of patient/microbe factors, as well as pharmacodynamics and pharmacokinetic drug effects.
- US for other causes using RUSH – Heart, IVC, FAST, Lungs.
- Balanced resuscitation – early vasopressor provision, which improves venoconstriction, arterial constriction, positive inotropy, improved cardiac output, and renal perfusion.
- Metabolic/Endocrine concerns – hypocalcemia, adrenal insufficiency, thyroid disease (myxedema coma).
- Abdominal Compartment Syndrome – measure bladder pressure.
- Transfusion of Products – look for bleeding.
- Respiratory Status – if declining, take airway with intubation, place on low tidal volume settings.
Sources/Further Reading:
- Elixhauser A, Friedman B, Stranges E. Septicemia in U.S. Hospitals, 2009. Agency for Healthcare Research and Quality, Rockville, MD. http://www.hcup-us.ahrq.gov/reports/statbriefs/sb122.pdf.
- Dellinger RP, Levy MM, Rhodes A, et al. Surviving sepsis campaign: international guidelines for management of severe sepsis and septic shock: 2012. Crit Care Med 2013;41:580–637.
- Strehlow MC, Emond SD, Shapiro NI, et al. National study of emergency department visits for sepsis, 1992 to 2001. Ann Emerg Med 2006;48:326–31.
- Brun-Buisson C, Doyon F, Carlet J, et al. Incidence, risk factors, and outcome of severe sepsis and septic shock in adults. A multicenter prospective study in intensive care units. French ICU Group for Severe Sepsis. JAMA 1995;274:968.
- Canetnacci MH, King K. Severe Sepsis and Septic Shock: Improving Outcome in the Emergency Department. Emerg Med Clin N Am 2008;26:603–623.
- Samaras, N, Chevalley, T, et al. “Older patients in the emergency department: a review.” Ann Emerg Med 2010;56:261-269.
- Ragsdale, L, Southerland, L. “Acute abdominal pain in the older adult.” Emerg Med Clin N Am 2011;29:429–448.
- Shenvi C. Uncomplicated Urinary Tract Infection in Older Adults: Diagnosis and Treatment (Part 1). http://www.aliem.com/uncomplicated-urinary-tract-infection-older-adults-diagnosis-treatment-1/#citation1
- Carpenter CR, Schuur JD, Everett WW, Pines JM. Evidence-based Diagnostics: Adult Septic Arthritis. Acad Emerg Med 2011;18(8):781-796.
- Rivers E, Nguyen B, Havstad S, et al. Early goal-directed therapy in the treatment of severe sepsis and septic shock. N Engl J Med 2001;345:1368.
- ProCESS Investigators, Yealy DM, Kellum JA, et al. A randomized trial of protocol-based care for early septic shock. N Engl J Med 2014;370:1683.
- ARISE Investigators, ANZICS Clinical Trials Group, Peake SL, et al. Goal-directed resuscitation for patients with early septic shock. N Engl J Med 2014;371:1496.
- Rudiger A, Singer M. Mechanisms of sepsis-induced cardiac dysfunction. Crit Care Med 2007;35:1599–608.
- Bouhemad B, Nicolas-Robin A, Arbelot C, et al. Isolated and reversible impairment of ventricular relaxation in patients with septic shock. Crit Care Med 2008;36:766–74.
- Finfer S, Bellomo R, Boyce N, et al. A comparison of albumin and saline for fluid resuscitation in the intensive care unit. N Engl J Med 2004;350:2247.
- Caironi P, Tognoni G, Masson S, et al. Albumin replacement in patients with severe sepsis or septic shock. N Engl J Med 2014;370:1412.
- Perner A, Haase N, Guttormsen AB, et al. Hydroxyethyl starch 130/0.42 versus Ringer’s acetate in severe sepsis. N Engl J Med 2012;367:124.
- Weingart S. Spontaneously Breathing Patients – EAT IT STONE. http://emcrit.org/podcasts/ivc-ultrasound-for-fluid-tolerance-in-spontaneously-breathing-patients/
- Kumar A, Roberts D, Wood KE, et al. Duration of hypotension before initiation of effective antimicrobial therapy is the critical determinant of survival in human septic shock. Crit Care Med 2006;34:1589–96.
- Larche J, Azoulay E, Fieux F, et al. Improved survival of critically ill cancer patients with septic shock. Intensive Care Med 2003;29:1688–95.
- Proulx N, Frechette D, Toye B, et al. Delays in the administration of antibiotics are associated with mortality from adult acute bacterial meningitis. QJM 2005;98:291–8.
- Green RS, Gorman SK. Optimizing initial antibiotic delivery for adult patients with severe sepsis and septic shock in the emergency department. EM Critical Care 2012;(Suppl):1–16.
- Leekha S, Terrell CL, Edson RS. General principles of antimicrobial therapy. Mayo Clin Proc 2011;86:156–67.
- Nicolau DP. Optimizing antimicrobial therapy and emerging pathogens. Am J Manag Care 1998;4(10 Suppl 2):S525–30.
- Varghese JM, Roberts JA, Lipman J. Pharmacokinetic and pharmacodynamic issues in the critically ill with severe sepsis and septic shock. Crit Care Clin 2011;27:19–34.
- Slavik RS, Jewesson PJ. Selecting antibacterials for outpatient parenteral antimicrobial therapy: pharmacokinetic-pharmacodynamic considerations. Clin Pharmacokinet 2003;42:793–817.
- Pankey GA, Sabath LD. Clinical relevance of bacteriostatic versus bactericidal mechanisms of action in the treatment of Gram-positive bacterial infections. Clin Infect Dis 2004;38:864–70.
- Seif D, Perera P, et al. Review Article: Bedside Ultrasound in Resuscitation and the Rapid Ultrasound Shock Protocol. Critical Care Research and Practice. Volume 2012. doi:10.1155/2012/503254.
- Weingart S, Duque D, Nelson B. The RUSH Exam: Rapid Ultrasound for Shock and Hypotension. http://emcrit.org/rush-exam/
- Waechter J, Kumar A, et al. Interaction between fluids and vasoactive agents on mortality in septic shock: a multicenter, observational study. Crit Care Med 2014 Oct;42(10):2158-68
- Morimatsu H, Singh K, et al. Early and exclusive use of norepinephrine in septic shock. Resuscitation 2004 Aug;62(2):249-54.
- Bai X, Yu W, et al. Early versus delayed administration of norepinephrine in patients with septic shock. Crit Care 2014 Oct 3;18(5):532.
- Weingart S. Podcast 107 – Peripheral Vasopressor Infusions and Extravasation. http://emcrit.org/podcasts/peripheral-vasopressors-extravasation/
- Herbert M, Weingart S, Orman R. Sepsis: A Review of Current Therapy. EM:RAP. (October 2012). https://www.emrap.org/episode/october2012/sepsisareviewof
- Russell JA, Walley KR, Singer J, et al. Vasopressin versus norepinephrine infusion in patients with septic shock. N Engl J Med 2008;358:877–87.
- Gordon AC, Wang N, Walley KR, et al. The cardiopulmonary effects of vasopressin compared with norepinephrine in septic shock. Chest 2012;142:593–605.
- Annane D, Sebille V, Charpentier C, et al. Effect of treatment with low doses of hydrocortisone and fludrocortisone on mortality in patients with septic shock. JAMA 2002;288:862–71.
- Sligl WI, Milner DA Jr, et al. Safety and efficacy of corticosteroids for the treatment of septic shock: A systematic review and meta-analysis. Clin Infect Dis 2009 Jul;49(1):93-101.
- Klubo-Gwiezdzinska J, Wartofsky L. Thyroid emergencies. Med Clin North Am 2012 Mar;96(2):385-403.
- Rehman SU, Cope DW, Senseney AD, et al. Thyroid disorders in elderly patients. South Med J 2005 May;98(5):543-9.
- Faisal Al-Mufarrej, et al. Understanding Intra-Abdominal Hypertension: From the Bench to the Bedside. J Intensive Care Med June 2012;27(3):145-160.
- Kirkpatrick, et al. Intra-abdominal hypertension and the abdominal compartment syndrome: updated consensus definitions and clinical practice guidelines from the World Society of the Abdominal Compartment Syndrome. Intensive Care Med 2013 Jul;39(7):1190–1206.
- Holst, LB, et al. Lower versus Higher Hemoglobin Threshold for Transfusion in Septic Shock N Engl J Med 2014;371:1381-1391.
- The Acute Respiratory Distress Syndrome Network. Ventilation with Lower Tidal Volumes as Compared with Traditional Tidal Volumes for Acute Lung Injury and the Acute Respiratory Distress Syndrome. N Engl J Med 2000;342:1301-1308.







5 thoughts on “The sepsis patient not improving after IV fluids and resuscitation: What should be considered? How can we improve?”
Good review. Remember that the when we talk about fluid resuscitation, the Surviving Sepsis Guidelines recommend 30cc/kg of crystalloid in the first 3-6 hours of recognition of severe sepsis or septic shock. I think many of us default to “give 2L” and think patients have received adequate fluid when in fact, given the weight of many people in America, 2L is nowhere near the 30cc/kg recommended. This becomes especially interesting and important with patients perceived at risk for volume overload (i.e, those with ESRD on HD or those with a pre-existing diagnosis of CHF). There is often hesitance to provide these specific patients with fluid in fear that they may become overloaded and require intubation. It is important to remember however, that there are not separate guidelines for the resuscitation of these patients and we may be doing them more harm by withholding or giving inadequate amount of fluids especially since patients with ESRD and HF at baseline have a much higher rate of mortality independent of being in septic shock.
Also have to keep in mind that patients with known right-sided heart failure and/or history of pulmonary hypertension will likely have very elevated right atrial pressures and assessment of the IVC in these patients will give you a false sense of adequate resuscitation as it will be full. IVC assessment for collapsibility as a target for volume resuscitation is of little use in this subset of patients and fluids should instead be targeted to other parameters (i.e., urine output, improvement in mentation, oxygenation, lactate clearance, etc).
Pingback: Sepsis: Failed Fluid Resuscitation | CritErEd
Pingback: emDOCs.net – Emergency Medicine EducationMimics of Sepsis: What do ED Physicians Need to Know? - emDOCs.net - Emergency Medicine Education
Pingback: emDOCs.net – Emergency Medicine EducationThe EM Educator Series: When sepsis care becomes not so straightforward - emDOCs.net - Emergency Medicine Education